Key takeaways:
~ There are two cannabinoid receptors, CB1 and CB2, that affect your response to cannabis and endocannabinoids.
~ Genetic variants in CB1 and CB2 affect your risk of cannabis abuse disorder.
Your Endocannabinoid System
Before we get into cannabis, let’s look at the body’s built-in system for cannabinoids. Your body has an endocannabinoid system that involves your own naturally produced molecules that bind to the same receptors as the psychoactive component in cannabis.
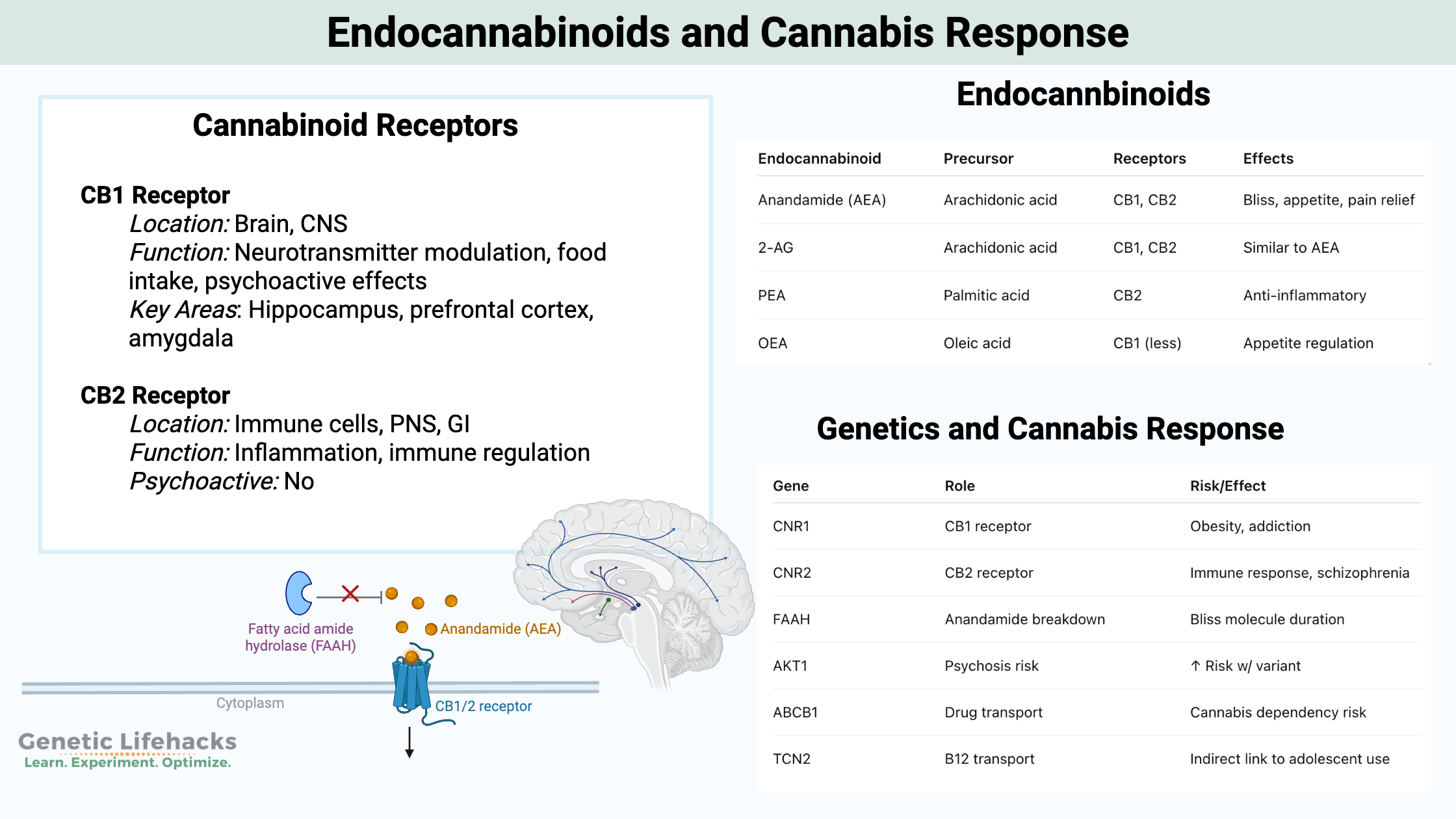
Cannabinoid Receptors:
The two cannabinoid receptor genes in the body were first sequenced in the early 1990s. The receptors, called CB1 and CB2, have different roles in the body.
| CB1 Receptor | CB2 Receptor | |
|---|---|---|
| Location | Central nervous system, brain, spinal cord, other tissues (pituitary, thyroid, liver, intestinal, fat cells) | Mainly immune cells, some neurons, T- and B-lymphocytes, CNS, PNS, GI tract |
| Main Functions | Modulates neurotransmitter release, maintains homeostasis, regulates food intake, involved in obesity | Moderates cytokine release, involved in immune response, studied in neuropathy, neurodegenerative diseases, autoimmune diseases, cancer |
| Psychoactive Effects | Yes (psychoactive effects via THC) | No significant psychoactive effects |
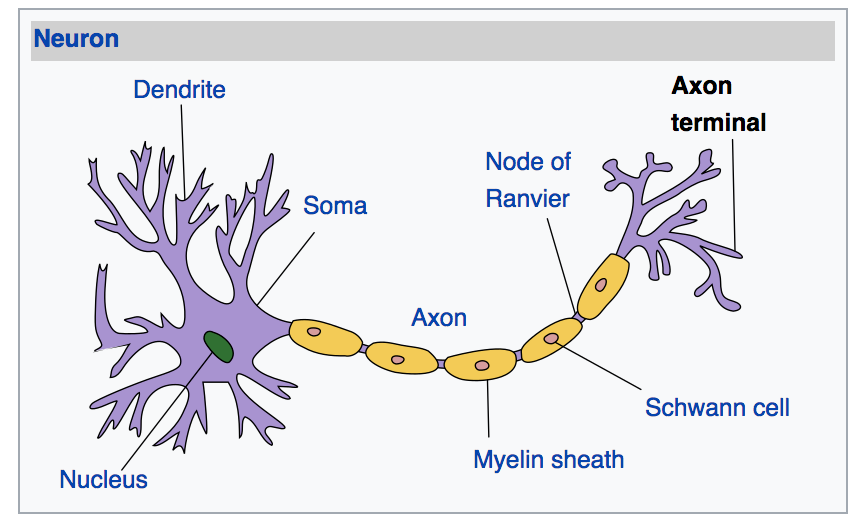
CB1 Receptor:
The CB1 receptor is a G protein-coupled receptor mainly expressed in the central nervous system.
The receptor is found on the axons and terminals of neurons and glial cells in the brain, with high levels in the amygdala, prefrontal cortex, and hippocampus.[ref][ref] CB1 receptors are also found in the spinal cord, as well as in other cells such as pituitary, thyroid, liver, intestinal, and fat cells.
Possibly, one role of the CB1 receptors in neurons could “modulate neurotransmitter release in a manner that maintains homeostasis in health and disease by preventing the development of excessive neuronal activity in the central nervous system”.[ref]
Basically, the release of endocannabinoids, which bind to the CB1 receptors, keeps the excitatory and the inhibitory neurotransmitters in check.[ref]
The CB1 receptor is also involved in processes that regulate food intake and is often studied for its role in obesity.[ref] (Yes, there is a reason why marijuana gives people the munchies…)
CB2 Receptor:
The CB2 receptor is found mainly in immune cells as well as in some neurons. CB2 receptors have been studied in a variety of pathologic conditions, including:
- neuropathy
- neurodegenerative diseases
- osteoporosis
- autoimmune diseases
- atherosclerosis
- cancer[ref]
CB2 receptors are found in the immune system, the central nervous system, the peripheral nervous system, and the gastrointestinal tract.[ref] Activation of the CB2 receptors moderates the release of cytokines from immune cells.
Endocannabinoids:
Our body produces several different endogenous cannabinoids derived from fatty acids and binds to the CB1 and CB2 receptors. The fatty acids used by the body when synthesizing endocannabinoids include arachidonic acid, palmitic acid, and oleic acid.
| Endocannabinoid | Precursor Fatty Acid | Binds to Receptor(s) | Main Effects |
|---|---|---|---|
| Anandamide (AEA) | Arachidonic acid | CB1, CB2 | Reward, pleasure, pain relief, feeding behavior |
| 2-AG | Arachidonic acid | CB1, CB2 | Similar to AEA |
| Palmitoylethanolamide (PEA) | Palmitic acid | CB2 | Anti-inflammatory |
| Oleoylethanolamide (OEA) | Oleic acid | CB1, CB2 (lesser extent) | Appetite regulation |
- Arachidonic acid-derived endocannabinoids include anandamide (AEA), 2-AG, and others.
- The oleic acid-derived EC is oleoylethanolamide (OEA).
Anandamide (AEA) is made in the body from arachidonic acid and is degraded by the fatty acid amide hydrolase (FAAH) enzyme.
Anandamide binds to both the CB1 and CB2 receptors and plays a role in reward, pleasure, and pain relief as well as feeding behavior. It is the ‘bliss’ molecule.
When less of the FAAH enzyme is around, anandamide is degraded more slowly. This leads to more of the bliss molecule.
The palmitic acid-derived endocannabinoid is palmitoylethanolamide (PEA). It acts as a natural anti-inflammatory and is produced on demand by cells in response to injury, inflammation, and pain. PEA primarily works by inhibiting the release of pro-inflammatory molecules from immune cells, like mast cells and macrophages.[ref]
Cannabis: Active components that bind to CB1 and CB2
THC, also called Δ9-tetrahydrocannabinol, activates both CB1 and CB2 receptors. It’s the psychoactive component that makes a person feel high. It has been shown to be a partial agonist to the CB1 receptor. It is thought that it may increase dopamine and acetylcholine in the brain.[ref]
Cannabidiol (CBD oil), on the other hand, is also a component of cannabis. However, it doesn’t bind directly to the CB1 or CB2 receptors, and instead, it influences the way other substances are able to bind to the receptors.[ref]
All of the effects of cannabis, both good and bad, point to or exaggerate the endogenous reasons for the endocannabinoids. For example:
- Endocannabinoids play a role in obesity (cannabis gives you the munchies)
- Anandamide makes you happy and takes away the pain (cannabis can do the same)
- Having fewer CB1 receptors can lead to depression and anxiety (cannabis takes away anxiety.)
| Component | Receptor Interaction | Psychoactive? | Main Effects/Mechanisms |
|---|---|---|---|
| THC | Partial agonist at CB1, CB2 | Yes | Euphoria, altered perception, increased appetite, possible anxiety/paranoia |
| CBD | Indirect (modulates binding) | No | Anti-inflammatory, anxiolytic, may counteract some THC effects |
Side effects of cannabis:
While there seems to be a lot of information available touting the promising medicinal effects of cannabis, such as a reduction in nausea for cancer patients, there are some serious considerations in recreational use. Interestingly, genetic variants can help explain who is more likely to have negative experiences with cannabis.
| Side Effect | Description/Notes |
|---|---|
| Psychotic-like states | Occur in 20-50% of users at least once; pregnenolone may be protective[ref][ref] |
| Schizophrenia risk | Frequent use doubles risk; genetic factors may play a role[ref] |
| Cannabinoid hyperemesis | Cyclical vomiting, nausea, abdominal pain after prolonged use |
| Cardiovascular events | Arrhythmia, stroke, heart attack (CB1 activation)[ref] |
| Acute anxiety disorders | Can be triggered by regular use[ref] |
Metabolism of cannabis: Genetic interactions
Phase I metabolism of THC in the liver involves the CYP2C9, CYP2C19, and/or CYP3A4 enzymes. The metabolites produced by the phase I metabolism of THC are then further altered by UGT1A3 for excretion in the feces and in the urine. [ref]
The primary route of metabolism is through the CYP2C9 gene. Genetic variants that decrease CYP2C9 function decrease the speed at which THC is metabolized and the overall amount of THC in someone’s system.[ref]
Genes involved in cannabis response:
Genetic variants in the cannabis receptors and the genes related to cannabis response can affect your risk of negative effects from cannabis, as well as overall subjective experience with it.
| Gene | Role | Risk/Effect |
|---|---|---|
| CNR1 | CB1 receptor | Obesity, addiction |
| CNR2 | CB2 receptor | Immune response, schizophrenia |
| FAAH | Anandamide breakdown | Bliss molecule duration |
| AKT1 | Psychosis risk | ↑ Risk w/ variant |
| ABCB1 | Drug transport | Cannabis dependency risk |
| TCN2 | B12 transport | Indirect link to adolescent use |
CNR1 gene (CB1 receptor) genetic variants:
Genetic variants in the CNR1 gene affect the body’s response to cannabis, including whether are, not you are likely to become dependent on cannabis. Additionally, the CB1 receptor is important in the way the brain regulates appetite; thus, a variant in CNR1 can affect someone’s susceptibility to obesity.
Genetic variants in CNR1 increase the risk of substance abuse (alcohol, drugs) and cannabis dependency. The variants also increase the risk of obesity. Interestingly, CNR1 is also linked to an increased susceptibility to stress-induced migraines. [ref][ref][ref][ref][ref] [ref][ref]
CNR2 gene (CB2 receptor) genetic variants:
The CNR2 gene encodes the CB2 receptor. The CB2 receptors are prevalent in the immune system.
Variants in CNR2 affect the immune response to viruses, including Covid and RSV.[ref][ref] Variants in CNR2 also affect the risk of schizophrenia, which may also be due to susceptibility to infection or due to altered immune response.[ref]
FAAH (fatty acid amide hydrolase) genetic variants:
FAAH breaks down the endocannabinoid anandamide, thus stopping the action of this ‘bliss molecule’.[ref] Currently, inhibitors of FAAH are being developed and studied for their role in treating pain and inflammation.[ref]
AKT1 gene:
The AKT1 gene encodes a protein kinase that is part of the dopamine receptor signaling cascade in the brain. It is activated by THC and is also involved in promoting neuronal cell survival and glucose metabolism.
Genetic variants in the AKT1 gene are associated with an increased risk of psychosis from cannabis. One study found that two copies of the variant (rs2494732, below in the genotype report), increased the risk of psychosis by sevenfold in regular cannabis smokers. [ref]
ABCB1 (multidrug resistance gene):
The ABCB1 gene involves transporting toxins (including drugs) out of the cell. Variants in ABCB1 affect the response to cannabis as well as to medications.
In a study done on heavy cannabis users, those with the rs1045642 A allele had lower plasma levels of cannabis due to the drug staying inside of cells longer.[ref] Another study showed that those with the G/G genotype were at a significantly greater risk for cannabis dependence.[ref]
TCN2 (B12 transport gene):
While this gene doesn’t have much to do with cannabis, per se, there is an interesting study tying maternal B12 levels during pregnancy to the risk of their children having substance abuse issues as an adolescents (including cannabis use). The study found that an estimated higher B12 (meat consumption) by a mom while pregnant correlated with a lower risk of substance abuse in teenage offspring. This was stratified by mom’s TCN2 genetic variant, rs1801198.[ref] The G allele is linked to lower plasma B12 levels.[ref]
Cannabinoid Receptor Genotype Report
Access this content:
An active subscription is required to access this content.
Lifehacks:
Substance Abuse:
If you or someone you know is struggling with substance use disorders, please seek help! Visit http://drughelpline.org/, talk to your doctor, talk with family or trusted friends. Reach out.
Access this content:
An active subscription is required to access this content.
Related Articles and Genes:
Problems sleeping? Your genes can point to individualized solutions
References: