Key takeaways:
~ The thyroid is a master regulator that controls many of your body’s systems including metabolism, body temperature, heart rate, breathing, and body weight.[ref]
~ There are two major forms of thyroid hormone: T4 and T3.
~ Your genes impact how your body produces and converts T4 to T3, the production of TSH, and your susceptibility to autoimmune thyroid problems.
Members will see their genotype report below, plus additional solutions in the Lifehacks section. Consider joining today.
Thyroid Hormones: Genetic polymorphisms, hypothyroidism, and conversion of thyroid hormones
Thyroid hormone levels play a vital role in how you feel and in your overall health and well-being. People often think of the thyroid in terms of metabolism and weight, but your thyroid hormones also affect your body temperature, gut health, muscle energy, heart rate, skin health, bone health, and immune health.
Why is this important now? We have an epidemic of hypothyroidism. In 2016, the #1 prescribed medication in the US was thyroid medication (Synthroid), with 123 million people taking the drug.[ref]
If you have thyroid problems, learning which genetic variants you carry may be a way to shed some light on what is going on with your thyroid.
Knowing where your genetic susceptibilities lie can help you figure out (along with your doctor) the best way to solve the problem.
Hypothyroidism vs hyperthyroidism:
Your body needs the right amount of thyroid hormones, at the right time.
- Hypothyroidism is caused by too little thyroid hormone;
- Hyperthyroidism is caused by too much thyroid hormone.
Symptoms of hypothyroidism include:
- Fatigue
- Feeling cold
- Weight gain
- Hair loss, puffiness
- Constipation
- Poor memory
- Slow pulse
- Lack of menstrual cycle
How is thyroid production regulated?
The hypothalamus, a region in the brain, and the pituitary gland control the thyroid gland’s rate of producing and releasing thyroid hormone.
- The hypothalamus releases thyrotropin-releasing hormone (TRH), which signals to the pituitary gland.
- The pituitary then creates and releases thyroid-stimulating hormone (TSH).
TSH then travels to the thyroid gland to signal for the production of thyroxine (T4) and triiodothyronine (T3).
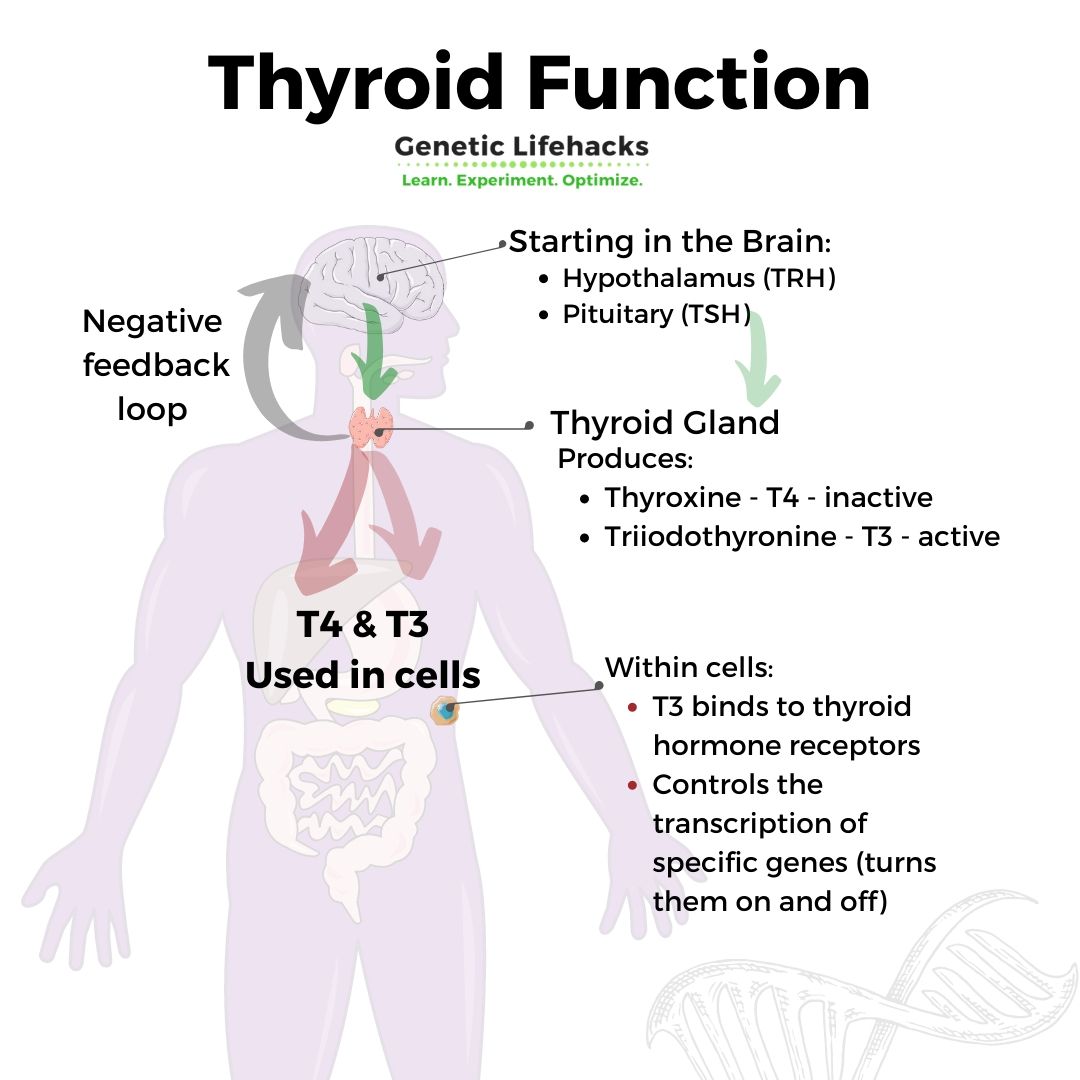
Balance between T3 and T4:
The balance of the two main types of thyroid hormones, T4 and T3, is important.
The thyroid gland produces and releases about 4x more T4 than T3.
- T3 is the active form of the thyroid hormone that your body uses
- T4 is the inactive form that can be converted into T3 when needed in your cells.
T4, the inactive form, is converted to the active T3 in your tissues and organs with the help of specific enzymes, called deiodinases.
Genetic variants in the deiodinase genes (DIO1, DIO2, and DIO3) affect how much T3 is likely to be produced from T4.
More on the DIO1 & DIO2 genes: The deiodinase 1 (DIO1) gene encodes a protein that converts T4 (inactive) to T3 (active). The enzyme is involved in the degradation of both T3 and T4 in the liver, kidney, thyroid, and pituitary gland, which controls overall hormone levels. Both iodine and selenium are cofactors in these reactions.[ref]
DIO2 (deiodinase 2) is also involved in converting T4 to T3, mainly in the skeletal muscles, central nervous system, pituitary, thyroid, heart, and brown fat.[ref] Some researchers think that hypothyroid individuals with DIO2 polymorphisms may benefit from a combination of T4 and T3.[ref]
Iodine in thyroid hormone:
Iodine molecules are an essential part of the T4 and T3 hormones. There are specific transporters within the thyroid gland to move iodine (in the form of iodide) into the follicular cells where the T3 and T4 are produced. More on this in the genetics section below.
The 4 in T4 relates to it containing 4 iodines, and the 3 in T3 refers to it containing 3 iodines.
Feedback loops regulate thyroid hormone levels:
T3 levels are strongly regulated by a feedback loop. Too much active T3 in cells will cause enzymes to inactivate the T3 into reverse T3 (rT3).[ref]
Thyroid hormone levels are an intricate balance between the production of T4, conversion to T3, inactivation to rT3, TSH levels, and the feedback loops controlling TRH and TSH.
In the liver, Liver X receptors are activated by thyroid hormone and control the activation of fat tissue. With less liver X receptor beta activation, more T3 remains in the system and drives the browning of white adipose tissue. Brown adipose tissue is more metabolically active, producing heat, and is linked with being less likely to be obese. [ref]
What does thyroid hormone do inside a cell?
Within cells, the thyroid hormone crosses into the nucleus and binds to a thyroid hormone receptor.
Thyroid hormone receptors then control the transcription of specific genes. Essentially, they turn on a gene so that whatever protein that gene codes for will get made.[ref]
In different cell types, the thyroid hormone receptors (THR) will control the production of different proteins.
- For example, T3 can enter the cell nucleus and bind to the thyroid hormone receptor that controls the liver’s transcription and production of fatty acids, called de novo lipogenesis.
- Thyroid hormone receptors also regulate the creation of mitochondria and the transcription of some genes within the mitochondria.[ref]
Going a little deeper:
The thyroid hormone receptors in the cell nucleus don’t act alone. They are often bound with a retinoic acid receptor, which vitamin A activates.
The thyroid hormone receptors also need zinc in how they bind to the DNA. It makes it important to have adequate vitamin A and zinc levels – along with producing enough thyroid hormone.[ref]
Let me clarify that there are actually two different thyroid hormone receptors: THR-alpha and THR-beta. While both are located in most types of cells, THR-beta is the major form in the liver and THR-alpha is the major form in the heart cells and the bone.[ref]
Thyroid Stimulating Hormone (TSH):
As mentioned above, TSH is released from the pituitary gland and signals for the thyroid gland to make T4 and T3.
The basic thyroid test most doctors run is to check the TSH level. According to the Cleveland Clinic, normal TSH levels for adults are 0.27 – 4.2 uIU/mL. Higher TSH levels indicate hypothyroidism, while very low TSH levels can indicate hyperthyroidism.
Genetic variants in the TSH-related genes are responsible for approximately 50 – 90% of thyroid hormone variability.[ref] Thus, you can be naturally higher or lower on TSH levels due to genetic variants.
Thyroid Hormone Level Genotype Report:
Members: Log in to see your data below.
Not a member? Join here.
Why is this section is now only for members? Here’s why…
Member Content:
Why join Genetic Lifehacks?
~ Membership supports Genetic Lifehack's goal of explaining the latest health and genetics research.
~ It gives you access to the full article, including the Genotype and Lifehacks sections.
~ You'll see your genetic data in the articles and reports.
Join Here
Lifehacks for thyroid problems:
If you are on thyroid medication or under a doctor’s care, please be sure to talk with your doctor about making any changes, including dietary changes, that could affect your thyroid hormone levels.
5 Supplements to Consider for Thyroid Health:
The following micronutrients and supplements have research showing that they may help thyroid function for some individuals. Talk with your doctor if you have any questions on supplements.
1) Selenium:
Selenium is an essential cofactor in the conversion of T4 to T3. Brazil nuts are a good source of selenium, and supplements are also available. Please note that selenium is not a mineral to go overboard on because it is toxic at higher levels.
Related article: Selenium and Your Genes
2) Melatonin:
The production of melatonin is regulated by TSH. Increasing melatonin increases thyroglobulin. The thyroid gland cells also synthesize melatonin.[ref] You can increase your melatonin levels naturally by blocking blue light at night or synthetically by taking melatonin at night.
Related article: Melatonin: key to health and longevity
Member Content:
Why join Genetic Lifehacks?
~ Membership supports Genetic Lifehack's goal of explaining the latest health and genetics research.
~ It gives you access to the full article, including the Genotype and Lifehacks sections.
~ You'll see your genetic data in the articles and reports.
Join Here
Related Articles and Topics:
Lithium Orotate + B12: Boosting mood and decreasing anxiety, for some people…
For some people, low-dose, supplemental lithium orotate is a game-changer for mood issues when combined with vitamin B12. But other people may have little to no response. The difference may be in your genes.
Is inflammation causing your depression or anxiety?
Research over the past two decades clearly shows a causal link between increased inflammatory markers and depression. Genetic variants in the inflammatory-related genes can increase the risk of depression and anxiety.
Green Smoothie Genes- Oxalates in Your Diet
Green smoothies have been a health fad for quite a while now, but these health drinks can be a double-edged sword for some people due to the high oxalate content. Primary Hyperoxaluria can cause oxalates to build up in the thyroid gland.
Detoxifying Phthalates: Genes and Diet
Plastics are everywhere – and a source of the chemicals that we are exposed to on a daily basis. One component of plastics is a class of compounds referred to as phthalates, which can act as an endocrine disruptor and mimic estrogen.
updated and revised 6/2020
References:
Akçay, Müfide Nuran, and Güngör Akçay. “The Presence of the Antigliadin Antibodies in Autoimmune Thyroid Diseases.” Hepato-Gastroenterology, vol. 50 Suppl 2, Dec. 2003, p. cclxxix–cclxxx.
Ballesteros, Virginia, et al. “Exposure to Perfluoroalkyl Substances and Thyroid Function in Pregnant Women and Children: A Systematic Review of Epidemiologic Studies.” Environment International, vol. 99, Feb. 2017, pp. 15–28. ScienceDirect, https://doi.org/10.1016/j.envint.2016.10.015.
Bunevicius, Adomas, et al. “Common Genetic Variations of Deiodinase Genes and Prognosis of Brain Tumor Patients.” Endocrine, vol. 66, no. 3, Dec. 2019, pp. 563–72. PubMed, https://doi.org/10.1007/s12020-019-02016-6.
Cao, Xinyuan, et al. “Exposure of Pregnant Mice to Triclosan Impairs Placental Development and Nutrient Transport.” Scientific Reports, vol. 7, Mar. 2017, p. 44803. PubMed, https://doi.org/10.1038/srep44803.
da-Silva, Wagner S., et al. “The Small Polyphenolic Molecule Kaempferol Increases Cellular Energy Expenditure and Thyroid Hormone Activation.” Diabetes, vol. 56, no. 3, Mar. 2007, pp. 767–76. PubMed, https://doi.org/10.2337/db06-1488.
Eriksson, Nicholas, et al. “Novel Associations for Hypothyroidism Include Known Autoimmune Risk Loci.” PLoS ONE, vol. 7, no. 4, Apr. 2012, p. e34442. PubMed Central, https://doi.org/10.1371/journal.pone.0034442.
Fernández, Lara P., et al. “New Insights into FoxE1 Functions: Identification of Direct FoxE1 Targets in Thyroid Cells.” PLoS ONE, vol. 8, no. 5, May 2013, p. e62849. PubMed Central, https://doi.org/10.1371/journal.pone.0062849.
Garcia-Marin, R., et al. “Melatonin in the Thyroid Gland: Regulation by Thyroid-Stimulating Hormone and Role in Thyroglobulin Gene Expression.” Journal of Physiology and Pharmacology: An Official Journal of the Polish Physiological Society, vol. 66, no. 5, Oct. 2015, pp. 643–52.
Gudmundsson, Julius, et al. “Common Variants on 9q22.33 and 14q13.3 Predispose to Thyroid Cancer in European Populations.” Nature Genetics, vol. 41, no. 4, Apr. 2009, pp. 460–64. PubMed Central, https://doi.org/10.1038/ng.339.
Inoue, Naoya, et al. “Functional Polymorphisms of the Type 1 and Type 2 Iodothyronine Deiodinase Genes in Autoimmune Thyroid Diseases.” Immunological Investigations, vol. 47, no. 5, July 2018, pp. 534–42. PubMed, https://doi.org/10.1080/08820139.2018.1458861.
Jorde, Rolf, et al. “The Phosphodiesterase 8B Gene Rs4704397 Is Associated with Thyroid Function, Risk of Myocardial Infarction, and Body Height: The Tromsø Study.” Thyroid: Official Journal of the American Thyroid Association, vol. 24, no. 2, Feb. 2014, pp. 215–22. PubMed, https://doi.org/10.1089/thy.2013.0177.
Kampf-Lassin, August, and Brian J. Prendergast. “Photoperiod History-Dependent Responses to Intermediate Day Lengths Engage Hypothalamic Iodothyronine Deiodinase Type III MRNA Expression.” American Journal of Physiology – Regulatory, Integrative and Comparative Physiology, vol. 304, no. 8, Apr. 2013, pp. R628–35. PubMed Central, https://doi.org/10.1152/ajpregu.00577.2012.
Llop, Sabrina, et al. “Association between Exposure to Organochlorine Compounds and Maternal Thyroid Status: Role of the Iodothyronine Deiodinase 1 Gene.” Environment International, vol. 104, July 2017, pp. 83–90. PubMed, https://doi.org/10.1016/j.envint.2016.12.013.
—. “Association between Exposure to Organochlorine Compounds and Maternal Thyroid Status: Role of the Iodothyronine Deiodinase 1 Gene.” Environment International, vol. 104, July 2017, pp. 83–90. PubMed, https://doi.org/10.1016/j.envint.2016.12.013.
—. “Association between Exposure to Organochlorine Compounds and Maternal Thyroid Status: Role of the Iodothyronine Deiodinase 1 Gene.” Environment International, vol. 104, July 2017, pp. 83–90. PubMed, https://doi.org/10.1016/j.envint.2016.12.013.
—. “Association between Exposure to Organochlorine Compounds and Maternal Thyroid Status: Role of the Iodothyronine Deiodinase 1 Gene.” Environment International, vol. 104, July 2017, pp. 83–90. PubMed, https://doi.org/10.1016/j.envint.2016.12.013.
Malinowski, Jennifer R., et al. “Genetic Variants Associated with Serum Thyroid Stimulating Hormone (TSH) Levels in European Americans and African Americans from the EMERGE Network.” PloS One, vol. 9, no. 12, 2014, p. e111301. PubMed, https://doi.org/10.1371/journal.pone.0111301.
NM_001128177.1(THRB):C.1357C>A (p.Pro453Thr) AND Thyroid Hormone Resistance, Generalized, Autosomal Dominant – ClinVar – NCBI. https://www.ncbi.nlm.nih.gov/clinvar/RCV000013377.22/. Accessed 10 Nov. 2021.
Nordio, Maurizio, and Sabrina Basciani. “Treatment with Myo-Inositol and Selenium Ensures Euthyroidism in Patients with Autoimmune Thyroiditis.” International Journal of Endocrinology, vol. 2017, 2017, p. 2549491. PubMed Central, https://doi.org/10.1155/2017/2549491.
Pałkowska-Goździk, Ewelina, et al. “Type of Sweet Flavour Carrier Affects Thyroid Axis Activity in Male Rats.” European Journal of Nutrition, vol. 57, no. 2, Mar. 2018, pp. 773–82. PubMed, https://doi.org/10.1007/s00394-016-1367-x.
Panicker, Vijay. “Genetics of Thyroid Function and Disease.” The Clinical Biochemist Reviews, vol. 32, no. 4, Nov. 2011, pp. 165–75.
Park, Choonghee, et al. “Associations between Urinary Phthalate Metabolites and Bisphenol A Levels, and Serum Thyroid Hormones among the Korean Adult Population – Korean National Environmental Health Survey (KoNEHS) 2012-2014.” The Science of the Total Environment, vol. 584–585, Apr. 2017, pp. 950–57. PubMed, https://doi.org/10.1016/j.scitotenv.2017.01.144.
Płoski, Rafał, et al. “Thyroid Stimulating Hormone Receptor (TSHR) Intron 1 Variants Are Major Risk Factors for Graves’ Disease in Three European Caucasian Cohorts.” PLOS ONE, vol. 5, no. 11, Nov. 2010, p. e15512. PLoS Journals, https://doi.org/10.1371/journal.pone.0015512.
Qian, Wei, et al. “Association between TSHR Gene Polymorphism and the Risk of Graves’ Disease: A Meta-Analysis.” Journal of Biomedical Research, vol. 30, no. 6, Nov. 2016, pp. 466–75. PubMed Central, https://doi.org/10.7555/JBR.30.20140144.
Roef, Greet, et al. “Heredity and Lifestyle in the Determination of Between-Subject Variation in Thyroid Hormone Levels in Euthyroid Men.” European Journal of Endocrinology, vol. 169, no. 6, Dec. 2013, pp. 835–44. eje.bioscientifica.com, https://doi.org/10.1530/EJE-13-0265.
Sachmechi, Issac, et al. “Autoimmune Thyroiditis with Hypothyroidism Induced by Sugar Substitutes.” Cureus, vol. 10, no. 9, Sept. 2018. www.ncbi.nlm.nih.gov, https://doi.org/10.7759/cureus.3268.
Sinha, Rohit, and Paul M. Yen. “Cellular Action of Thyroid Hormone.” Endotext, edited by Kenneth R. Feingold et al., MDText.com, Inc., 2000. PubMed, http://www.ncbi.nlm.nih.gov/books/NBK285568/.
Thyroid Hormone Receptors. http://www.vivo.colostate.edu/hbooks/pathphys/endocrine/thyroid/receptors.html. Accessed 10 Nov. 2021.
“Top-Selling, Top-Prescribed Drugs for 2016.” Medscape, http://www.medscape.com/viewarticle/886404. Accessed 10 Nov. 2021.
VCV000009790.1 – ClinVar – NCBI. https://www.ncbi.nlm.nih.gov/clinvar/variation/9790/. Accessed 10 Nov. 2021.
Wright, Caroline F., et al. “Assessing the Pathogenicity, Penetrance, and Expressivity of Putative Disease-Causing Variants in a Population Setting.” American Journal of Human Genetics, vol. 104, no. 2, Feb. 2019, pp. 275–86. PubMed Central, https://doi.org/10.1016/j.ajhg.2018.12.015.

