Key takeaways:
- The MTHFR enzyme converts folate (vitamin B9) to its active form, 5-MTHF.
- Folate is used to create methyl groups, which cells use to synthesize neurotransmitters, detoxify toxicants, methylate DNA, and maintain a healthy heart.
- The common MTHFR C677T and A1298C variants lower enzyme function (up to ~70–80% for C677T homozygous, ~20% for A1298C homozygous), especially when folate is limited.
- You don’t need expensive testing for MTHFR; you can check your C677T and A1298C SNPs in 23andMe, AncestryDNA, or similar raw data files.
- Optimizing your diet and nutrient intake to align with your genetic variants may help to improve overall wellness and healthy aging.
What is the MTHFR gene?
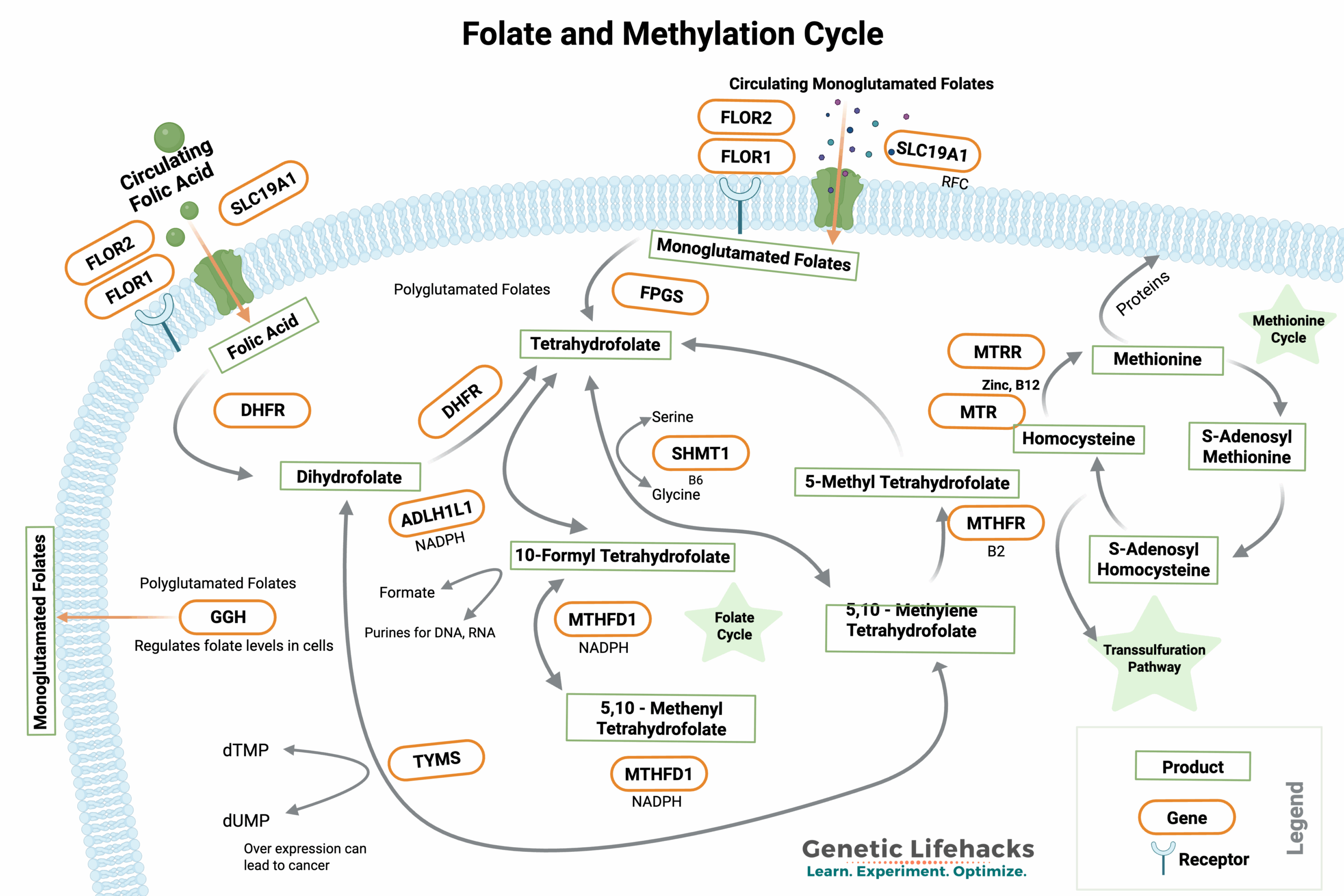
MTHFR is a central gene in the methylation cycle and is a limiting factor for producing methyl groups from folate (vitamin B9).
Specifically, the MTHFR gene codes for an enzyme called methylenetetrahydrofolate reductase that turns folate into the active form, 5-methyltetrahydrofolate. This enzyme, along with the active form of vitamin B-12 (methylcobalamin), drives an essential portion of the methylation cycle.
Common genetic variants, called MTHFR C677T and A1298C, affect how the enzyme works and impact the availability of methyl groups.[ref] Genetic variants are variations in the DNA sequence that can influence the function of a gene or the protein it encodes.
Which DNA tests cover MTHFR?
You don’t need expensive or specialized testing to find out your MTHFR status. The MTHFR genetic variants are available in 23andMe and AncestryDNA raw data files which are free to download if you’ve done a test with them.
Here’s a quick list of inexpensive testing options:
- 23andMe
- AncestryDNA
- MyHeritage
- Sequencing .com
- SelfDecode
- MyGene Food
- TellMeGen
In addition to understanding your MTHFR variants from your DNA raw data, you may also want to consider testing your homocysteine levels to see what your current status is.
MTHFR: Key role in the methylation cycle
The MTHFR gene encodes an enzyme that is a key part of the methylation cycle.
Methylation is the addition and removal of a methyl group (-CH3) to amino acids, DNA, and other enzymes or proteins. Within the methylation cycle, folate can be used to donate a methyl group that is used by SAMe for methylation reactions. This is a cycle that also involves homocysteine (which we will come back to in a minute).
Related article: Methylation cycle report (MTHFR, COMT, B12, and more)
Methyl groups are used to:
- Form new molecules, including neurotransmitters
- Control DNA gene expression (turn on and off genes)
- Detoxify certain substances
Let’s look at each of these:
1. Forming new molecules:
Most of the molecules in our body are chains of hydrocarbons — carbons plus hydrogens. So adding a methyl group, a carbon plus three hydrogens, stacks on one more link in a hydrocarbon chain. The methyl group changes the original molecule into something different. A methyl group makes the molecule non-polar, which means that it isn’t able to mix with water and instead can pass through lipid membranes more easily.
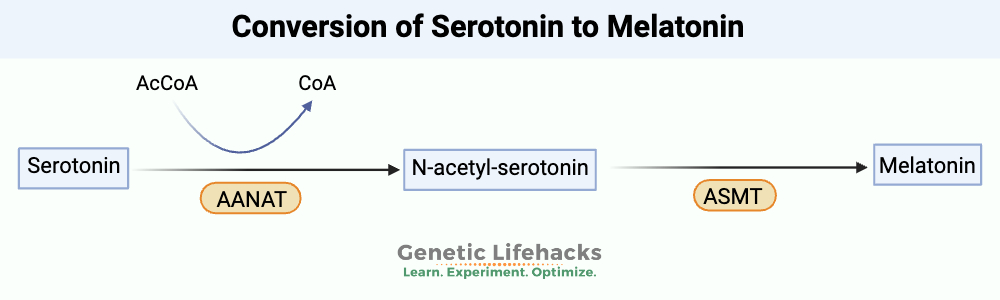
Example: The synthesis of melatonin involves methylation
A methyl group is added to serotonin in the two-step process that forms melatonin.
2. DNA methylation:
DNA methylation is the addition of a methyl group to specific locations on a chromosome. By binding to these spots, methylation can turn genes on and off, and maintain and repair your DNA. This is an ongoing, essential, and continual use of methyl groups.
3. Detoxification and breaking down substances:
Methylation is essential in the nervous system, in the production and breakdown of neurotransmitters, and in detoxifying some specific environmental toxicants such as arsenic.
The folate and methylation cycle is a series of biochemical reactions that involve the transfer of methyl groups. For those who like a visual pathway, here’s what the complete folate and methylation cycle looks like. Notice that MTHFR is in a key spot, tying together 5-methyltetrahydrofolate and homocysteine.
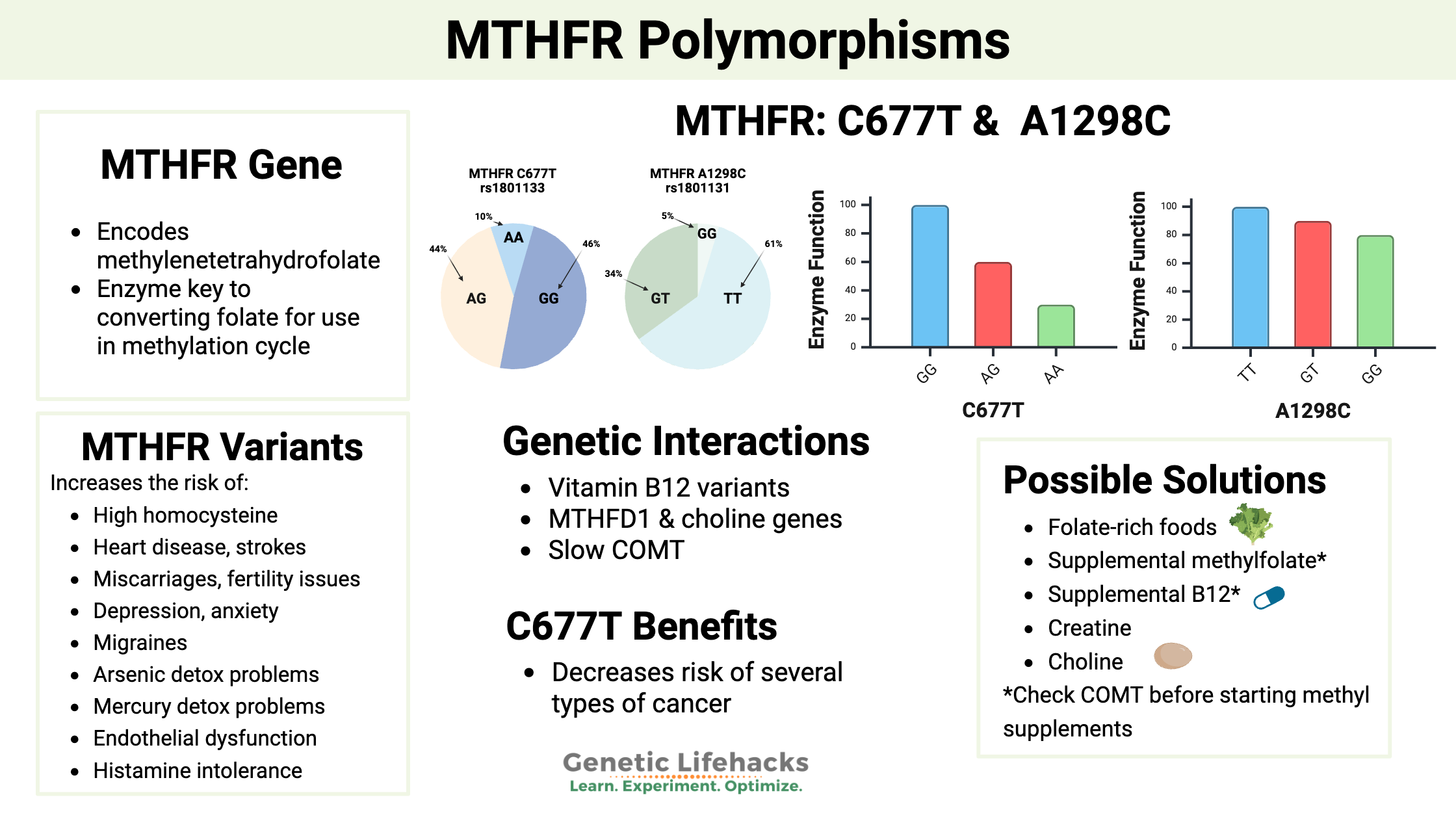
The MTHFR C677T and A1298C variants:
Two common variants in the MTHFR gene, C677T and A1298C, cause the enzyme not to function normally. Let’s look at what this means and the implications for your health.
Understanding the terminology:
The MTHFR C677T variant is a single nucleotide polymorphism (SNP). A SNP is a variation in a single nucleotide base pair (the As, Cs, Gs, and Ts) that differs from the typical nucleotide at that spot in the gene for part of the population. Similarly, A1298C is another SNP in the MTHFR gene.
Both MTHFR C677T and MTHFR A1298C are common variants (SNPs). Both polymorphisms are found in approximately half the population. The substitution of a different nucleotide – the A instead of a G – makes the MTHFR enzyme function a little differently.
| Variant/SNP | Genotype | Enzyme function effect | Notes |
|---|---|---|---|
| C677T (rs1801133) | G/G | Typical | No major reduction in activity. |
| C677T (rs1801133) | A/G | ~35–40% decreased | Thermolabile enzyme; more sensitive to low folate. |
| C677T (rs1801133) | A/A | ~70–80% decreased | Strongest impact on folate-dependent methylation. |
| A1298C (rs1801131) | T/T | Typical | No major reduction. |
| A1298C (rs1801131) | G/T | Slightly decreased | Mild effect alone. |
| A1298C (rs1801131) | G/G | ~20% decreased | Moderate reduction. |
| Compound C677T + A1298C | A/G + G/T | ~50% decreased | One copy of each |
Let’s look at the details of what this means:
What is the impact of MTHFR C677T:
The MTHFR C677T variant changes the enzyme structure in a way that makes it break down faster at normal body temperature (more thermolabile). This faster enzyme breakdown then reduces the amount of enzyme available in each cell. This can be a problem when folate is limited.[ref][ref][ref]
- One copy of the C677T variant reduces enzyme function by about 35-40%.
- Two copies (homozygous) cause a ~70% reduction in enzyme function.
Why is it called C677T?
The variant involved in C677T is a change from Cytosine (C) to Thymine (T) at position 677. This variant was originally defined on the minus strand of the DNA, but most genetic raw data files report it on the plus strand. That is why in your genetic raw data, you will see G for the typical and A for the variant, but many research studies will still include C for the typical and T for the variant. Just remember that C=G and T=A.
What is the functional impact of A1298A?
The A1298C variant causes less of a change to the way the enzyme works, with two copies of the variant decreasing enzyme function by around 20%. [ref]
Compound heterozygous: MTHFR C677T + A1298C:
Having one copy of each variant, called compound heterozygous, reduces enzyme function by around 50%. You may be wondering about what happens if someone has two copies of each variant. That is a combination that is rarely, if ever, seen (likely incompatible with life).
Research studies on MTHFR C677T or A1298C variants:
Having an MTHFR variant increases the relative risk (not certainty!) of many chronic diseases, but this does not mean it will cause you to have that disease.
According to multiple meta-analyses, MTHFR C677T and A1298C increase the relative risk for high homocysteine, cardiovascular disease, stroke, neural tube defects, certain pregnancy issues, depression, migraines, and some neuropsychiatric conditions, especially with low folate/B vitamins.[ref] These same variants can lower the risk for certain conditions as well, illustrating tradeoffs of both positive and negative effects. Importantly, dietary changes or supplemental vitamins can eliminate many of the problems associated with the MTHFR variants.
The MTHFR gene is extremely well researched, with over 6,000 studies investigating the C677T variant. There is also a lot of misinformation and hype about MTHFR, so this article sticks just to the high-quality studies on the topic.
Studies show that the C667T and A1298C variants increase the relative risk:
- high homocysteine, stroke, and heart disease[ref][ref][ref][ref][ref]
- neural tube defects and cleft lip[ref]
- preeclampsia and hypertension in pregnancy[ref][ref]
- miscarriage[ref][ref]
- depression or anxiety[ref][ref][ref][ref][ref]
- Alzheimer’s[ref][ref]
- autism spectrum disorder (C677T only according to 2026 meta-analysis)[ref]
- rare problems with nitrous oxide[ref], but most have no problems[ref]
Let’s dig into the details of what research studies and clinical trials show.
Studies on MTHFR variants show:
1. Depression and the MTHFR Gene Variants: A1298C and C677T
- A meta-analysis of 26 studies found that the MTHFR C677T variant was associated with an increased risk of depression.[ref] Age and gender may also play a role here. Postmenopausal women who carried the C677T variant had a 2 to 3-fold increased risk of depression.[ref]
- Women with two copies of the A1298C variant were at twice the risk of major depressive disorder (MDD). The risk of MDD was even higher in people who also had COMT slow (MET) alleles. (read about COMT)[ref]
Not all studies agree, and some studies show that the MTHFR C677T variant has little to no impact on depression risk. The difference in study results could be due to diet. People who eat a diet that includes more folate (green vegetables, legumes, liver) may not be at an increased risk of mood disorders, while people who eat few folate-rich foods may be more susceptible to depression.[ref][ref]
Related article with more details: MTHFR: depression and anxiety
2. High Homocysteine with MTHFR variants:
The methylation cycle also controls the level of homocysteine, an important marker of heart disease risk. It is also involved in cholesterol levels.[ref] Genetic variants in the methylation pathway, including MTHFR, are strongly linked to high homocysteine levels and heart disease in many studies. For example, a study in acute coronary syndrome patients showed that MTHFR C677T increased severity as well as homocysteine levels. High homocysteine is also linked to increased blood clots.[ref][ref][ref][ref]
Related article: Homocysteine- Genetics and Solutions
3. Increased risk of heart disease
Studies show a link between MTHFR C677T and an increased risk of cardiovascular disease. For example, a meta-analysis found that two copies of the MTHFR C677T variant (A/A, homozygous) increase the relative risk of heart disease by 38%.[ref][ref] However, not all studies agree, and a large prospective study (n=6,000) found that older people with two copies of the C677T allele were at a 30% decreased risk of death from cardiovascular disease when other parameters were included.[ref]
The increased risk is not just due to high homocysteine. People with the C677T variant (AG or AA) have reduced endothelial function, even when homocysteine levels are normalized by increasing folate intake. The endothelium is the lining of blood vessels, and endothelial function controls how the blood vessels relax or contract to control blood pressure.[ref][ref]
Related article: MTHFR, riboflavin, and reducing high blood pressure
4. Pregnancy, infertility, and MTHFR:
One of the first researched links to MTHFR variants was neural tube defects in infants. Babies need folate for the spinal column to develop fully, and the MTHFR variants increase the risk of problems.[ref] This is why prenatal vitamins contain folate and why folic acid is added to white rice and white flour.
Related article: MTHFR, pregnancy, and infertility
5. Breakdown of estrogen, interaction with histamine:
Additionally, the methylation pathway involves the regulation of hormones, such as estrogen, and plays a role in histamine levels through breaking down high histamine.
Related article: Histamine metabolism and estrogen receptors
6. Migraines
Numerous studies show that MTHFR variants are linked to a significantly increased risk of migraines. Some studies indicate that the risk is also due to higher homocysteine levels[ref], while other studies show that it may be due to the methylation of certain genes.[ref]
Meta-analyses showed that the MTHFR C677T variant increased the risk of migraines with aura for all population ancestry groups. In non-Caucasians, the C677T variant increased migraine risk by 3-fold.[ref][ref] In North Indians, the A1298C variant was associated with the risk of migraines.[ref]
Related article: Getting to the root genetic cause of migraines
7. Detoxification and MTHFR
A methyl group is needed in the detoxification reaction for arsenic. The enzyme (arsenite methyltransferase) that metabolizes arsenic depends on the availability of methyl groups. The C677T variant is linked to decreased arsenic detoxification and increased skin lesions with exposure.[ref][ref]
Related article: Arsenic detoxification genes
Additionally, methylation is important in detoxifying mercury. In fact, MTHFR variants are tentatively linked to being more likely to have problems detoxifying mercury (small study).[ref][ref]
Related article: Mercury detoxification genes
Tradeoffs! Positive benefits of MTHFR SNPs
You may wonder why MTHFR variants – with such negative effects – are so common. It seems like a variant that should have been weeded out with natural selection. For common genetic variants, there is almost always a positive effect that balances the negatives. Plus, there are interactions between our modern world
The big positive for MTHFR C677T is that it protects against several common types of cancer. Folate is needed for cell growth, especially fast-dividing cancer cells, and the decrease in folate with MTHFR variants can protect against growth in fast-growing tumors.
Studies finding protective effects against cancer:
- A meta-analysis found that two copies of the C677T variant (AA genotype) decrease the risk of colon cancer by about 20%![ref]
- Another meta-analysis found that two copies of the C677T variant (AA genotype) were protective against prostate cancer.[ref]
- The MTHFR C677T variant decreases the relative risk of retinoblastoma and oral squamous cell cancer.[ref][ref]
- Two copies of the C677T variant (AA genotype) are protective against gastric cancer.[ref]
However, for someone who has cancer, the MTHFR C677T variant is associated with poorer outcomes.[ref]
The MTHFR A1298C variant (GG or GT) is associated with a decreased risk of hypothyroidism.[ref]
The flip side: Folate can also help protect against cancerous mutations in the first place. A lack of folate is linked to double-strand mutations in DNA replication. If those mutations occur in a gene important to cancer prevention, then a tumor can occur.[ref] Thus, folate helps to prevent the mutations that cause cancer, yet in someone who has cancer, folate and methyl groups can fuel cancer cell growth.
Table: Risks and Benefits: Overview of MTHFR Tradeoffs
| Variant | Increased Risks | Potential Benefits |
|---|---|---|
| C677T | Heart disease, stroke, neural tube defects, | ↓ Colon, prostate, gastric cancer |
| depression, migraines, pregnancy complications | ||
| A1298C | Migraines, depression (with two copies) | ↓ Hypothyroidism |
Gene expression and MTHFR:
MicroRNAs are short strands of RNA that can bind to an mRNA strand and block it from being translated into its protein. In cell studies, folate deficiency upregulates miR-22 and miR-149. These two miRNAs bind to the MTHFR mRNA and keep it from being turned into the MTHFR enzyme when folate is lacking. Interestingly, the effects of folate deficiency regulate miRNA differently in cancer cells vs. non-cancerous cells. Those same miRNAs also affect tumor suppressor genes.[ref]
While a lot more research is needed here, the impact of miRNA on both tumor suppressors and MTHFR may be part of why folate is both beneficial for preventing cancer and then detrimental during some types of cancer.
Lifehacks: Diet and Supplements for MTHFR
Knowing that you carry an MTHFR genetic variant can help guide your choice of foods and supplements. By optimizing your diet, you can easily mitigate the risks from the MTHFR variants.[ref]
Let’s explore some diet and supplement options for individuals with MTHFR variants.
Access this content:
An active subscription is required to access this content.
Digging deeper: MTHFR is more than just C677T and A1298C
While the C677T and A1298C are the most well-studied variants, there are several other genetic variants in the MTHFR gene that either increase or decrease the enzyme’s function.
Genotype report: Additional MTHFR variants
Additional variants to check that decrease MTHFR enzyme function:
Access this content:
An active subscription is required to access this content.
Related B Vitamins and Methylation
Frequently Asked Questions (FAQ) About MTHFR:
What is the MTHFR gene, and what does it do?
The MTHFR gene codes for an enzyme (methylenetetrahydrofolate reductase) that converts folate (vitamin B9) into its active form.
Why it matters: This process produces methyl groups used for:
- DNA methylation (gene regulation)
- Neurotransmitter synthesis (brain health)
- Detoxification (removing toxins)
- Heart health (managing homocysteine)
What are the most common MTHFR variants?
The two most common variants are C677T and A1298C. These are single nucleotide polymorphisms (SNPs), not rare mutations, and are found in about half the population.
How can I check if I have an MTHFR variant?
You don’t need expensive genetic testing. You can check your raw data from 23andMe or AncestryDNA for the C677T (rs1801133) and A1298C (rs1801131) SNPs.
What do the C677T and A1298C results mean for enzyme function?
C677T:
- G/G: typical function
- A/G: enzyme function decreased by ~40%
- A/A: enzyme function decreased by 70–80%
A1298C:
- T/T: typical function
- G/T: slightly decreased function
- G/G: enzyme function decreased by ~20%
Are there other MTHFR variants that matter?
Yes, variants like G1793A (rs2274976) can also significantly decrease enzyme function, while others, such as rs9651118 and rs13306560, are associated with positive health outcomes like lower blood pressure or reduced risk of certain diseases.
How do MTHFR variants affect my health?
Variants like C677T and A1298C can reduce enzyme efficiency, potentially leading to higher homocysteine levels and impacting processes like detoxification, neurotransmitter production, and cardiovascular health.
What can I do if I have an MTHFR variant?
Optimizing your diet is key. Increase intake of natural folate (leafy greens, lentils, liver, asparagus, broccoli), choline (egg yolks, beef liver, wheat germ), and betaine (beets, quinoa, spinach). These nutrients can help mitigate the risks associated with reduced enzyme function.
Is folate the same as folic acid?
No. Folate is the natural form found in foods, while folic acid is a synthetic form used in supplements and fortified foods. Not everyone with MTHFR variants processes folic acid efficiently, so focus on natural food sources of folate.
Can increasing dietary folate help with MTHFR?
Yes. Studies show that increasing folate-rich foods can lower homocysteine and inflammatory markers, especially in people with the C677T variant.
Do MTHFR variants affect both men and women?
Yes. For example, MTHFR variants in fathers can also affect fertility and miscarriage risk Dads and MTHFR.
Should I get MTHFR testing done?
Understanding your MTHFR variants can help you dial in your diet and prevent many age-related chronic conditions. Fortunately, the MTHFR SNPs are readily available in your raw data if you’ve already done genetic testing, such as through 23andMe or AncestryDNA.
What should I eat if I have MTHFR C677T?
Optimizing your diet to include plenty of natural sources of folate helps to mitigate the negative effects of the MTHFR variants. Folate-rich foods include leafy greens, lentils, many green vegetables, and liver. Vitamin B12 is also important, along with other B vitamins. Vitamin B12 is only found in animal-based foods, so people eating a primarily vegan diet may need to consider supplementing with a B-complex. Talk with your healthcare provider if you have questions about this.
What does compound heterozygous mean?
When an article on MTHFR mentions ‘compound heterozygous’, it refers to having one copy of the MTHFR C677T variant and one copy of the A1298C variant. This combination reduces the efficiency of the folate-related enzyme by about 50% and makes it important to consume plenty of folate-rich foods in the diet.
Is MTHFR dangerous?
While the variants in MTHFR increase the relative risk of several chronic conditions, they do not cause a specific disease on their own. Instead, it is a combination of diet and environment, which interacts with increased susceptibility due to the MTHFR variants.
Related Articles and Topics:
Histamine Intolerance, MTHFR, and the Methylation Cycle
References:
Pan, Yining, et al. “The Contribution of Ethnicity to the Association of MTHFR Variants C677T and A1298C with Autism Spectrum Disorder: A Meta-Analysis.” Brain Sciences, vol. 16, no. 1, Jan. 2026, p. 93. DOI.org (Crossref), https://doi.org/10.3390/brainsci16010093.
Maurya, Anil Kumar, et al. “Association of MTHFR C677T and A1298C Polymorphisms in Metabolic Alterations, Neuroimaging, and Cognitive Decline in Alzheimer’s Disease: Case–Control and Bioinformatics Insights.” Molecular Neurobiology, vol. 63, no. 1, Dec. 2025, p. 312. Springer Link, https://doi.org/10.1007/s12035-025-05621-x.
Abhinand, P. A., Shaikh, F., Bhakat, S., Radadiya, A., Bhaskar, L. V. K. S., Shah, A., & Ragunath, P. K. (2016). Insights on the structural perturbations in human MTHFR Ala222Val mutant by protein modeling and molecular dynamics. Journal of Biomolecular Structure & Dynamics, 34(4), 892–905. https://doi.org/10.1080/07391102.2015.1057866
Adaikalakoteswari, A., Finer, S., Voyias, P. D., McCarthy, C. M., Vatish, M., Moore, J., Smart-Halajko, M., Bawazeer, N., Al-Daghri, N. M., McTernan, P. G., Kumar, S., Hitman, G. A., Saravanan, P., & Tripathi, G. (2015). Vitamin B12 insufficiency induces cholesterol biosynthesis by limiting s-adenosylmethionine and modulating the methylation of SREBF1 and LDLR genes. Clinical Epigenetics, 7(1), 14. https://doi.org/10.1186/s13148-015-0046-8
Bereket-Yücel, S. (2015). Creatine supplementation alters homocysteine level in resistance trained men. The Journal of Sports Medicine and Physical Fitness, 55(4), 313–319.
Bodenmann, S., Xu, S., Luhmann, U. F. O., Arand, M., Berger, W., Jung, H. H., & Landolt, H. P. (2009). Pharmacogenetics of modafinil after sleep loss: Catechol-O-methyltransferase genotype modulates waking functions but not recovery sleep. Clinical Pharmacology and Therapeutics, 85(3), 296–304. https://doi.org/10.1038/clpt.2008.222
Branched chain amino acids selectively promote cardiac growth at the end of the awake period. (2021). Journal of Molecular and Cellular Cardiology, 157, 31–44. https://doi.org/10.1016/j.yjmcc.2021.04.005
Bueno, O., Molloy, A. M., Fernandez-Ballart, J. D., García-Minguillán, C. J., Ceruelo, S., Ríos, L., Ueland, P. M., Meyer, K., & Murphy, M. M. (2016). Common polymorphisms that affect folate transport or metabolism modify the effect of the mthfr 677c > t polymorphism on folate status. The Journal of Nutrition, 146(1), 1–8. https://doi.org/10.3945/jn.115.223685
Chen, J., Lipska, B. K., Halim, N., Ma, Q. D., Matsumoto, M., Melhem, S., Kolachana, B. S., Hyde, T. M., Herman, M. M., Apud, J., Egan, M. F., Kleinman, J. E., & Weinberger, D. R. (2004). Functional analysis of genetic variation in catechol-O-methyltransferase (Comt): Effects on mRNA, protein, and enzyme activity in postmortem human brain. American Journal of Human Genetics, 75(5), 807–821. https://doi.org/10.1086/425589
Choi, Y., Kim, J. O., Shim, S. H., Lee, Y., Kim, J. H., Jeon, Y. J., Ko, J. J., Lee, W. S., & Kim, N. K. (2016). Genetic variation of methylenetetrahydrofolate reductase (Mthfr) and thymidylate synthase (Ts) genes is associated with idiopathic recurrent implantation failure. PLoS ONE, 11(8), e0160884. https://doi.org/10.1371/journal.pone.0160884
Chung, J.-O., Lee, S.-B., Jeong, K.-H., Song, J.-H., Kim, S.-K., Joo, K.-M., Jeong, H.-W., Choi, J.-K., Kim, J.-K., Kim, W.-G., Shin, S.-S., & Shim, S.-M. (2018). Quercetin and fisetin enhanced the small intestine cellular uptake and plasma levels of epi-catechins in in vitro and in vivo models. Food & Function, 9(1), 234–242. https://doi.org/10.1039/c7fo01576c
Dietary quercetin exacerbates the development of estrogen-induced breast tumors in female ACI rats. (2010). Toxicology and Applied Pharmacology, 247(2), 83–90. https://doi.org/10.1016/j.taap.2010.06.011
El-Hadidy, M. A., Abdeen, H. M., Abd El-Aziz, S. M., & Al-Harrass, M. (2014). Mthfr gene polymorphism and age of onset of schizophrenia and bipolar disorder. BioMed Research International, 2014, 318483. https://doi.org/10.1155/2014/318483
Ganz, A. B., Shields, K., Fomin, V. G., Lopez, Y. S., Mohan, S., Lovesky, J., Chuang, J. C., Ganti, A., Carrier, B., Yan, J., Taeswuan, S., Cohen, V. V., Swersky, C. C., Stover, J. A., Vitiello, G. A., Malysheva, O. V., Mudrak, E., & Caudill, M. A. (2016). Genetic impairments in folate enzymes increase dependence on dietary choline for phosphatidylcholine production at the expense of betaine synthesis. The FASEB Journal, 30(10), 3321–3333. https://doi.org/10.1096/fj.201500138RR
García-Minguillán, C. J., Fernandez-Ballart, J. D., Ceruelo, S., Ríos, L., Bueno, O., Berrocal-Zaragoza, M. I., Molloy, A. M., Ueland, P. M., Meyer, K., & Murphy, M. M. (2014). Riboflavin status modifies the effects of methylenetetrahydrofolate reductase (Mthfr) and methionine synthase reductase (Mtrr) polymorphisms on homocysteine. Genes & Nutrition, 9(6), 435. https://doi.org/10.1007/s12263-014-0435-1
Hall, K. T., Buring, J. E., Mukamal, K. J., Vinayaga Moorthy, M., Wayne, P. M., Kaptchuk, T. J., Battinelli, E. M., Ridker, P. M., Sesso, H. D., Weinstein, S. J., Albanes, D., Cook, N. R., & Chasman, D. I. (2019). Comt and alpha-tocopherol effects in cancer prevention: Gene-supplement interactions in two randomized clinical trials. JNCI: Journal of the National Cancer Institute, 111(7), 684–694. https://doi.org/10.1093/jnci/djy204
Hall, K. T., Loscalzo, J., & Kaptchuk, T. J. (n.d.-a). Systems pharmacogenomics – gene, disease, drug and placebo interactions: A case study in COMT. Pharmacogenomics, 20(7), 529–551. https://doi.org/10.2217/pgs-2019-0001
Hall, K. T., Loscalzo, J., & Kaptchuk, T. J. (n.d.-b). Systems pharmacogenomics – gene, disease, drug and placebo interactions: A case study in COMT. Pharmacogenomics, 20(7), 529–551. https://doi.org/10.2217/pgs-2019-0001
Hall, K. T., Nelson, C. P., Davis, R. B., Buring, J. E., Kirsch, I., Mittleman, M. A., Loscalzo, J., Samani, N. J., Ridker, P. M., Kaptchuk, T. J., & Chasman, D. I. (2014). Polymorphisms in catechol-o-methyltransferase modify treatment effects of aspirin on risk of cardiovascular disease. Arteriosclerosis, Thrombosis, and Vascular Biology, 34(9), 2160–2167. https://doi.org/10.1161/ATVBAHA.114.303845
Husemoen, L. L. N., Skaaby, T., Jørgensen, T., Thuesen, B. H., Fenger, M., Grarup, N., Sandholt, C. H., Hansen, T., Pedersen, O., & Linneberg, A. (2014). MTHFR C677T genotype and cardiovascular risk in a general population without mandatory folic acid fortification. European Journal of Nutrition, 53(7), 1549–1559. https://doi.org/10.1007/s00394-014-0659-2
Hustad, S., Schneede, J., & Ueland, P. M. (2013). Riboflavin and methylenetetrahydrofolate reductase. Landes Bioscience. https://www.ncbi.nlm.nih.gov/books/NBK6145/
Jadavji, N. M., Emmerson, J. T., MacFarlane, A. J., Willmore, W. G., & Smith, P. D. (2017). B-vitamin and choline supplementation increases neuroplasticity and recovery after stroke. Neurobiology of Disease, 103, 89–100. https://doi.org/10.1016/j.nbd.2017.04.001
Kang, K. S., Yamabe, N., Wen, Y., Fukui, M., & Zhu, B. T. (2013). Beneficial effects of natural phenolics on levodopa methylation and oxidative neurodegeneration. Brain Research, 1497, 1–14. https://doi.org/10.1016/j.brainres.2012.11.043
Kang, Minsu, et al. “MTHFR Polymorphism Is Associated with Increased Adverse Events and Poor Clinical Outcomes in Gastric Cancer Patients with Adjuvant S-1 Chemotherapy.” Scientific Reports, Mar. 2026. www.nature.com, https://doi.org/10.1038/s41598-026-38429-3.
Li, A., Shi, Y., Xu, L., Zhang, Y., Zhao, H., Li, Q., Zhao, X., Cao, X., Zheng, H., & He, Y. (2017). A possible synergistic effect of MTHFR C677T polymorphism on homocysteine level variations increased risk for ischemic stroke. Medicine, 96(51), e9300. https://doi.org/10.1097/MD.0000000000009300
Li, M.-N., Wang, H.-J., Zhang, N.-R., Xuan, L., Shi, X.-J., Zhou, T., Chen, B., Zhang, J., & Li, H. (2017). MTHFR C677T gene polymorphism and the severity of coronary lesions in acute coronary syndrome. Medicine, 96(49), e9044. https://doi.org/10.1097/MD.0000000000009044
Li, W.-X., Dai, S.-X., Zheng, J.-J., Liu, J.-Q., & Huang, J.-F. (2015). Homocysteine metabolism gene polymorphisms (Mthfr c677t, mthfr a1298c, mtr a2756g and mtrr a66g) jointly elevate the risk of folate deficiency. Nutrients, 7(8), 6670–6687. https://doi.org/10.3390/nu7085303
Li, Y., Qiu, S., Shi, J., Guo, Y., Li, Z., Cheng, Y., & Liu, Y. (2020). Association between MTHFR C677T/A1298C and susceptibility to autism spectrum disorders: A meta-analysis. BMC Pediatrics, 20, 449. https://doi.org/10.1186/s12887-020-02330-3
Liew, S.-C., & Gupta, E. D. (2015). Methylenetetrahydrofolate reductase (Mthfr) C677T polymorphism: Epidemiology, metabolism and the associated diseases. European Journal of Medical Genetics, 58(1), 1–10. https://doi.org/10.1016/j.ejmg.2014.10.004
Lisboa, J. V. de C., Ribeiro, M. R., Luna, R. C. P., Lima, R. P. A., do Nascimento, R. A. F., Monteiro, M. G. C. A., Lima, K. Q. de F., Fechine, C. P. N. dos S., de Oliveira, N. F. P., Persuhn, D. C., Veras, R. C., Gonçalves, M. da C. R., Ferreira, F. E. L. de L., Lima, R. T., da Silva, A. S., Diniz, A. da S., de Almeida, A. T. C., de Moraes, R. M., Verly Junior, E., & Costa, M. J. de C. (2020b). Food intervention with folate reduces tnf-α and interleukin levels in overweight and obese women with the mthfr c677t polymorphism: A randomized trial. Nutrients, 12(2), 361. https://doi.org/10.3390/nu12020361
Lok, A., Bockting, C. L. H., Koeter, M. W. J., Snieder, H., Assies, J., Mocking, R. J. T., Vinkers, C. H., Kahn, R. S., Boks, M. P., & Schene, A. H. (2013). Interaction between the MTHFR C677T polymorphism and traumatic childhood events predicts depression. Translational Psychiatry, 3(7), e288. https://doi.org/10.1038/tp.2013.60
Maurya, Anil Kumar, et al. “Association of MTHFR C677T and A1298C Polymorphisms in Metabolic Alterations, Neuroimaging, and Cognitive Decline in Alzheimer’s Disease: Case–Control and Bioinformatics Insights.” Molecular Neurobiology, vol. 63, no. 1, Dec. 2025, p. 312. Springer Link, https://doi.org/10.1007/s12035-025-05621-x.
Miller, R. J., Jackson, K. G., Dadd, T., Nicol, B., Dick, J. L., Mayes, A. E., Brown, A. L., & Minihane, A. M. (2012). A preliminary investigation of the impact of catechol-O-methyltransferase genotype on the absorption and metabolism of green tea catechins. European Journal of Nutrition, 51(1), 47–55. https://doi.org/10.1007/s00394-011-0189-0
Mthfr gene: Medlineplus genetics. (n.d.). Retrieved August 13, 2021, from https://medlineplus.gov/genetics/gene/mthfr/
Nowak, I., Bylińska, A., Wilczyńska, K., Wiśniewski, A., Malinowski, A., Wilczyński, J. R., Radwan, P., Radwan, M., Barcz, E., Płoski, R., Motak-Pochrzęst, H., Banasik, M., Sobczyński, M., & Kuśnierczyk, P. (2017). The methylenetetrahydrofolate reductase c.c.677 C>T and c.c.1298 A>C polymorphisms in reproductive failures: Experience from an RSA and RIF study on a Polish population. PLoS ONE, 12(10), e0186022. https://doi.org/10.1371/journal.pone.0186022
Rai, V. (2017). Association of C677T polymorphism (Rs1801133) in MTHFR gene with depression. Cellular and Molecular Biology (Noisy-Le-Grand, France), 63(6), 60–67. https://doi.org/10.14715/cmb/2017.63.6.13
Rai, V., Yadav, U., Kumar, P., Yadav, S. K., & Gupta, S. (2017). Methylenetetrahydrofolate reductase A1298C genetic variant & risk of schizophrenia: A meta-analysis. The Indian Journal of Medical Research, 145(4), 437–447. https://doi.org/10.4103/ijmr.IJMR_745_14
Sak, K. (2017c). The Val158Met polymorphism in COMT gene and cancer risk: Role of endogenous and exogenous catechols. Drug Metabolism Reviews, 49(1), 56–83. https://doi.org/10.1080/03602532.2016.1258075
Scoditti, E. (2020). Neuroinflammation and neurodegeneration: The promising protective role of the citrus flavanone hesperetin. Nutrients, 12(8). https://doi.org/10.3390/nu12082336
Stead, L. M., Au, K. P., Jacobs, R. L., Brosnan, M. E., & Brosnan, J. T. (2001). Methylation demand and homocysteine metabolism: Effects of dietary provision of creatine and guanidinoacetate. American Journal of Physiology. Endocrinology and Metabolism, 281(5), E1095-1100. https://doi.org/10.1152/ajpendo.2001.281.5.E1095
The extra virgin olive oil phenolic oleacein is a dual substrate-inhibitor of catechol-O-methyltransferase. (2019). Food and Chemical Toxicology, 128, 35–45. https://doi.org/10.1016/j.fct.2019.03.049
Troesch, B., Weber, P., & Mohajeri, M. H. (2016). Potential links between impaired one-carbon metabolism due to polymorphisms, inadequate b-vitamin status, and the development of alzheimer’s disease. Nutrients, 8(12), 803. https://doi.org/10.3390/nu8120803
Wan, L., Li, Y., Zhang, Z., Sun, Z., He, Y., & Li, R. (2018). Methylenetetrahydrofolate reductase and psychiatric diseases. Translational Psychiatry, 8, 242. https://doi.org/10.1038/s41398-018-0276-6
Wang, L.-J., Lee, S.-Y., Chen, S.-L., Chang, Y.-H., Chen, P. S., Huang, S.-Y., Tzeng, N.-S., Chen, K. C., Lee, I. H., Wang, T.-Y., Yang, Y. K., & Lu, R.-B. (2015). A potential interaction between COMT and MTHFR genetic variants in Han Chinese patients with bipolar II disorder. Scientific Reports, 5, 8813. https://doi.org/10.1038/srep08813
Wang, P., Heber, D., & Henning, S. M. (2012). Quercetin increased the antiproliferative activity of green tea polyphenol (−)-epigallocatechin gallate in prostate cancer cells. Nutrition and Cancer, 64(4), 580–587. https://doi.org/10.1080/01635581.2012.661514
Wu, X., Yang, K., Tang, X., Sa, Y., Zhou, R., Liu, J., Luo, Y., & Tang, W. (2015). Folate metabolism gene polymorphisms MTHFR C677T and A1298C and risk for preeclampsia: A meta-analysis. Journal of Assisted Reproduction and Genetics, 32(5), 797–805. https://doi.org/10.1007/s10815-014-0408-8
Xu, B., Kong, X., Xu, R., Song, Y., Liu, L., Zhou, Z., Gu, R., Shi, X., Zhao, M., Huang, X., He, M., Fu, J., Cai, Y., Li, P., Cheng, X., Wu, C., Chen, F., Zhang, Y., Tang, G., … Huo, Y. (2017). Homocysteine and all-cause mortality in hypertensive adults without pre-existing cardiovascular conditions. Medicine, 96(8), e5862. https://doi.org/10.1097/MD.0000000000005862
Yan, L., Zhao, L., Long, Y., Zou, P., Ji, G., Gu, A., & Zhao, P. (2012). Association of the maternal mthfr c677t polymorphism with susceptibility to neural tube defects in offsprings: Evidence from 25 case-control studies. PLoS ONE, 7(10), e41689. https://doi.org/10.1371/journal.pone.0041689
Yang, B., Fan, S., Zhi, X., Li, Y., Liu, Y., Wang, D., He, M., Hou, Y., Zheng, Q., & Sun, G. (2014). Associations of mthfr gene polymorphisms with hypertension and hypertension in pregnancy: A meta-analysis from 114 studies with 15411 cases and 21970 controls. PLoS ONE, 9(2), e87497. https://doi.org/10.1371/journal.pone.0087497
Zawieja, Emilia, et al. “Betaine Supplementation Modulates Betaine Concentration by Methylenetetrahydrofolate Reductase Genotype, but Has No Effect on Amino Acid Profile in Healthy Active Males: A Randomized Placebo-Controlled Cross-over Study.” Nutrition Research (New York, N.Y.), vol. 127, July 2024, pp. 63–74. PubMed, https://doi.org/10.1016/j.nutres.2024.05.003.
Debbie Moon is a biologist, engineer, author, and the founder of Genetic Lifehacks where she has helped thousands of members understand how to apply genetics to their diet, lifestyle, and health decisions. With more than 10 years of experience translating complex genetic research into practical health strategies, Debbie holds a BS in engineering from Colorado School of Mines and an MSc in biological sciences from Clemson University. She combines an engineering mindset with a biological systems approach to explain how genetic differences impact your optimal health.