Key takeaways:
- There’s a strong genetic component to anxiety that interacts with environmental factors.
- Different physiological changes in the brain can cause anxiety.
- Understanding your genetic susceptibility may help you find the right solutions, tailored to your underlying cause.
Anxiety Disorders and Genetics: What role does heredity play?
If you’re dealing with anxiety, you are not alone! The lifetime risk of anxiety disorder is over 20%, and researchers estimate the heritability at 50%. Anxiety disorder is a term that includes generalized anxiety, panic disorders, social anxiety, PTSD, phobias, and more.
These disorders are a complex group with various ways of presenting; however, there are some very interesting underlying biological similarities in their causes.[ref]
First, here are some of the terms for anxiety disorders:
- Social anxiety disorder is really common and affects up to 10% of the population. People with social anxiety disorder have severe enough anxiety in social situations to cause them to alter their behavior in order to avoid social situations. This can lead to stress or an overall inability to function well in society.[ref]
- Generalized anxiety disorder (GAD) is a condition in which people worry more than normal about everyday things. People with GAD may feel on edge, irritable, tense, and they may have problems sleeping well.
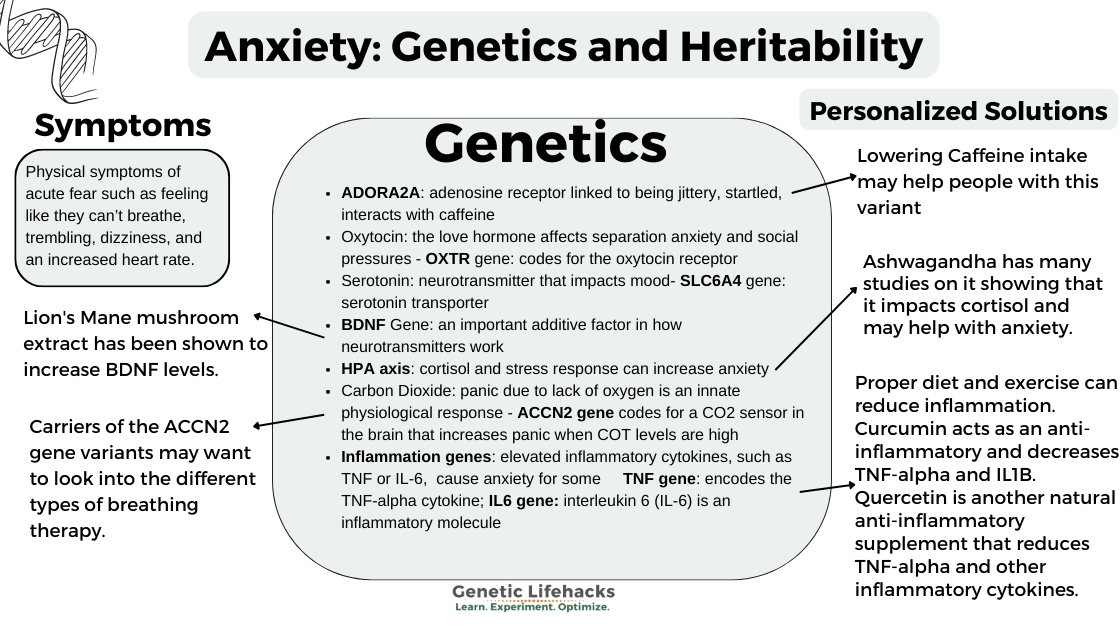
- People with panic disorders have a sudden onset of anxiety. They have physical symptoms of acute fear, such as feeling like they can’t breathe, trembling, dizziness, and an increased heart rate.[source]
Genetic research explains the underlying causes of anxiety disorders:
Genetics plays a role in anxiety disorders, with heritability estimated to be up to 50%. The other half of the picture is environmental and lifestyle factors. Genes + Environment shape our physiological reactions. [ref]
The big takeaway here is that research shows:
- Anxiety disorders have a genetic component for most people.
- Genetics explains the physical alteration in the way the brain works or in the physiological response to stress.
Knowing and understanding your genetic kryptonite may help you find the right solution to your anxiety problems.
How can genetics show the underlying cause?
When it comes to understanding the physiological causes of anxiety, researchers investigate how genes influence the risk of a disease/condition through several methods:
- One way is to first assume that genes in a certain pathway are important and then investigate those genetic variants (SNPs) in detail.
- Another way is to do a Genome-wide Association Study (GWAS), which looks at all the genes in people with a disease to see which variants differ from a control group.
- A final approach is to look at how specific genetic variants interact with lifestyle factors to influence disease risk.
All of these methods have their pros and cons, and often all of the different methods end up being used by different research groups investigating a topic.
Here’s an overview of specific genes linked to anxiety disorders:
| Gene/Pathway | Function/Role | Effect on Anxiety/Disorder | Notes |
|---|---|---|---|
| ADORA2A | Adenosine receptor, neuron signaling | Jitteriness, caffeine sensitivity, startle response | Common variant alters caffeine response |
| Oxytocin (OXTR) | Social bonding, stress response | Social anxiety, separation anxiety | OXTR variants linked to adult separation anxiety |
| Serotonin | Neurotransmitter, mood regulation | General anxiety, depression, PTSD | Multiple serotonin pathway variants |
| BDNF | Neuroplasticity, neurotransmitter support | Additive risk for anxiety and depression | Val66Met (Met allele: reduced activity) |
| HPA Axis | Cortisol/stress response | Panic disorder, altered cortisol rhythms | Altered feedback in PTSD |
| ACCN2 | CO2/pH sensing in amygdala | Panic attacks triggered by CO2 | Acid-sensing ion channel variants |
| Inflammation genes | Immune response, cytokines | Chronic inflammation as root cause | Various cytokine gene variants |
Let’s go into each of these in more detail…
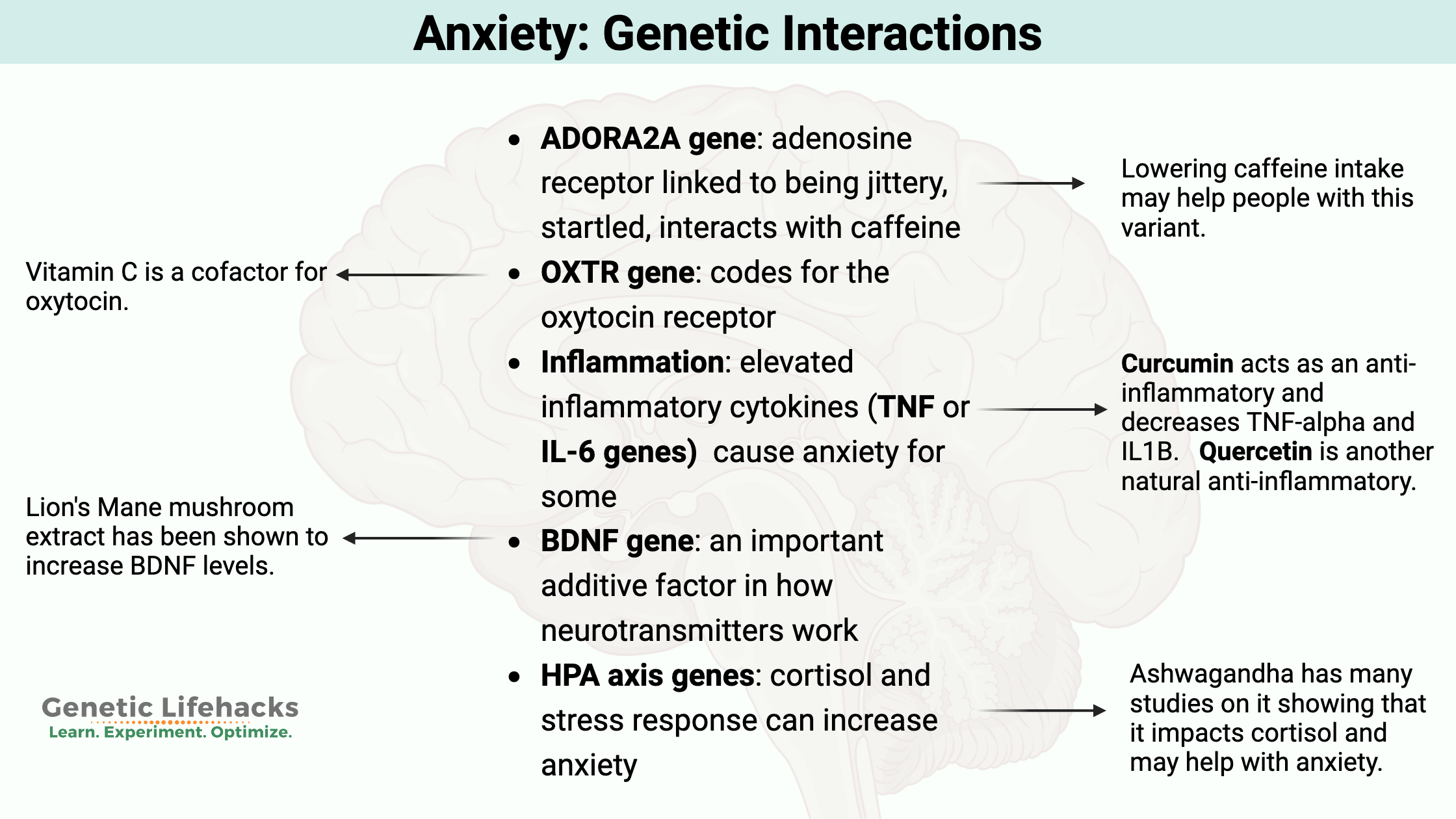
1) ADORA2A and Adenosine: Jittery and startled
One gene linked with anxiety in multiple studies is the ADORA2A gene. ADORA2A codes for the adenosine 2A receptor, which is important in the way that the brain works.
Adenosine is made up of an adenine molecule and a d-ribose sugar molecule. It’s found in every cell in the body.
- Brain balance: In the brain, adenosine is important in the way that the neurons work. It helps to fine-tune the way that neurons communicate, and it also helps to balance the inhibitory and excitatory neurons.[ref]
- DNA: Adenine is a nucleotide (the “A” in your DNA raw data). D-ribose is the sugar that makes up part of the DNA molecule (deoxyribonucleic acid).
- ATP: Adenosine also may sound familiar because it is part of the ATP molecule (adenosine triphosphate). ATP is made in the mitochondria and used for energy in every cell of the body.
Adenosine levels in the brain increase over the course of the day. This higher level of adenosine is what causes you to feel sleepy at night, called the homeostatic sleep drive.
Caffeine works to make you feel awake by blocking the adenosine receptor so that the adenosine can’t attach to it. Genetic variants in the adenosine receptor alter people’s response to caffeine.
Animal studies show that anxiety increases greatly when researchers knock down the ADORA2A (adenosine receptor) gene expression. They had increased heart rate, increased platelet aggregation, and an altered pain response — adenosine is important in a lot of different functions in the body.[ref]
But what about in humans? There is a common genetic variant in the ADORA2A gene that alters its function. An fMRI study found that ADORA2A variant carriers had increased connectivity between two regions of the brain: the prefrontal cortex and the insula (part of the cerebral cortex).[ref]
One more way ADORA2A variants impact the risk of anxiety disorder is through the impact on the startle response and the maladaptive emotional reactivity.[ref]
All in all, this paints a picture of the adenosine receptor being important in how neurons communicate. Alterations in this pathway may cause people to be more on alert, startled easily, and jittery (especially with high caffeine).
2) Oxytocin and separation anxiety
Oxytocin is a hormone and a neuropeptide that researchers link to both emotional functioning and social empathy. In women, large quantities of oxytocin are released during childbirth and breastfeeding. It’s a big part of mother-child bonding. In fact, oxytocin is involved in a mother’s neuroendocrine response to their baby’s crying. Researchers study this using fMRI brain imaging on mothers listening to crying babies.[ref] It is a built-in, physiological response based on oxytocin.
Going beyond parent-infant bonding, oxytocin, in general, plays a role in social attachment and trust. For example, if you give normal adults oxytocin intranasally (up the nose), there is a substantial increase in trust among people, which increases the benefits of social interaction.[ref][ref]
Stressful situations cause the body to increase oxytocin levels, which acts quickly to decrease anxiety. But oxytocin creates an anti-anxiety effect only under stressful conditions.[ref] In other words, boosting oxytocin when you’re not stressed doesn’t change anything. It is only a ‘chill pill’ in situations that have your body on alert.
Adult separation anxiety is characterized by feeling anxious when separated from a loved one, a pet, or even being away from home. It leaves people feeling anxious or worried in anticipation of being away from someone/something, and it also can involve fear or nightmares about being separated from someone.
The disorder is linked to oxytocin levels, and variants in the oxytocin receptor increase susceptibility to adult separation anxiety. [ref][article]
Related article: Oxytocin genes
3) Serotonin can play a role in anxiety and depression
Serotonin is a neurotransmitter involved in mood. This pathway is a common target of antidepressant and antianxiety medications known as SSRIs. Serotonin is also important in gut motility and in sleep (as the precursor for melatonin).
Researchers still have a lot of questions about how serotonin works in anxiety. The key may be that serotonin acts differently in situations of fear or panic, compared to situations that cause general anxiety.[ref][ref]
Genetic connections: Several genetic variants in serotonin-related pathways have been linked to PTSD, a type of anxiety disorder.
Related article: Serotonin genes
4) BDNF: Additive factor
Brain-derived neurotrophic factor (BDNF) has multiple functions in the brain, including neuroplasticity and potentiating neurotransmitter function. Basically, it helps grow new neurons and helps neurotransmitters work better.
Genetic connections: BDNF genetic variants are linked with an increased risk of depression and anxiety, but the common variants in this gene also interact with other genes and with stress in changing the risk for depression. The Val66Met variant (listed below) has been investigated in over a thousand studies. The Met allele carriers have reduced BDNF activity.[ref][ref]
A recent animal study tested the theory that BDNF genetic variants interact with stress as a ‘two-hit’ combo for impacting mood and brain function. The researchers gave normal mice and BDNF-reduced mice (Val/Met heterozygous mice) a stress hormone (cortisol) in their water. Then they tested the effects of different environments. Mice that were housed in an enriched environment (toys, tunnels, more open housing, exercise, mazes, etc) did not have brain changes in the BDNF + stress hormone mice. But the female mice in a non-enriched environment exposed to the ‘two-hits’ had changes in spatial memory, maze performance, and the hippocampus.[ref]
Related article: Increasing BDNF
5) HPA Axis: Cortisol and stress response
The HPA axis – hypothalamic, pituitary, adrenal cortex – controls the body’s cortisol release in times of stress.
The adrenals release cortisol after exposure to physical or mental stressors. This system works great when you are being chased by a tiger once in a while, but chronically elevated cortisol is linked to a number of health problems, including depression.
The body’s baseline level of cortisol rises and falls in a circadian rhythm over the course of 24 hours. Having this rhythm in sync with the rest of the body is important for both physical and mental health.
Altered cortisol levels with low cortisol and a stronger negative feedback loop are found in people with PTSD. While the research is still ongoing, there seems to be a subset of people for whom an altered cortisol response is at the root of either their anxiety or depression.[ref]
Research shows that people with panic disorder have higher overnight cortisol levels, and they have an exaggerated response to novel stressors.[ref]
Related article: HPA Axis genes
6) Carbon dioxide: Physical trigger for panic attacks
In severe anxiety or panic disorders, people often feel like they are short of breath or suffocating. Interestingly, people with panic disorder also have a heightened sensitivity to carbon dioxide. Most people, when inhaling higher percentages of carbon dioxide, don’t have a response. However, in people with panic disorder, inhaling a higher-than-normal amount of CO2 can actually trigger a panic attack.[ref]
Increasing the CO2 levels in the blood increases the acidity (lower pH). Researchers theorize that a chemical sensor in the amygdala is involved in the detection of pH levels. In mouse studies, inhaled CO2 drops the pH in the amygdala and causes fear behaviors. This is mediated by the acid-sensing ion channel-1a subunit (coded for by the ACCN2 gene in humans).[ref][ref]
Genetic connection: The ACCN2 (or ASIC1) gene codes for a protein important in sensing CO2 levels in the brain. Variants in ACCN2 cause a greater sensitivity to higher CO2 (low O2), which triggers panic more easily. The variants are found more commonly in people who are diagnosed with panic disorder. In people without panic disorder, the variant still alters the response in the amygdala to fearful situations. [ref]
7) Inflammation as a physiological cause of anxiety:
For some, a root cause of depression and/or anxiety can be chronic inflammation. Some people are just wired to produce a higher inflammatory response, and this can cause the physical alterations linked to anxiety.
This is such a huge topic, I’ve created a whole article on: Is Inflammation Causing Your Depression or Anxiety
Anxiety Genotype Report
This section gets specific on genetic variants of the pathways discussed above. The variants included below are found in 23andMe or AncestryDNA data and also have multiple studies to back up the assertions.
Access this content:
An active subscription is required to access this content.
Lifehacks for Anxiety (personalized for your genes):
The following are solutions that may work for specific genetic variants. Everyone is different, of course. Use your best judgment and talk with your doctor if you are under a doctor’s care for anxiety.
Here’s an overview of where we are going:
| Gene/Variant | Personalized Solution | Notes/References |
|---|---|---|
| ADORA2A | Reduce caffeine intake | Try half-caf, <150mg/day |
| OXTR | Boost oxytocin (pet a dog, watch puppy videos, vitamin C) | Useful for separation/social anxiety |
| BDNF | Enrich environment, exercise | Especially important for Met allele carriers |
| HPA Axis | Maintain circadian rhythm, manage stress | Consider sleep hygiene, stress reduction |
| GABA | Sufficient vitamin B6 as P5P | Increasing GABA helps calm the brain |
| Inflammation | Address chronic inflammation | See related article on inflammation |
ADORA2A:
If you carry the ADORA2A receptor variant and drink beverages containing caffeine, try cutting down on caffeine to see if it helps with your anxiety.
- Low doses, half-calf: Caffeine at low doses seems to help some people with anxiety, so cutting out coffee altogether may not be the right move. If you’re a coffee drinker, try switching to half decaf/half regular, and decrease caffeine levels to less than 150mg per day.[ref]
OXTR variants:
Understanding the way that social interaction and empathy are important to you personally, may help you to modify your interactions. If you carry the G/G allele and you have anxiety when separated from loved ones, you may want to see about boosting oxytocin levels at times when you feel anxious. Here are a couple of ways to raise oxytocin levels:
- Gazing into a dog’s eyes and petting a dog both increase oxytocin levels.[ref][ref] Don’t have a dog nearby? Try puppy videos.
- Vitamin C is a cofactor for creating oxytocin. Perhaps some extra vitamin C before going to a group function might help to increase oxytocin.[ref][ref]
Increasing BDNF:
If you carry the BDNF variants, you may want to experiment with boosting your BDNF levels on a long-term basis.
- Exercise: Even a single bout of exercise increases BDNF levels in the hippocampus.[ref][ref] Find some kind of activity that you can enjoy daily: walking, jogging, weight lifting, tennis, pilates, biking…
- Sunlight: Exposure to sunlight or bright light during the day increases BDNF levels.[ref] Go outside!
Animal studies show that increased branched-chain amino acid (BCAA) intake promoted BDNF growth and made the animals less anxious and more resilient to stress. This was shown to affect the same pathway that exercise affects, but adding BCAA to exercise didn’t have a synergistic effect.[ref] Thus, it may be worthwhile to experiment with increasing BCAAs on days that you don’t exercise to see if there is an effect on anxiety. Supplemental branch chain amino acids are often sold in the weight-lifting section of a health foods store.
Access this content:
An active subscription is required to access this content.
Graphical Abstract:
Putting it all together:
It is entirely likely that there is more than one genetic pathway involved in anxiety for most people. Figure out which tools you need in your toolbox – e.g. cortisol regulation plus breath work, or focusing on sleep, along with avoiding caffeine.
Not finding your answer here? Check out these articles as well:
GABA could be playing a role in anxiety
Lithium Orotate + Vitamin B12 can be helpful for some people
L-theanine: research studies and how it works
Histamine intolerance and/or Mast Cell Activation Syndrome can cause anxiety symptoms
Related Articles and Topics:
Is Inflammation Causing Your Depression and Anxiety? The Science Behind the Link
References
Abelson, James L., et al. “HPA Axis Activity in Patients with Panic Disorder: Review and Synthesis of Four Studies.” Depression and Anxiety, vol. 24, no. 1, 2007, pp. 66–76. Wiley Online Library, https://doi.org/10.1002/da.20220.
Anderson, I. M., and C. Mortimore. “5-HT and Human Anxiety. Evidence from Studies Using Acute Tryptophan Depletion.” Advances in Experimental Medicine and Biology, vol. 467, 1999, pp. 43–55. PubMed, https://doi.org/10.1007/978-1-4615-4709-9_6.
Chen, Frances S., et al. “Common Oxytocin Receptor Gene (OXTR) Polymorphism and Social Support Interact to Reduce Stress in Humans.” Proceedings of the National Academy of Sciences of the United States of America, vol. 108, no. 50, Dec. 2011, pp. 19937–42. PubMed, https://doi.org/10.1073/pnas.1113079108.
Childs, Emma, et al. “Association between ADORA2A and DRD2 Polymorphisms and Caffeine-Induced Anxiety.” Neuropsychopharmacology: Official Publication of the American College of Neuropsychopharmacology, vol. 33, no. 12, Nov. 2008, pp. 2791–800. PubMed, https://doi.org/10.1038/npp.2008.17.
Corchs, Felipe, et al. “Evidence for Serotonin Function as a Neurochemical Difference between Fear and Anxiety Disorders in Humans?” Journal of Psychopharmacology (Oxford, England), vol. 29, no. 10, Oct. 2015, pp. 1061–69. PubMed, https://doi.org/10.1177/0269881115590603.
Costa, Barbara, Stefano Pini, David S. Baldwin, et al. “Oxytocin Receptor and G-Protein Polymorphisms in Patients with Depression and Separation Anxiety.” Journal of Affective Disorders, vol. 218, Aug. 2017, pp. 365–73. PubMed, https://doi.org/10.1016/j.jad.2017.03.056.
—. “Oxytocin Receptor and G-Protein Polymorphisms in Patients with Depression and Separation Anxiety.” Journal of Affective Disorders, vol. 218, Aug. 2017, pp. 365–73. PubMed, https://doi.org/10.1016/j.jad.2017.03.056.
Costa, Barbara, Stefano Pini, Pamela Gabelloni, et al. “Oxytocin Receptor Polymorphisms and Adult Attachment Style in Patients with Depression.” Psychoneuroendocrinology, vol. 34, no. 10, Nov. 2009, pp. 1506–14. PubMed, https://doi.org/10.1016/j.psyneuen.2009.05.006.
Domschke, Katharina, et al. “ADORA2A Gene Variation, Caffeine, and Emotional Processing: A Multi-Level Interaction on Startle Reflex.” Neuropsychopharmacology, vol. 37, no. 3, Feb. 2012, pp. 759–69. PubMed Central, https://doi.org/10.1038/npp.2011.253.
Field, David T., et al. “High‐dose Vitamin B6 Supplementation Reduces Anxiety and Strengthens Visual Surround Suppression.” Human Psychopharmacology: Clinical and Experimental, vol. 37, no. 6, Nov. 2022, p. e2852. DOI.org (Crossref), https://doi.org/10.1002/hup.2852.
Geiger, Maximilian J., et al. “ADORA2A Genotype Modulates Interoceptive and Exteroceptive Processing in a Fronto-Insular Network.” European Neuropsychopharmacology: The Journal of the European College of Neuropsychopharmacology, vol. 26, no. 8, Aug. 2016, pp. 1274–85. PubMed, https://doi.org/10.1016/j.euroneuro.2016.05.007.
Grech, Adrienne M., et al. “Sex-Dependent Effects of Environmental Enrichment on Spatial Memory and Brain-Derived Neurotrophic Factor (BDNF) Signaling in a Developmental ‘Two-Hit’ Mouse Model Combining BDNF Haploinsufficiency and Chronic Glucocorticoid Stimulation.” Frontiers in Behavioral Neuroscience, vol. 12, 2018, p. 227. PubMed, https://doi.org/10.3389/fnbeh.2018.00227.
“Help for Anxiety and Panic Disorders.” American Family Physician, vol. 91, no. 9, May 2015, p. 617.
Henckens, Marloes J. A. G., et al. “Interindividual Differences in Stress Sensitivity: Basal and Stress-Induced Cortisol Levels Differentially Predict Neural Vigilance Processing under Stress.” Social Cognitive and Affective Neuroscience, vol. 11, no. 4, Apr. 2016, pp. 663–73. PubMed Central, https://doi.org/10.1093/scan/nsv149.
Hohoff, Christa, et al. “Adenosine A(2A) Receptor Gene: Evidence for Association of Risk Variants with Panic Disorder and Anxious Personality.” Journal of Psychiatric Research, vol. 44, no. 14, Oct. 2010, pp. 930–37. PubMed, https://doi.org/10.1016/j.jpsychires.2010.02.006.
Kosfeld, Michael, et al. “Oxytocin Increases Trust in Humans.” Nature, vol. 435, no. 7042, June 2005, pp. 673–76. PubMed, https://doi.org/10.1038/nature03701.
Ledent, C., et al. “Aggressiveness, Hypoalgesia and High Blood Pressure in Mice Lacking the Adenosine A2a Receptor.” Nature, vol. 388, no. 6643, Aug. 1997, pp. 674–78. PubMed, https://doi.org/10.1038/41771.
Maud, Catherine, et al. “The Role of Oxytocin Receptor Gene (OXTR) DNA Methylation (DNAm) in Human Social and Emotional Functioning: A Systematic Narrative Review.” BMC Psychiatry, vol. 18, May 2018, p. 154. PubMed Central, https://doi.org/10.1186/s12888-018-1740-9.
McQuaid, Robyn J., et al. “Distress of Ostracism: Oxytocin Receptor Gene Polymorphism Confers Sensitivity to Social Exclusion.” Social Cognitive and Affective Neuroscience, vol. 10, no. 8, Aug. 2015, pp. 1153–59. PubMed, https://doi.org/10.1093/scan/nsu166.
Neumann, Inga D., and David A. Slattery. “Oxytocin in General Anxiety and Social Fear: A Translational Approach.” Biological Psychiatry, vol. 79, no. 3, Feb. 2016, pp. 213–21. www.biologicalpsychiatryjournal.com, https://doi.org/10.1016/j.biopsych.2015.06.004.
Otowa, Takeshi, et al. “Meta-Analysis of Genome-Wide Association Studies of Anxiety Disorders.” Molecular Psychiatry, vol. 21, no. 10, Oct. 2016, pp. 1391–99. PubMed Central, https://doi.org/10.1038/mp.2015.197.
—. “Meta-Analysis of Genome-Wide Association Studies of Anxiety Disorders.” Molecular Psychiatry, vol. 21, no. 10, Oct. 2016, pp. 1391–99. PubMed Central, https://doi.org/10.1038/mp.2015.197.
Perna, G., et al. “Carbon Dioxide/Oxygen Challenge Test in Panic Disorder.” Psychiatry Research, vol. 52, no. 2, May 1994, pp. 159–71. PubMed, https://doi.org/10.1016/0165-1781(94)90085-x.
Silove, Derrick, et al. “Pediatric-Onset and Adult-Onset Separation Anxiety Disorder Across Countries in the World Mental Health Survey.” The American Journal of Psychiatry, vol. 172, no. 7, July 2015, pp. 647–56. PubMed Central, https://doi.org/10.1176/appi.ajp.2015.14091185.
Skvortsova, Aleksandrina, et al. “Effects of Oxytocin Administration and Conditioned Oxytocin on Brain Activity: An FMRI Study.” PLoS ONE, vol. 15, no. 3, Mar. 2020, p. e0229692. PubMed Central, https://doi.org/10.1371/journal.pone.0229692.
Smoller, Jordan W., et al. “The Human Ortholog of Acid-Sensing Ion Channel Gene ASIC1a Is Associated With Panic Disorder And Amygdala Structure And Function.” Biological Psychiatry, vol. 76, no. 11, Dec. 2014, pp. 902–10. PubMed Central, https://doi.org/10.1016/j.biopsych.2013.12.018.
Stein, Murray B., et al. “Genetic Risk Variants for Social Anxiety.” American Journal of Medical Genetics. Part B, Neuropsychiatric Genetics : The Official Publication of the International Society of Psychiatric Genetics, vol. 174, no. 2, Mar. 2017, pp. 120–31. PubMed Central, https://doi.org/10.1002/ajmg.b.32520.
Temido-Ferreira, Mariana, et al. “Novel Players in the Aging Synapse: Impact on Cognition.” Journal of Caffeine and Adenosine Research, vol. 9, no. 3, Sept. 2019, pp. 104–27. PubMed Central, https://doi.org/10.1089/caff.2019.0013.
Tsai, Shih-Jen. “Critical Issues in BDNF Val66Met Genetic Studies of Neuropsychiatric Disorders.” Frontiers in Molecular Neuroscience, vol. 11, May 2018, p. 156. PubMed Central, https://doi.org/10.3389/fnmol.2018.00156.
Ziemann, Adam E., et al. “The Amygdala Is a Chemosensor That Detects Carbon Dioxide and Acidosis to Elicit Fear Behavior.” Cell, vol. 139, no. 5, Nov. 2009, pp. 1012–21. PubMed, https://doi.org/10.1016/j.cell.2009.10.029.