Key takeaways:
- TRPM3 is an ion channel that is sensitive to heat and certain hormones.
- When activated on sensory neurons, TRPM3 sends the signal for pain. In other parts of the body, TRPM3 signaling influences insulin release, immune system function, and blood vessel function.
- Variants in the TRPM3 gene influence pain, cataracts, and susceptibility to ME/CFS.
Members will see their genotype report below and the solutions in the Lifehacks section. Consider joining today.
What is TRPM3?
TRPM3 is a calcium ion channel that is activated by high heat and certain compounds, such as pregnenolone sulfate. Activating the ion channel, such as from heat, causes the signal for increased pain — like sensing that the oven or a car is too hot. TRPM3 channels also play a role in insulin secretion from the pancreas and in vascular constriction.
Where TRPM3 gets interesting is that variants in the gene change your susceptibility to migraines, pain syndromes, and even chronic fatigue syndrome (ME/CFS).
Let’s dig into the details and then look at the genetic variants and solutions.
Background science on TRPM3:
When TRPM3 is activated, it allows calcium ions to flow into the cell, and calcium ions cause different functions, different signals, depending on the type of cell. For example, calcium influx into neurons can cause the neuron to fire, sending a signal, such as for pain.
TRPM3 is involved in:
- heat sensing
- pain transmission, especially with inflammation
- blood vessel relaxation
- neuropeptide release
- insulin release
- immune defense
TRPM3 can also allow zinc ions into cells, such as in the pancreas, where it’s involved in insulin release.[ref]
1) Heat sensing by TRPM3:
Noxious heat is heat that is warm enough to be painful. The TRPM3 is one of several thermal receptors that can detect heat, opening up the ion channel and sending signals through peripheral nerves that heat has been detected. [ref]
In addition, when the body is warmer than normal, the TRPM3 channel is somewhat activated and makes you more sensitive to pain and inflammation. Think about how, when you have a fever, everything kind of hurts — sometimes even your hair hurts. TRPM3 is one of the heat-activated ion channels (along with TRPV1) that is involved there. Genetic variants in TRPM3 make everyone a bit different in their threshold for pain with heat.[ref][ref]
Related article: TRPV1 ion channel genetic variants
Note that TRPM3 is part of a bigger family of channels (e.g TRPM2, TRPM4) that are activated at different temperature ranges.
2) Inflammation, heat, and chronic pain:
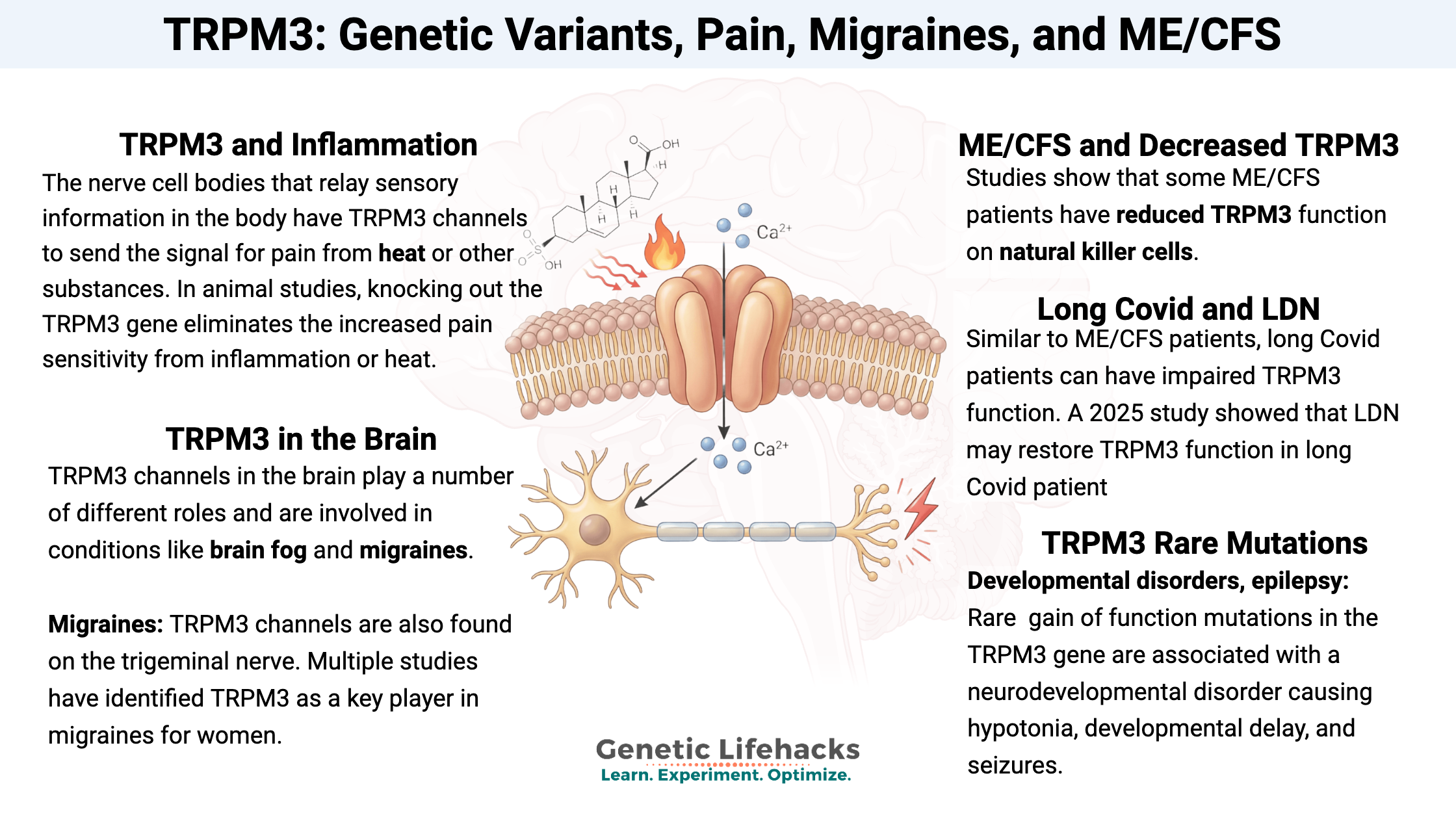
About 60% of the dorsal root ganglion – the nerve cell bodies that relay sensory information – in the body have TRPM3 channels to send the signal for pain from heat or other substances, such as pregnenolone sulfate, that activate the channel. In animal studies, knocking out the TRPM3 gene eliminates the increased pain sensitivity from inflammation or heat. [ref]
Bladder inflammation and cystitis: Animal studies show that pain in bladder inflammation is at least partly due to TRPM3 activation.[ref]
Pancreatitis pain: TRPM3 channels are found in the sensory nerves in the pancreas, and activation of TRPM3 from inflammation is part of what causes the acute pain in pancreatitis. Researchers are looking at blocking TRPM3 to target pain in chronic pancreatitis.[ref]
3) Pregnenolone sulfate activation, progesterone suppression:
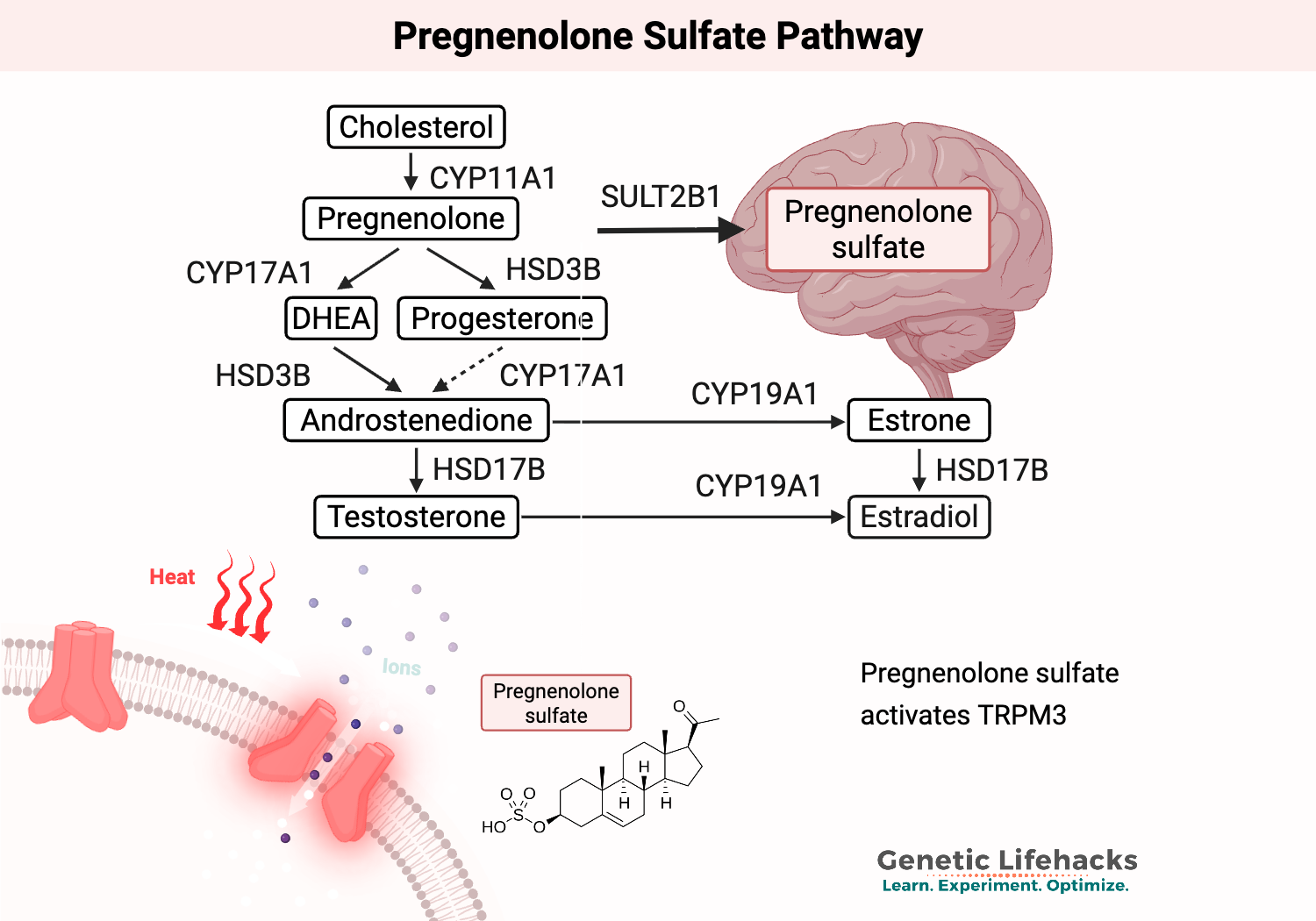
Pregnenolone sulfate is a neurosteroid that modulates the excitatory neurotransmission in the glutamate pathways. It is synthesized from the hormone pregnenolone using the enzyme SULT2B1. Pregnenolone is synthesized from cholesterol in the adrenal glands and is the precursor for other steroid hormones (progesterone, testosterone, estrogen, and cortisol) as well as pregnenolone sulfate, allopregnanolone, and DHEA-S.[ref]
TRPM3 is activated by pregnenolone sulfate and DHEA-S, but progesterone and dihydrotestosterone (DHT) can suppress TRPM3 activation. In vascular smooth muscle cells, the muscles that cause blood vessel relaxation or constriction, progesterone has been shown to inhibit TRPM3 activation by pregnenolone sulfate.[ref][ref]
Here’s an overview of how pregnenolone (and pregnenolone sulfate) fits in with other steroid hormones.
To make this a little more confusing, while progesterone decreases TRPM3 activation, sulfated progesterone metabolites can activate TRPM3. This is mainly seen during pregnancy and can be a problem in gestational diabetes.[ref]
Endometriosis pain: A 2025 study showed that pregnenolone sulfate activating TRPM3 channels on neurons in the endometrium causes endometriosis-associated pain. Pregnenolone sulfate and DHEA-S were shown to be increased in the endometrial tissue, and their activation of TRPM3 causes pain signaling in endometriosis. This activation of TRPM3 is exacerbated by inflammation, which is also involved in endometriosis. While TRPM3 doesn’t explain why endometriosis happens, it does explain some of the pain involved.[ref][ref]
4) Metabolic health and insulin release:
The TRPM3 ion channel is also found in the pancreas, specifically on pancreatic beta cells that release insulin. The activation of TRPM3 by pregnenolone sulfate in the pancreas enhances insulin release in response to glucose.[ref]
5) TRPM3 in the brain: Migraines, neuropeptides, and GABA/glutamate balance
TRPM3 channels in the brain play a number of different roles and are involved in conditions like brain fog and migraines. TRPM3 is also involved in basal glutamate release.
Neurosteroids and sphingolipids: The neurosteroid pregnenolone sulfate acts on TRPM3 channels in the brain, as well as activating other receptors. In addition, the sphingolipid sphingosine and its derivative, sphingosylphosphorylcholine, can also activate TRPM3. These sphingolipids are involved in the myelin sheath, which is the protective, fatty layer that acts like electrical insulation around neurons.[ref]
Restless Leg: A genetic variant in the TRPM3 gene was found to increase the relative risk of restless leg syndrome, likely due to GABA/glutamate balance changes in the brain.[ref] This may be why some people find that changing temperature and cooling off the legs helps with RLS.
Migraines: TRPM3 channels are also found on the trigeminal nerve, which is the main branching nerve in the face and head that is involved in migraines. Multiple studies have identified TRPM3 as a key player in migraines for women.
Let’s look at the mechanism for migraines in more depth:
In migraines, the trigeminal nerve that sends signals from your head and face to your brain is activated, along with changes to the way blood vessels constrict. The trigeminal nerve has lots of TRPM3 channels, and activating TRPM3 allows calcitonin gene-related peptide (CGRP) to be released. CGRP release is key to migraines.
In women, migraines are often triggered by changing hormone levels. I mentioned above that progesterone decreases or inhibits TRPM3. When progesterone and estrogen levels drop during the menstrual cycle, the inhibition on TRPM3 channels is removed. If pregnenolone sulfate is high or another trigger, such as heat, is present, it activates TRPM3, allowing calcium ion influx into the trigeminal nerve endings. The calcium influx then causes CGRP release into the extracellular space, where it binds to receptors on nearby vessels, altering blood flow and triggering nerves to release additional factors, including glutamate, further depolarizing the trigeminal nerves. Researchers think that the sustained activation can cause the nerves to be in a hyperexcitable state, leading to the symptoms of a migraine (including pain).[ref] A 2025 study in animals confirmed that TRPM3 activation triggers CGRP release and vasodilation in the trigeminal nerve, and this was more active in female animals.[ref] There’s a lot of research on this topic right now because a TRPM3 antagonist is in phase II clinical trials for migraines.[ref]
Immune system involvement of TRPM3:
In addition to signaling for heat and pain through sensory nerves, TRPM3 calcium ion channels are found on immune system cells, and particularly natural killer (NK) cells. Activation of TRPM3 in NK cells allows calcium ions to flow in and facilitates the NK cell function.
It may seem, from the above functions – inflammatory pain, bladder cystitis, endometriosis pain – that TRPM3 activation is always a negative. But this ion channel is necessary for both sensory neurons and, importantly, for the way that natural killer cells work.
ME/CFS and decreased TRPM3 function:
Studies show that some ME/CFS (chronic fatigue syndrome) patients have reduced TRPM3 function on natural killer (NK) cells. This is an interesting medical detective story, with researchers finding NK cell dysfunction, tracing it to calcium signaling, and landing on TRPM3.
Let me try to lay out the theory of why this is important:
- Activation of TRPM3 on small nerve fibers causes CGRP and other neuropeptides to be released. This causes vasodilation (relaxation of the muscles surrounding the blood vessels).
- CGRP release is also important in skeletal muscle function, and it stimulates sodium/potassium-ATPase (Na+/K+-ATPase) activity. This is an essential regulator of ions, electrolytes, and muscle function during activity.
- Insufficient Na+/K+-ATPase activity causes excess sodium in cells and an ion imbalance, leading to mitochondrial damage and muscles not responding correctly to exertion.
Thus, a dysregulation or downregulation of TRPM3 could cause many of the symptoms of ME/CFS. Researchers have found that some ME/CFS patients have a higher latency for warmth perception — meaning that they don’t detect hot things as quickly. Cold perception (different ion channel) wasn’t affected.[ref]
TRPM3 activation on natural killer cells allows for calcium ions to flow in at the right level. In NK cells, the sustained calcium ion influx through TRPM3 is needed for the exocytosis of cytotoxic granules. Essentially, NK cells detect virus-infected cells and release toxic granules into the infected cell to kill it. When the natural killer cells aren’t functioning correctly, the body thinks there is an immune system disturbance and acts as if it is sick.
In ME/CFS, TRPM3 dysfunction causes blunted calcium-dependent signaling in the NK cells, which causes cytotoxicity and alters neuroimmune function. Researchers have found that ME/CFS patients’ natural killer cells have reduced TRPM3 calcium influx in response to pregnenolone sulfate and fewer cells that are responsive to TRPM3. There are still a lot of questions as to exactly what is going on here, since calcium ions are used in multiple pathways in the immune cells.[ref]
In addition to the natural killer cell / immune dysregulation pathway, patients with ME/CFS often have neurocognitive dysfunction. This also makes sense with the TRPM3 function being altered, since TRPM3 is also involved in the spontaneous glutamate release in the brain. Glutamate is an excitatory neurotransmitter that is released in large amounts when neurons are firing in the brain due to thinking, moving, etc. But TRPM3 channels, along with TRPV1, are slightly open to allow a little calcium influx, causing low-level glutamate activity all the time. This is something of a keep-alive signal in the brain. Researchers theorized that the TRPM3 dysfunction causes a mismatch of energy demand that then disrupts neurological function, leading to GABA/glutamate imbalance in the brain and skeletal muscles. One more connection with ME/CFS and TRPM3 is the altered vascular response, which could contribute to orthostatic intolerance. [ref][ref][ref]
Importantly, multiple genetic variants in TRPM3 are associated with an increased risk of ME/CFS. This strongly points towards an underlying, important role of TRPM3 in the pathophysiology of ME/CFS.
Long Covid:
Common symptoms reported by many long COVID patients include fatigue, post-exertional malaise, pain, brain fog, and sleep problems, which are symptoms that overlap with ME/CFS. Not everyone with long Covid has the same symptoms, though, so this pathway may not be a solution for everyone with long Covid.
Multiple studies now show that in some long Covid patients, TRPM3 function is also impaired, similar to what is seen in ME/CFS. Moreover, a 2025 study showed that taking low-dose naltrexone (LDN) could restore TRPM3 function in long Covid patient cells to a normal function similar to healthy control cells.[ref][ref]
Low-dose naltrexone, mu-opioid receptors:
I mentioned above that low-dose naltrexone could be used to restore TRPM3 function. Here’s why:
TRPM3 channels are blocked from opening by mu-opioid receptor interactions. This is where low-dose naltrexone (LDN) comes into play. Activated mu-opioid receptors inhibit TRPM3, and low-dose naltrexone is an antagonist of the mu-opioid receptor (temporarily blocks it). Thus, LDN allows for the TRPM3 function to be restored on natural killer cells.[ref]
Studies show that low-dose naltrexone (LDN) can restore TRPM3 function and calcium ion flux in natural killer cells for ME/CFS and long Covid patients.[ref]
A 2025 study involving natural killer cells that were isolated from ME/CFS patients showed that LDN could restore TRPM3 function within 24-hours.[ref]
Genetic diseases associated with TRPM3 mutations:
One way of understanding the function of a gene is to look at the effects of mutations that either increase or decrease the function significantly.
Developmental disorders, epilepsy: Mutations in the TRPM3 gene are associated with a neurodevelopmental disorder (TRPM3-NDD) that can cause hypotonia, developmental delay, and seizures. The mutations that cause seizures are gain-of-function mutations, causing excessive TRPM3 activation. An anticonvulsant medication called primidone acts on TRPM3 to prevent seizures.[ref][ref]
Cataracts: TRPM3 is highly expressed in the retina. Rare mutations that allow excess calcium into the retina can cause early-onset cataracts.[ref][ref]
Circadian rhythm:
For anyone with a chronic pain condition, they often will note that pain is worse at certain times of the day. Many molecules in the body fluctuate over the course of 24-hours due to control by the body’s circadian clock. This holds true for pregnenolone sulfate, a primary activator of TRPM3 and likely involved in pain sensitization.[ref]
As mentioned above, TRPM3 is also highly expressed in the retina and may play a role in modulating photoreceptor function. It isn’t a core ion channel needed for photoreceptor response, though.[ref] (This may be why, anecdotally, some people note slightly improved eyesight when taking pregnenolone.)
Genotype report: TRPM3
TRPM3 gene: encodes a calcium ion channel that impacts the function of natural killer cells. It is also expressed in the neurons, the trigeminal nerve, and the brain.
Check your genetic data for rs962001 (AncestryDNA)
- C/C: altered sphingomyelin levels (sphingomyelin is a phospholipid in the brain’s myelin sheath)[ref]
- C/T: altered sphingomyelin levels (sphingomyelin is a phospholipid in the brain’s myelin sheath)
- T/T: typical
Members: Your genotype for rs962001 is —.
Check your genetic data for rs10780944 (23andMe v5; AncestryDNA):
- G/G: slightly taller on average (males)[ref]
- A/G: slightly taller on average (males)
- A/A: typical
Members: Your genotype for rs10780944 is —.
ME/CFS connection: Multiple studies show that TRPM3 channels show dysfunction in ME/CFS, with most studies showing decreased TRPM3 channel activation and decreased calcium ion influx.[ref]
Check your genetic data from rs6560200 (23andMe v4):
Members: Your genotype for rs6560200 is —.
Check your genetic data for rs1891301 (AncestryDNA):
Members: Your genotype for rs1891301 is —.
Check your genetic data for rs11142508 (23andMe v5):
- T/T: typical
- C/T: increased relative risk of ME/CFS[ref]
- C/C: increased relative risk of ME/CFS
Members: Your genotype for rs11142508 is —.
Check your genetic data for rs1160742 (23andMe v4):
- G/G: typical
- A/G: increased relative risk of ME/CFS[ref]
- A/A: increased relative risk of ME/CFS
Members: Your genotype for rs1160742 is —.
Check your genetic data for rs1328153 (23andMe v4; AncestryDNA)
- A/A: typical
- A/G: increased relative risk of ME/CFS[ref]
- G/G: increased relative risk of ME/CFS
Members: Your genotype for rs1328153 is —.
Check your genetic data for rs7865858 (23andMe v5):
- G/G: typical
- A/G: increased relative risk of ME/CFS[ref]
- A/A: increased relative risk of ME/CFS
Members: Your genotype for rs7865858 is —.
Lifehacks for TRPM3:
If you are dealing with ME/CFS, long Covid, or chronic pain, such as endometrial pain, you may want to consider trying to increase or decrease TRPM3 activation.
Increasing TRPM3 activation:
Research shows that decreased or dysfunctional TRPM3 activation plays a role in ME/CFS and long Covid, at least for some patients.
Pregnenolone: It is the precursor hormone for pregnenolone sulfate, as well as other hormones such as estrogen, DHEA-s, and progesterone. Pregnenolone is readily available as a supplement.
Taking pregnenolone may increase your pregnenolone sulfate levels with the help of the SULT2B1 enzyme. However, your body may prioritize using pregnenolone for other hormone synthesis if needed. While theoretically, pregnenolone may help with ME/CFS or long Covid or other conditions with low TRPM3 function, I haven’t found any research studies addressing this directly with a clinical trial.
Note that pregnenolone may interact with other medications, such as benzodiazepines, sedatives, or exogenous hormones (HRT). Be sure to talk with your doctor about any questions.
Low-dose naltrexone (LDN): The connection with LDN is a bit complicated. Essentially, TRPM3 channels are held in check when they are located near mu-opioid receptors, and the mu-opioid receptor is not blocked. Low-dose naltrexone blocks the mu-opioid receptor when taken at the right dosage. Studies in ME/CFS and in long Covid patients show that LDN can help to restore TRPM3 ion channel function in NK cells.[ref] Talk with your doctor about the pros and cons of LDN (Rx).
Nifedipine: Nifedipine is a calcium channel blocker (Rx) that blocks voltage-gated calcium channels in the heart. But it also acts as a potent, direct agonist of TRPM3 channels. It activates TRPM3, causing calcium influx. In studies, it is often combined with pregnenolone sulfate to synergistically activate TRPM3.[ref]
Phosphatidylinositol-4,5-bisphosphate (PIP2) is a phospholipid that is part of the cell membrane. It’s found on the inner membrane and is part of how cell membranes recruit and use ion channels. PIP2 can also be used in the cell as a precursor for signaling molecules. TRPM3 function is dependent on PIP2, as are many other ion channels.[ref][ref]
Inositol is a precursor for PIP2. If you are deficient in inositol, it is readily available as a supplement.[ref]
Decreasing TRPM3 activation:
There’s a lot of current research on blocking TRPM3 channels for chronic pain. Keep in mind, though, that TRPM3 activation or inhibition may have effects on many systems in the body, including insulin release and blood pressure. Currently, there are few TRPM3 antagonist drugs available, but there is one in phase 2 clinical trials for migraines.[ref]
Here are some of the natural options and medications that target TRPM3.
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
TRPV1 Gene: Receptor for Spicy Foods, Sensing Cold, and CBD Oil
ME/CFS and Genetics: Chronic Fatigue Syndrome Genes, Immune Pathways, and Natural Solutions