Key takeaways:
- ME/CFS is a multi-system disease, not just fatigue, with some being unable to work, housebound, or bedridden.
- Many cases start after an infection such as EBV, West Nile, or COVID-19, while others follow immune stressors like mold or mycotoxin exposure, yet they converge on similar long‑lasting fatigue and pain.
- Studies show core biological changes, including reduced NK cell function, activation of the NLRP3 inflammasome, altered interferon and cytokine signaling, and evidence of mitochondrial and metabolic dysfunction (lactate/pyruvate, haptoglobin).
- There may be multiple subtypes of ME/CFS that result in similar symptoms.
- Genetic studies do not show a single ME/CFS gene but instead highlight multiple pathways that increase susceptibility. Genetic variants in immune and autoimmune regulation, complement system, NK-cell signaling, mitochondrial enzymes, and neurological/circadian genes have been identified in studies.
- The Lifehacks section includes multiple natural ways of influencing the different genetic pathways, which may help with some symptoms.
This article is for you if you have been diagnosed with ME/CFS, post-viral fatigue, or long COVID and want to understand the genetic and immune pathways underlying your condition. If you have genetic raw data from 23andMe, AncestryDNA, or whole genome sequencing, the genotype report below checks your variants in immune, inflammatory, mitochondrial, and neurological pathways linked to ME/CFS susceptibility.
Members will see their genotype report below and the solutions in the Lifehacks section. Consider joining today.What Is ME/CFS? Symptoms, Prevalence, and Why It’s Complex
Myalgic encephalomyelitis, also known as chronic fatigue syndrome or ME/CFS, is a debilitating condition that causes severe fatigue and inability to recover normally from exertion. ME/CFS is a multi-systemic disease affecting somewhere between 0.1% and 2.5% of the population.[ref] In the US, around 1% of the population is diagnosed with ME/CFS.[ref]
In some parts of the world, it is commonly referred to as myalgic encephalomyelitis (ME), while in other areas, such as the US, it is sometimes still called chronic fatigue syndrome (CFS). I’m going to go with ME/CFS here just for consistency.
The symptoms of ME/CFS include:
- chronic exhaustion (more than tired – difficulty staying upright, feeling ill)
- post-exertional malaise, PEM (inability to recover from activity)
- pain and flu-like symptoms
- cognitive dysfunction (brain fog+)
- a general reduction in the overall quality of life, such as being unable to keep a job or regularly attend school
This is more than just feeling tired. The majority of people with ME/CFS are unable to work, and about 25% are homebound or bedridden.[ref]
History of ME/CFS:
You would think that there would be better answers for a condition that has been defined since at least the 1930s. ME/CFS was known as ‘chronic Epstein-Barr virus syndrome’ in the ’80s, pointing towards one of many possible viral causes.[ref]
It hasn’t been a smooth road for people looking for medical help. Chronic fatigue was considered a psychiatric condition that was all in the patient’s head for decades. In fact, ME/CFS was derisively called the ‘yuppie flu’ at one point.[ref][ref] Dr. Anthony Fauci, as the head of the NIAID from the 80s – 2020s, was strongly criticized for a distinct lack of research focus on the condition for more than two decades.[ref]
Viral Triggers: EBV, HHV-6, West Nile, and COVID-19
Different people with ME/CFS report different starting points: EBV or other herpesviruses, West Nile virus, Covid, traumatic injury, or mold/mycotoxin exposure. These triggers all interact with the immune system and may leave a ‘stuck’ danger signal even when the infection is no longer obvious. Beyond viruses, transposable elements and endogenous retroviruses add another layer by mimicking viral signals from within our own DNA.
Link between ME/CFS and infection:
For many, ME/CFS seems to be triggered by an acute infection, usually viral. One study found that almost two-thirds of ME/CFS patients reported an infection-related onset.[ref]
- EBV: Recent studies point to latent or reactivated Epstein-Barr virus being implicated in a portion of ME/CFS patients. One study found that 24% of chronic fatigue patients had DNA from the Epstein-Barr virus in their plasma, in comparison with only 4% of the control group.[ref]
- Herpes simplex, CMV: Other studies point to a variety of other possible viral triggers for ME/CFS, including herpes simplex or cytomegalovirus, but not all studies agree.[ref]
- HHV-6: Researchers examined brain and spinal cord postmortem tissue samples from ME/CFS patients. They found that HHV-6 (human herpesvirus 6) miRNA was abundant in these tissues, in stark contrast to control tissue samples. HHV-6 is a latent virus that remains in the body throughout life, integrating into the telomeres of host cell chromosomes.
- Additionally, the researchers found active EBV infection in the ME/CFS tissue. [ref]
Related article: Epstein-Barr virus genetic susceptibility
Non-viral root causes of ME/CFS:
It’s important to note that not all ME/CFS cases can be traced to a viral infection. Again, these causes interact with the immune system in various ways.
Fungal and mycotoxin connection to ME/CFS:
While much of the research on ME/CFS focuses on viral persistence, another possibility is that the immune response can be altered due to mold exposure. A study in Florida of 236 patients with ME/CFS found a high presence of myocotoxins in their urine. OTA and aflatoxins were the most common mycotoxins found.[ref] Another study involving 112 ME/CFS patients in Kansas City found that 93% of chronic fatigue patients had mycotoxins in their urine, while none of the healthy control group (n=55) had mycotoxins.[ref]
Mycotoxins are the metabolites produced by certain fungi (molds) that are harmful if inhaled, consumed, or exposed to. Many studies show that mycotoxin exposure can significantly alter immune response.[ref]
Transposable Elements: Viral infection mimicry
One area of research in ME/CFS ties the immune system activation to the viral DNA encoded in our human genome. Not everyone with ME/CFS, fibromyalgia, or similar diagnoses has altered levels of viruses or a known viral onset. A possible answer may lie in the ‘fossil viruses’ encoded into the human genome.
Your DNA – your genome – is the ‘code’ for your genes, but most of your DNA doesn’t code for protein-coding genes. In fact, about 45% of the genome is made up of transposable elements. These sections of DNA move around within the genome and are sometimes called ‘jumping genes’.
Methylation represents one method of controlling which genes – or which sections of the genome – get transcribed into RNA. This is just one epigenetic way cells control which genes turn off or on. In methylation, a methyl tag that binds to the right spot on your DNA turns off a gene.
A recent study looked at methylation patterns in people with fibromyalgia and ME/CFS. The researchers found that methylation patterns were different from the healthy control group, and one large difference was in the methylation patterns in the transposable elements.
The study focused on a section known as HERV-K, which is an endogenous retrovirus found in the non-coding part of the genome. An endogenous retrovirus is a section of our human DNA that is likely the integration of a virus or provirus incorporated into the genome millions of years ago. Researchers estimate that about 4-8% of the human genome’s composition includes endogenous retroviruses. (Here’s a good article on HERVs, if you’re interested)
The results showed that people with fibromyalgia had increased expression of the HERV-H, HERV-K, and HERV-K. This corresponded with increased interferon-beta and interferon-gamma. The researchers theorize that an infection mimicry state could be a cause of fibromyalgia and ME/CFS.[ref]
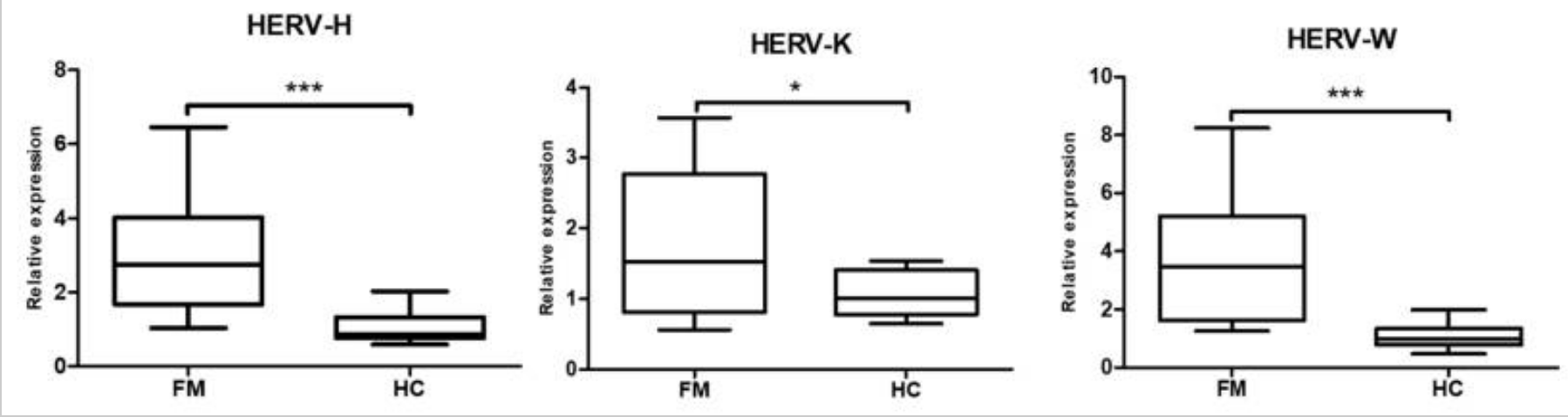
A 2025 study from the University of East Anglia and Oxford BioDynamics showed that a blood test, called EpiSwitch, was fairly accurate for diagnosing ME/CFS. The study found epigenetic markers that were unique to ME/CFS patients.[ref]
Autoimmune component:
An autoimmune component or response to traumatic injury likely exists for some ME/CFS patients. Research shows genetic variants with links to autoimmune diseases also have links to ME/CFS in patients with an initial viral cause.[ref]
Past infections, reactivations:
Everyone has a latent viral presence from a variety of different viruses. For some, the reactivation or continued, low-level presence of a virus or bacteria may cause chronic immune system changes.
Chronic Active Epstein-Barr virus:
Epstein-Barr is a herpes virus that causes few symptoms in children, but in teens and young adults, it causes mononucleosis. The virus is spread through contact with saliva, and almost everyone (>90% of people) has it by the time they are an adult. The virus sticks around in a latent form for the rest of your life.
Chronic active Epstein-Barr virus (CAEBV) is a rare syndrome where the virus stays active, causing long-term illness. Patients have prolonged mono-like symptoms, and most show unusual T-cells or natural killer cells.[ref] Rare mutations linked to immunodeficiency have been tied to chronic active Epstein-Barr.[ref]
For some with ME/CFS, it starts not long after mononucleosis and can be directly tied to EBV infection — and infection that doesn’t go away rather than being reactivated.[ref]
For most people, though, the Epstein-Barr virus hangs out in a latent state and avoids immune system detection in unique ways. One of the proteins coded for by the virus is very similar to human IL-10, which is an immune system molecule that dampens the immune response. Additionally, Epstein-Barr is an enveloped virus, and the host’s cell membrane creates the envelope. So it escapes detection by looking like ‘self’.[ref] Autoimmune diseases, such as lupus, rheumatoid arthritis, and Sjögren’s syndrome, are linked to Epstein-Barr as a contributing factor.[ref]
Prolonged fatigue after West Nile Virus:
Research shows that a lot of people experience prolonged fatigue for months to years after having West Nile virus. West Nile is a mosquito-borne illness that is prevalent across the US in some years, with an estimated 3 million people in the US having the disease by 2010.[ref]
While the majority of people with West Nile virus are asymptomatic, about 20% of people will experience fever, headache, weakness, and muscle aches. Around 1% of people will develop severe neurological symptoms, including encephalitis and myocarditis. [ref][ref]
- In a study of people with West Nile in Houston, TX, about 20% of the symptomatic people in the study still had continuing fatigue up to 8 years later. The study participants with continuing symptoms also had elevated cytokine levels.[ref]
- Other research points to almost half of the people with more severe cases of West Nile having long-term symptoms from it. The NLRP3 inflammasome activation is important in fighting off West Nile, as is interferon.[ref]
Long COVID and ME/CFS: Overlap, EBV Reactivation, and Shared Genetics
Early on in the pandemic, a research study found that about 3% of long covid patients also met the criteria for ME/CFS. The researchers also included PTSD in the study and found no overlap between patients with PTSD and CFS/ME.[ref] Interestingly, a May 2021 study found that 67% of the long COVID patients had reactivated Epstein-Barr virus titers (compared to only 10% in the control group). Symptoms of Epstein-Barr virus (fatigue, brain fog, sleep problems, muscle aches, headaches, gastrointestinal issues, and skin rash) overlap completely with long COVID symptoms.[ref]
Read more about long Covid genes and studies here.
Core Biology: Immune, Mitochondrial, and Metabolic Changes in ME/CFS
The immune system ties the possible triggers together. A lingering or latent infection points toward a change in the immune response. Similarly, an autoimmune, traumatic injury, or continued mold exposure could also trigger a change in the immune response. This leads to altered cellular energy and chronic fatigue.
| Finding | System / pathway | What changes in ME/CFS | Why it might matter for symptoms |
|---|---|---|---|
| Reduced NK cell function | Innate immunity | Lower NK cell cytotoxicity, altered TRPM3/TRPM8 channels on NK cells | Impaired clearance of virally infected cells and tumors; may contribute to persistent infections and fatigue |
| NLRP3 inflammasome activation | Innate immunity / inflammasome | Activation linked to fatigue behavior in mouse models and implicated in ME/CFS and MS | Drives IL‑1β and IL‑18 release, pyroptosis, blood–brain barrier changes, sickness behavior |
| Mitochondrial damage and interferon | Mitochondria / cytokines | Damaged mitochondria leak mtDNA, activating interferon and other cytokines; some viruses trigger mitophagy | Creates a feedback loop of immune activation and impaired ATP production, sustaining fatigue |
| T cell exhaustion | Adaptive immunity | CD8+ T cells show exhaustion markers and metabolic reprogramming; Treg differences vs long COVID | Exhausted T cells respond poorly to ongoing antigens, possibly allowing low-level viral or inflammatory signals to persist |
| Lactate/pyruvate alterations | Metabolism / mitochondria | Evidence of impaired pyruvate dehydrogenase, elevated lactate in CSF and blood, correlating with PEM | Points to a metabolic bottleneck after exertion, matching clinical post-exertional crashes |
| Haptoglobin isoform changes | Oxidative stress / RBC handling | Post-exertional drop in specific haptoglobin isoforms in ME/CFS vs controls | May reflect altered handling of oxidative stress after exertion, contributing to prolonged malaise |
Let’s dive into the research studies that show how the immune response is altered in ME/CFS patients. Keep in mind that studies tend to look at just one aspect of the immune response, so we need to pull together the threads from different studies to weave a fuller picture of what is going on.
Natural killer (NK) cells and impaired calcium channels in ME/CFS:
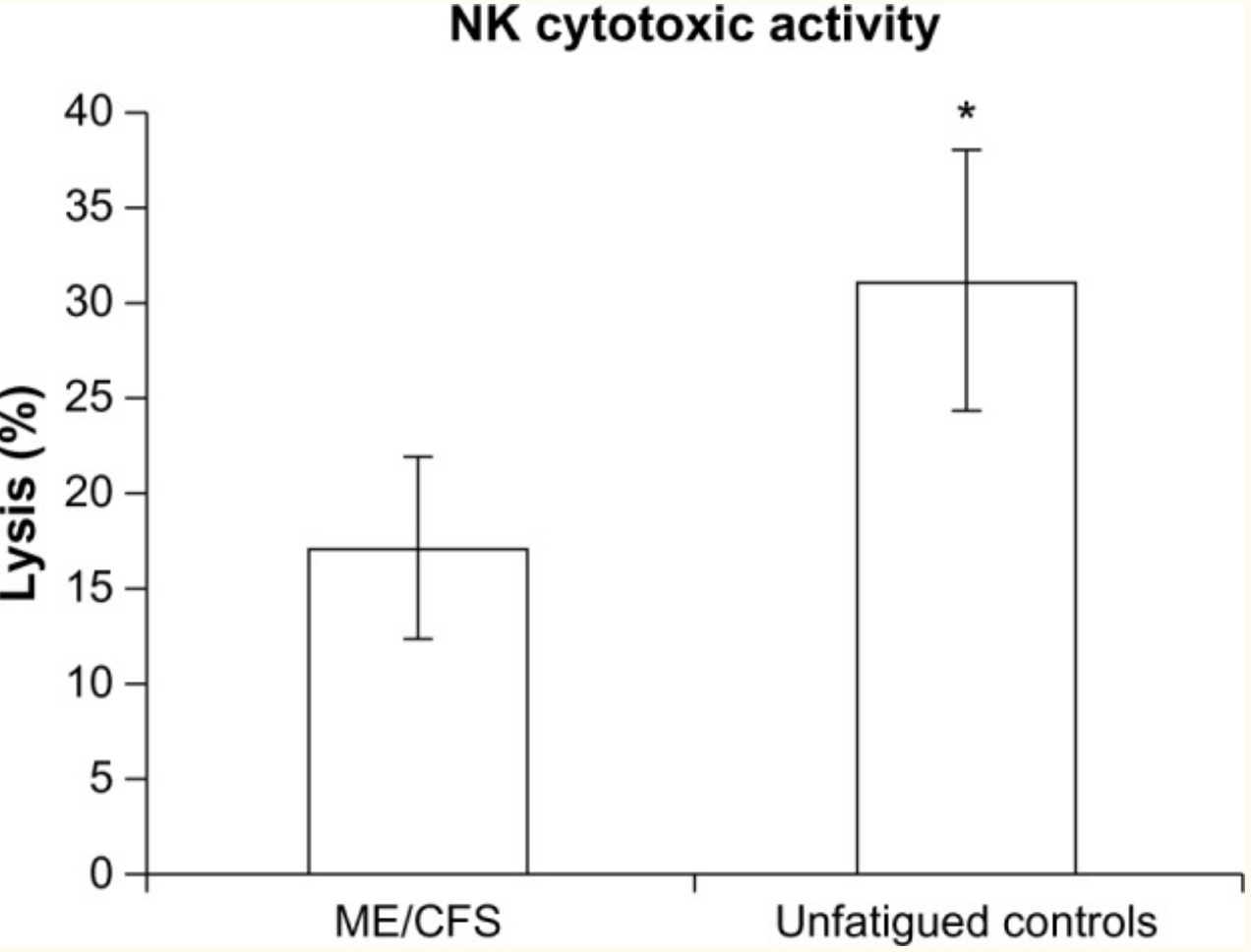
One fairly consistent finding for ME/CFS patients is a reduction in natural killer cells. A type of white blood cell, natural killer (NK) cells are part of the innate immune response. They target tumor cells for destruction and are also vital for responding to viral infections by destroying infected cells.[ref]
Calcium ions play numerous roles in the activation of different cellular processes. Researchers have found that patients with ME/CFS are likely to have impaired calcium ion channel function via the TRPM3 channel. They link this to reduced TRMP3 function on natural killer cells in people with ME/CFS compared to healthy controls. Several variants in the TRPM3 gene are more often found in people with ME/CFS.[ref]
NLRP3 inflammasome activation and fatigue:
Another innate immune system component, the NLRP3 inflammasome, is also implicated in fatigue syndromes, including ME/CFS and MS.[ref][ref]
NLRP3 is a linchpin at the start of the immune cascade. Essentially, when NLRP3 activates, it causes caspase-1 activation, which in turn activates interleukin-1β (IL-1β) and interleukin-18 (IL-18). IL-1β and IL-18 are proinflammatory cytokines that cause rapid cell death (pyroptosis) as well as initiate other inflammatory responses. IL-1β also alters the integrity of the blood-brain barrier.[ref]
NLRP3 activation can develop due to microbes (including coronaviruses) as well as ethanol, amyloid-beta (Alzheimer’s), and alpha-synuclein (Parkinson’s).
Researchers often use animal models in chronic fatigue research, and animal studies point to the continued activation of NLRP3.
- Using a mouse model of ME/CFS but with the NLRP3 gene inactivated, the researchers showed that the mice had reduced fatigue behavior after forced exercise. The knock-out model also showed decreased IL-1β.
- On the other hand, mice with NLRP3 intact had increased fatigue behavior and increased IL-1β after repeated forced exercise.[ref]
In addition to NLRP3 being activated by viral pathogens, it can also be triggered by mitochondrial dysfunction.[ref] This brings us to the next topic of mitochondrial dysfunction in chronic fatigue.
Related article: NLRP3 inflammasome genetics
Mitochondria, cellular energy, and interferon:
Mitochondria are the organelles responsible for the majority of cellular energy — the ‘powerhouse’ of the cell.
When you learn about mitochondria, the Krebs cycle, and the electron transport chain in biology class, a dimensional picture forms of a cellular battery cranking out energy in the form of ATP.
Cellular energy is, of course, vital to well-being. Your muscles and brain can’t work well when lacking energy. So a mitochondrial connection to fatigue is common sense. But why would the fatigue continue after an illness resolves?
The role of mitochondria in the cell goes beyond just generating energy. Mitochondria are constantly changing – fusing together and splitting into two. When a mitochondrion is no longer functioning correctly, it degrades and recycles via a process called mitophagy (autophagy of mitochondria).
In addition to activating the NLRP3 inflammasome cascade (IL-1β and IL-18), damaged mitochondria can trigger interferons and other pro-inflammatory cytokines.
Specifically, when mitochondrial DNA leaks, it triggers the immune response, including interferon activation, as a danger signal from the damaged mitochondria.[ref]
Some viral infections also trigger mitophagy, or mitochondrial destruction, which works to help the virus evade the immune response.[ref]
Interleukin-2 is an inflammatory cytokine that plays an integral role in activating T cells, NK cells, and macrophages. Dysregulation of IL-2 is reported in ME/CFS (chronic fatigue) patients.
- IL-2 is downregulated, along with other markers for CD8+ T cell exhaustion, in some ME/CFS patients. The researchers also found differences between Tregs in ME/CFS compared to long Covid.[ref]
- However, other studies show IL2 is significantly higher in extracellular vesicles in ME/CFS patients.[ref]
Related article: IL-2 variants and immune balance
Thus, we have the circle of immune system activation causing mitochondrial damage, as well as mitochondrial damage causing immune system activation. In a trap of decreased cellular energy, fatigue persists.
Let’s move on to some of the other mechanisms seen in ME/CFS that add to the emerging picture of how and why the condition persists.
MicroRNAs change gene expression:
MicroRNAs (miRNAs) are short strands of RNA that can bind to mRNA and prevent it from being translated into its protein. This is one way that the cells moderate gene expression — fine-tuning how much of a specific protein is made from a gene.
The idea that certain cellular processes are turned down in ME/CFS makes sense and fits with symptoms and the research. MiRNAs are one way that this could happen.
One key here is that each miRNA can affect gene expression for multiple genes. There are about 5,000 different miRNAs, with different miRNAs active in specific cell types. MiRNAs can also be released from cells in extracellular vesicles, circulating and affecting the body as a system.
Here’s my full article on miRNA if you want more background.
Let’s take a look at some of the research studies on miRNA changes in ME/CFS patients:
- Endothelial function and eNOS: A 2021 study looked at five specific miRNAs involved in endothelial function. The results showed: “miR-21, miR-34a, miR-92a, miR-126, and miR-200c are jointly increased in ME/CFS patients compared to healthy controls.”[ref] MiR-21 and miR-126 regulate eNOS and endothelial function.[ref] MiR-21 is found abundantly in macrophages and plays a key role in regulating the anti-inflammatory response in macrophages.[ref]
- Immune response, cellular energy: A study involving 40 ME/CFS patients and 20 healthy controls found five upregulated miRNAs (miR-127-3p, miR-142-5p, miR-143-3p, miR-150-5p, and miR-448), and one downregulated (miR-140-5p). This study chose to look at miRNAs that had previously been identified as differentially expressed in people with ME/CFS. A pathway analysis of how those specific miRNAs interact shows that they affect cytokine signaling, mTOR, and the immune response to microbial infections.[ref]
To add a little more complexity here, miRNAs are affected by SNPs in their DNA as well as SNPs in the mRNAs that they bind to.
Haptoglobin changes:
Haptoglobin is a hemoglobin-binding protein that mitigates oxidative damage when red blood cells are damaged. A study in ME patients with post-exertional stress (PEM) showed that they have altered haptoglobin isoforms when compared to a normal control group. Haptoglobin is encoded by two genes, HP1 and HP2, which then form six different isoforms of haptoglobin. At baseline, both the ME/CFS patients and a healthy control group had similar protein levels for 107 proteins, but after a stress test, the ME/CFS patients showed significant reductions in haptoglobin proteins that weren’t seen in the control group.[ref]
ME/CFS and T cell exhaustion:
A 2024 study that analyzed T cell subsets found T cell exhaustion to be common in ME/CFS patients.[ref] Prior research had also shown that CD8+ T cells had metabolic reprogramming indicative of T cell exhaustion.[ref][ref][ref]
Related article: T cell exhaustion and checkpoint genes
Lactate and pyruvate alterations:
Pyruvate and lactate are integral to the mitochondrial processes for ATP production. Both have been identified as being abnormal in some people with ME/CFS.
- A December 2016 study of metabolites in ME/CFS (Chronic Fatigue Syndrome) patients found indicators showing impaired pyruvate dehydrogenase, inadequate ATP production, and elevated lactate. [ref]
- Studies also show increased lactate in the cerebrospinal fluid in ME/CFS.[ref]
- Elevated lactate levels correlate with the presence of post-exertional malaise (PEM).[ref]
Related articles: Lactate and Pyruvate
Changes in cardiac output and cerebral blood flow:
A 2024 study showed that 91% of the ME/CFS patients in the study had an abnormal cardiac output and abnormal cerebral blood flow in response to going from lying down to standing up (tilt table). The researchers found that the ME/CFS patients didn’t have POTS, which causes changes in heart rate when standing up, but instead, there was a lack of the normal compensatory blood vessel vasodilation to maintain blood flow to the brain. This resulted in the blood flow to the brain being reduced, but without the change in heart rate that is seen in POTS when the body is compensating. This suggests that the endothelium – the lining of the blood vessels in the brain – isn’t reacting normally. [ref]
Related article: POTS genetic susceptibility
ME/CFS Genetics: Genes, Variants, and Susceptibility Pathways
Essentially, by looking at how different genetic variants affect the statistical risk of ME/CFS, we can get a good idea of which pathways are likely to play a causal role in the condition. In general, the genetic studies on ME/CFS show lots of connections to the immune response and inflammatory cytokines. (Members will see their SNPs below for these in the Genotype Report section)
DecodeME: 2025 GWAS on ME patients and immune response
A 2025 genome-wide association study (GWAS) called DecodeME looked at the genetic risk associated with ME/CFS. The study identified 8 genetic locations that were associated with either a slight increase or a slight decrease in the relative risk of ME/CFS. Three of the genes, BTN2A2, OLFM4, and RABGAP1L, are involved in the response to viral or bacterial infection.
For an individual, this genome-wide association study is not going to tell them much about why they have ME/CFS, since the relative risk shown is pretty small. However, from a big picture scientific point of view, the pathways involved — particularly the immune response to viruses — are very interesting.
Actionable ME/CFS study findings:
While the DecodeME study was important for research into the immune system pathways, prior studies have also identified important genes related to ME/CFS susceptibility. Understanding these genes and your genotypes may help you further refine where to focus for healing.
Inflammatory cytokine elevations:
Genetic studies also show that variants in the TNF gene, which encodes TNF-alpha, increase susceptibility to ME/CFS. These variants are associated with increased TNF response to pathogens or mycotoxins.[ref]
The NLRP3 inflammasome helps to rev up a strong inflammatory response. Genetic variants in the CAIS1 gene that encodes NLRP3 are associated with an increased relative risk of ME/CFS.
These inflammatory cytokine elevations also show up in blood tests in some ME/CFS. It’s likely that this adds to the symptoms of fatigue and overall immune system dysregulation.
TRP ion channels:
A 2016 study involving gene expression on natural killer cells of people with ME/CFS compared to a healthy control group found that there were differences in several of the TRP ion channels, including TRPM8 and TRPM3. [ref] TRPM8 is a thermoreceptor that is activated by extreme cold or heat. It is also activated by menthol and tacrolimus.[ref]. TRPM3 is a similar ion channel that is activated by noxious heat as well as pregnenolone sulfate. It plays a role in pain signaling and is found in sensory neurons.
Complement and innate immune response:
The body has multiple ways that it can fight off pathogens – from the adaptive immune response (B cells, T cells), to the innate immune system (Natural killer cells, interferon, mast cells), and the complement system (bridges the innate and adaptive response). Variants (below) in the complement pathway increase susceptibility to ME/CFS, and patients with ME/CFS are about twice as likely to have mannose-binding lectin deficiency, which increases susceptibility to fungal and viral infections.[ref]
IDO trap: IDO2 gene
One theory of ME/CFS is that there is a metabolic trap in the tryptophan pathway that shifts the way tryptophan is metabolized. People with IDO2 gene mutations are more likely to rely on IDO1, which then feedsback to inhibit tryptophan conversion under certain conditions. This results in a pathological state with high cellular tryptophan and low kynurenine.[ref] There’s still more research that needs to be done here, so check back for updates.
Related article: Tryptophan metabolism, IDO1, IDO2
Glutamate receptor changes:
A genomics study using data from 40 patients with ME/CFS alongside a matched, healthy control group looked at over 100,000 different genetic SNPs as well as gene expression for 20,000 genes. The results showed that a genetic variant in the GRIK2 gene was more common in people with ME/CFS, and that the gene expression for that gene was decreased by 10-fold in patients compared to controls.[ref]
Related article: Glutamate receptor gene variants
Circadian clock gene, NPAS2:
The study mentioned above also identified a circadian clock gene variant as differentially expressed (10-fold difference) and a variant in the gene as increasing susceptibility to ME/CFS.[ref]
Rare mutations:
A 2025 Stanford study using the HEAL2 deep learning framework analyzed whole genome sequences from 1,075 ME/CFS patients. The whole genome sequencing found 115 loss-of-function mutations that were likely to be playing a role in ME/CFS for the individuals who had the mutations. These genes were in immune cells and had a role in synaptic function in the central nervous system. Patient data showed reduced expression of these pathways in B cells and T cells.[ref]
This study points to the individuality of underlying genetic dysfunction that can affect multiple pathways, resulting in the symptoms of ME/CFS. In other words, the underlying cause for one person with ME/CFS is likely not to be the same as for another person with ME/CFS. Similarly, there is likely to be individuality in response to treatments.
Genotype Report: ME/CFS
Genetic studies do not point to a single ‘ME/CFS gene’ but instead highlight pathways: immune and autoimmune regulation, inflammatory cytokines, complement and innate defense, neuronal/circadian signaling, and mitochondrial enzymes.
Together, these variants can tilt the system toward stronger or prolonged responses to infections or toxins. This may make it harder to return to baseline after a trigger. Note that many of these immune system genetic variants also overlap with autoimmune diseases and the innate response to different pathogens.
Access this content:
An active subscription is required to access this content.
Lifehacks: From pathways to possible interventions
Please talk with your doctor before starting any diet or supplements. What works in research studies may not be right for your individual case!
Interventions based on genetic pathways:
Please note that there are no direct clinical trials showing that inhibiting these pathways will help with ME/CFS. Most of this is theoretical, based on the genetic connection. Where there are specific research studies on ME/CFS it is noted.
Naltrexone (TRMP3 variant):
A 2019 cell study using CFS/ME cells from 8 different patients showed that Naltrexone restored TRPM3 function.[ref] Keep in mind that this is just a cell study (not a randomized, double-blind, placebo-controlled trial), but it may be worth talking with your doctor about this study, especially if you carry a TRPM3 variant linked to CFS.
Related article: Naltrexone: LDN and Your Genes
Saffron (TNF):
Saffron has been shown in studies to modulate TNF-alpha levels, thus impacting chronic inflammation.[ref] It has been shown to reduce fatigue in autoimmune disease patients.[ref]
Luteolin (PTPN22 variant, TNF variant):
Luteolin has been shown to decrease elevated TNF levels significantly.[ref]
Related article: Read all about luteolin.
(Also check COMT — possible interaction with slow COMT)
Hesperidin (PTPN22 variant, TNF variant):
A natural flavonoid from citrus fruits, hesperidin, inhibits the release of TNF-alpha.[ref][ref]
Related article: Read more about hesperidin.
Rosmarinic acid (PTPN22 variant, TNF variant, NLRP3):
Found in rosemary, basil, holy basil, lemon balm, and perilla oil, rosmarinic acid is a natural TNF-alpha inhibitor.[ref] Carnosic acid is a terpene also found in rosemary and rosemary essential oil. Studies show that carnosic acid attenuates NLRP3 overactivation.[ref]
Curcumin (TNF variant):
A component of the spice turmeric, curcumin is a natural TNF-alpha inhibitor.[ref] (Read more about curcumin. Also check COMT)
Related article: TNF-alpha and natural inhibitors.
Ketone metabolites (NLRP3 variant):
β-hydroxybutyrate, a metabolite formed in ketosis, blocks the NLRP3 inflammasome.[ref] The body produces β-hydroxybutyrate on a ketogenic diet (low carb, high fat). Additionally, you can buy β-hydroxybutyrate supplements.
Parthenolide (NLRP3):
Parthenolide, a plant compound found in feverfew, is a medicinal plant traditionally used to treat fever and inflammation. Parthenolide has been shown to inhibit the activation of the NLRP3 inflammasome.[ref]
Plasmalogens (NLRP3, TNF):
Plasmalogens are a unique type of phospholipid and an essential component of cell membranes. They can also be used in the cell as an antioxidant to neutralize oxidative stress, especially important in the brain.
Plasmalogen levels are significantly decreased in people with ME/CFS and for some long Covid patients.[ref] Another study on ME/CFS patients also had a similar finding of patients having uniformly low plasmalogen levels. The study looked at hundreds of metabolic markers, and the effect on ethanolamine plasmalogen levels was seen across the board in the ME/CFS patients.[ref]
Plasmalogen supplements are available from a few manufacturers online. One in particular to look up is ProdromeNeuro.
Related article: Plasmalogens and your genes
Fluids, salt, and compression garments: (possibly ties to GRIK2, TRPM3)
A 2024 study showed that most of the ME/CFS patients in the study had reduced blood flow to the brain upon standing. This was due to the blood vessels in the brain not correctly responding to cardiac output. The researchers suggested that increasing circulation by drinking more water, getting more salt, and wearing compression garments may help. In addition, there are prescription medications that may improve cardiac outflow. [ref]
Boosting overall mitochondrial function:
These interventions have research studies showing efficacy in ME/CFS patients.
CoQ10:
People with ME/CFS often have lower levels of CoQ10, another molecule important in mitochondrial health. Supplementing with 150mg of ubiquinol, a form of CoQ10, improved cognitive function in people with ME/CFS.[ref]
Related article: CoQ10 genes
Sleep / Circadian Rhythm:
Sleep disruption is often a component of ME/CFS, fibromyalgia, and other long-term fatigue and pain syndromes. The NPAS2 gene variant and changes in gene expression seen in ME/CFS show that circadian rhythm can play an integral role in the condition.
Light in the blue wavelengths sets the circadian clock, and exposure to blue light after dark can interfere with the circadian rhythm.
Important actions here: Blocking blue light before bedtime, sleeping on a regular schedule, and getting outside or having exposure to bright light in the morning.
Related article: Circadian rhythm genes
L-carnitine:
Carnitine is important in producing energy in the mitochondria, and people with ME/CFS often have low carnitine levels. A clinical trial showed that l-carnitine supplementation improved the studied parameters in people with ME/CFS after a month.[ref]
Related article: Carnitine genes
Nutrient interactions: Check your genetic susceptibility
Several studies have specifically looked for nutritional deficiencies as the root cause of ME/CFS without finding much of note as a single underlying cause.
However, studies do show that nutrient deficiencies can interact with and exacerbate the symptoms of ME/CFS. Thus, testing or looking for genetic reasons for nutrient deficiencies can help to support overall quality of life and promote healing.
The links below are to additional Genetic Lifehacks articles to check for variants in these pathways. You can use the information to prioritize what is likely to be important for you to look at first, but you can’t eliminate anything based just on your genes.
Vitamins and minerals noted in studies include: [ref]
- Vitamin C
- B-complex (B6, riboflavin, folate, B12, B5, thiamine)
- Magnesium
- Zinc
- L-carnitine
- L-tryptophan (read section on IDO block in the article)
- CoQ10
A website called Born Free has protocols for reversing nutrient deficiency and mitochondrial dysfunction. It may be worthwhile to read through it and talk with your doctor about whether the protocols would be right for your situation.
Histamine connections (anecdotal):
Many with ME/CFS report problems with mast cell activation or histamine intolerance symptoms.
If you have delayed reactions to foods, unexplained itching or hives, sinus drainage, headaches after foods, heartburn, gastrointestinal issues, or early morning insomnia, read through the articles on histamine intolerance and mast cell activation.
Here are the genetic variants related to histamine intolerance (check for multiple variants in AOC1 plus HNMT).
| Gene | RS ID | Your Genotype | Effect Allele | Effect Allele Frequency | Notes About Effect Allele |
|---|---|---|---|---|---|
| AOC1 | rs10156191 | -- | T | 0.26 | Reduced production of DAO |
| AOC1 | rs2052129 | -- | T | 0.23 | Reduced production of DAO |
| AOC1 | rs1049742 | -- | T | 0.07 | Reduced production of DAO |
| AOC1 | rs1049793 | -- | G | 0.31 | Reduced production of DAO |
| AOC1 | rs2071514 | -- | A | 0.19 | possibly slightly higher DAO |
| HMNT | rs1050891 | -- | A | 0.79 | Reduced breakdown of serum histamine |
| HMNT | i3000469 | -- | T | 0.1 | Reduced breakdown of serum histamine |
| HMNT | rs2071048 | -- | T | 0.58 | T/T: Reduced breakdown of serum histamine (common) |
| HMNT | rs11558538 | -- | T | 0.1 | Reduced breakdown of serum histamine |
| HDC | rs2073440 | -- | G | 0.02 | Decreased histamine production |
| HDC | rs267606861 | -- | A | 0 | rare pathogenic mutation, linked to Tourettes |
| HRH1 | rs901865 | -- | T | 0.17 | Increased H1 receptor, increased asthma risk |
| HRH2 | rs2067474 | -- | A | 0.04 | Decreased H2 receptor |
| HRH4 | rs11662595 | -- | G | 0.09 | decreased HRH4 activation (receptor dysfunction), increased risk of progression in non-small cell lung cancer |
| MTHFR | rs1801133 | -- | A | 0.33 | MTHFR C677T, decreased enzyme function, affects methylation cycle |
| MTHFR | rs1801131 | -- | G | 0.3 | MTHFR A1298C, slightly decreased enzyme function, slightly affects methylation cycle |
Multiple chemical sensitivity:
Another condition that overlaps with ME/CFS in the literature is multiple chemical sensitivity. The CDC classifies (or used to classify) multiple chemical sensitivity as a chemical intolerance in people with ME/CFS, acknowledging an overlap in patients and symptoms.[ref]
Related article: Multiple chemical sensitivity
Recap of your ME/CFS genes:
| Gene | RS ID | Your Genotype | Effect Allele | Effect Allele Frequency | Notes About Effect Allele |
|---|---|---|---|---|---|
| PTPN22 | rs2476601 | -- | A | 0.08 | increased risk of autoimmune diseases, increased susceptibility to CFS/ME (in patients with infectious disease onset) |
| CTLA4 | rs3087243 | -- | G | 0.57 | increased risk of autoimmune conditions, decrease CTLA4 expression; increased risk of CFS/ME (patients with infectious disease onset only) |
| TNF | rs1799724 | -- | T | 0.12 | higher TNF-alpha levels, increased susceptibility to CFS/ME |
| INFG | rs2430561 | -- | A | 0.39 | decreased risk of ME/CFS |
| NLRP3 | rs35829419 | -- | A | 0.03 | Increased susceptibility to fatigue / pain after EBV or other viruses |
| NLRP3 | rs121908147 | -- | A | 0.007 | mutation linked to autoinflammatory disease in combo with other genes (rare) |
| TRPM8 | rs11563204 | -- | A | 0.2 | increased risk of CFS/ME (cold, menthol receptor) |
| TRPM3 | rs6560200 | -- | C | 0.5 | CC: common genotype, higher risk of CFS/ME (Naltrexone may work?) |
| TRPM3 | rs1891301 | -- | T | 0.51 | TT: higher risk of CFS/ME |
| CFB | rs4151667 | -- | A | 0.03 | AA: increased risk of CFS/ME |
| CFH | rs1061170 | -- | C | 0.37 | CC: increased risk of AMD, higher immune response; TT: increased risk of CFS/ME |
| GRIK2 | rs2247215 | -- | G | 0.26 | increased relative risk of ME/CFS |
| NPAS2 | rs356653 | -- | A | 0.31 | increased relative risk of ME/CFS |
[/mepr-show]
Related Articles and Topics:
BH4: Tetrahydrobiopterin Synthesis, Recycling, and Genetic SNPs
Is Inflammation Causing Your Depression and Anxiety? The Science Behind the Link
HPA Axis Dysfunction: Understanding Cortisol and Genetic Interactions

