will see their genotype report below and the solutions in the Lifehacks section.
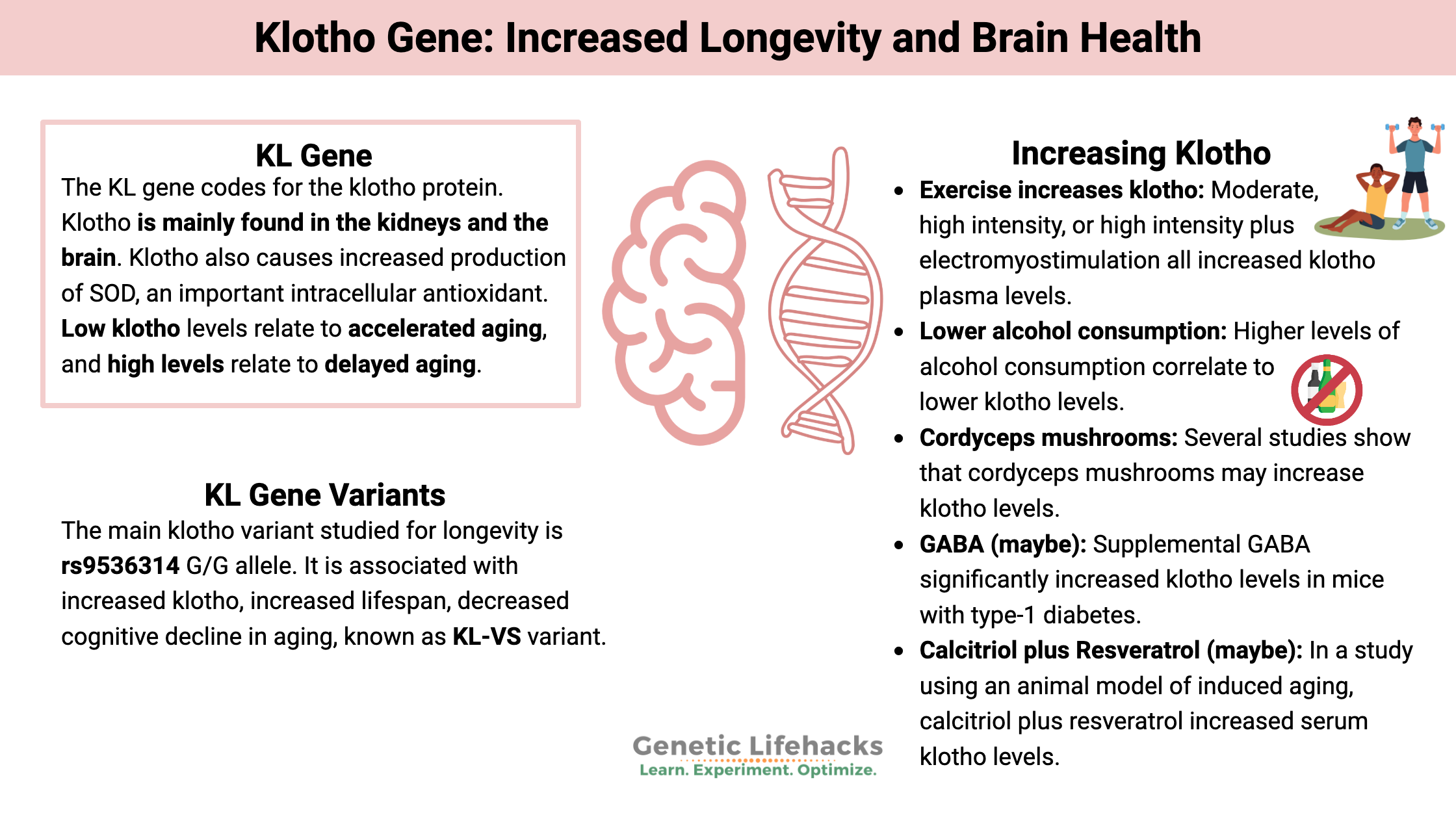
Klotho proteins, which include αKlotho and βKlotho, are produced in the brain, kidneys, and a few other tissues. This article primarily focuses on αKlotho, and for simplicity’s sake, it will be referred to just as Klotho.
The name “Klotho” comes from the Greek Fate who spins the thread of life—an apt metaphor, as this protein is deeply tied to lifespan and aging. Studies show that:[ref]
The klotho protein was originally identified in mice that carried a mutation that caused accelerated aging. These mice were found to have a loss-of-function mutation in the Klotho gene, which caused atherosclerosis, osteoporosis, muscle loss, kidney disease, and sagging skin – starting at 3 to 4 weeks of age. Likewise, creating a mouse strain that overexpresses the klotho gene causes a longer lifespan.[ref][ref]
Klotho, found in the cerebrospinal fluid, plasma, and membranes, is involved in maintaining phosphorus and calcium homeostasis in the kidneys and controlling insulin by reducing insulin-like growth factor-1 (IGF-1). Klotho also causes increased production of SOD (superoxide dismutase), an important intracellular antioxidant.[ref][ref]
Fibroblast growth factors, FGFs, are a family of signaling molecules that control cell growth, tissue repair, development, and more.
Klotho works with FGF23, which is produced by the bone marrow and plays an important role in how the kidneys regulate phosphate and calcium levels, as well as vitamin D metabolism. When active vitamin D (1,25(OH)2) and phosphate levels are high, FGF23 is produced in the bones and binds to its receptor with Klotho in the kidneys, promoting phosphate excretion and inhibiting vitamin D production.[ref][ref][ref]
Just as a little context, phosphate and calcium levels are critical for the way that muscles and nerves function. We can’t live without them at the right levels. The levels are tightly controlled in the body, with both being stored in the bones as a reservoir. Magnesium also comes into play here in the way that the levels of calcium and phosphorus are regulated. Essentially, we get calcium, phosphorus, and magnesium from food, and calcium is then stored in the bones until needed. The levels of calcium and phosphorus in the body are regulated by parathyroid hormone, vitamin D, calcitonin, and FGF23. Phosphorus is used for ATP, phospholipids, and DNA synthesis, while calcium is integral to the way that ion channels work, such as in neurons and the heart muscle.[ref] Basically, when the diet is high in phosphate, klotho causes lower active vitamin D (1,25 OH-D).[ref]
In humans, klotho levels have been shown to predict mortality. A six-year-long study of 804 adults aged 65 or older found that those with lower klotho levels had a 78% greater mortality risk. Lower klotho levels were defined as being in the bottom 25%.[ref]
Let’s put that into context. Everyone knows that uncontrolled high blood pressure will kill you, right? An extensive study that followed participants for almost 20 years found that people with uncontrolled high blood pressure had an increased all-cause mortality risk of 69%.[ref] Compare this to the low klotho level correlating to a 78% increase in mortality…
In studies, researchers look at cognitive impairment, cardiovascular disease, kidney stones, cancer, and longevity to determine the effects of klotho levels. Genetic variants linked to higher klotho levels also increase longevity.[ref]
The studies on how klotho levels influence cognitive decline or dementia have conflicting results.
The difference may be the presence of the APOE E4 allele (risk factor for Alzheimer’s). A fascinating study found that older adults who carry the APOE E4 allele had lower β-amyloid if they also carried the KL variant that increases klotho.[ref]
A study looked at older adults (Caucasian population) with the genetic variant associated with increased klotho compared to a similar group without the klotho variant. They found that the KL variant carriers had increased brain volume in a prefrontal cortex region. The variant carriers (more klotho) also had better executive function, which included better working memory and processing speed.[ref]
Another study also showed that older adults (age 52-85) who carry the KL variant associated with higher Klotho levels also had a significantly better cognitive function. Importantly, this increase in Klotho was stable as people aged. For example, people in their 50s with higher klotho had better cognitive function scores when matched with people their same age with typical klotho levels. Likewise, people in their 70s with higher klotho had better cognitive scores than their age-matched peers.[ref]
Animal studies are better able to define the function of this gene.
In diabetic mice, a pre-treatment of klotho injections showed protection from diabetic cardiomyopathy. It reduced the oxidative stress triggered by high blood glucose levels. [ref]
Adema, Aaltje Y., et al. “Influence of Exogenous Growth Hormone Administration on Circulating Concentrations of α-Klotho in Healthy and Chronic Kidney Disease Subjects: A Prospective, Single-Center Open Case-Control Pilot Study.” BMC Nephrology, vol. 19, no. 1, Nov. 2018, p. 327. PubMed, https://doi.org/10.1186/s12882-018-1114-z.
Agoro, Rafiou, and Kenneth E. White. “Regulation of FGF23 Production and Phosphate Metabolism by Bone–Kidney Interactions.” Nature Reviews Nephrology, vol. 19, no. 3, Mar. 2023, pp. 185–93. www.nature.com, https://doi.org/10.1038/s41581-022-00665-x.
Almeida, Osvaldo P., et al. “Longevity Klotho Gene Polymorphism and the Risk of Dementia in Older Men.” Maturitas, vol. 101, July 2017, pp. 1–5. PubMed, https://doi.org/10.1016/j.maturitas.2017.04.005.
Amaro-Gahete, F. J., et al. “Exercise Training Increases the S-Klotho Plasma Levels in Sedentary Middle-Aged Adults: A Randomised Controlled Trial. The FIT-AGEING Study.” Journal of Sports Sciences, vol. 37, no. 19, Oct. 2019, pp. 2175–83. PubMed, https://doi.org/10.1080/02640414.2019.1626048.
Amin, Hasnat A., et al. “No Evidence That Genetic Variation at the Klotho Locus Is Associated With Longevity in Caucasians from the Newcastle 85+ Study and the UK Biobank.” The Journals of Gerontology Series A: Biological Sciences and Medical Sciences, vol. 77, no. 3, Dec. 2021, pp. 457–61. PubMed Central, https://doi.org/10.1093/gerona/glab361.
Chen, Ci-Di, et al. “The Antiaging Protein Klotho Enhances Oligodendrocyte Maturation and Myelination of the CNS.” The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, vol. 33, no. 5, Jan. 2013, pp. 1927–39. PubMed, https://doi.org/10.1523/JNEUROSCI.2080-12.2013.
Dehghani, Ali, et al. “Resveratrol and 1,25-Dihydroxyvitamin D Co-Administration Protects the Heart against D-Galactose-Induced Aging in Rats: Evaluation of Serum and Cardiac Levels of Klotho.” Aging Clinical and Experimental Research, vol. 31, no. 9, Sept. 2019, pp. 1195–205. PubMed, https://doi.org/10.1007/s40520-018-1075-x.
Dërmaku-Sopjani, Miribane, et al. “Significance of the Anti-Aging Protein Klotho.” Molecular Membrane Biology, vol. 30, no. 8, Dec. 2013, pp. 369–85. DOI.org (Crossref), https://doi.org/10.3109/09687688.2013.837518.
Dubal, Dena B., et al. “Life Extension Factor Klotho Enhances Cognition.” Cell Reports, vol. 7, no. 4, May 2014, pp. 1065–76. PubMed Central, https://doi.org/10.1016/j.celrep.2014.03.076.
Erickson, Claire M., et al. “KLOTHO Heterozygosity Attenuates APOE4-Related Amyloid Burden in Preclinical AD.” Neurology, vol. 92, no. 16, Apr. 2019, pp. e1878–89. PubMed, https://doi.org/10.1212/WNL.0000000000007323.
Freathy, Rachel M., et al. “The Functional ‘KL-VS’ Variant of KLOTHO Is Not Associated with Type 2 Diabetes in 5028 UK Caucasians.” BMC Medical Genetics, vol. 7, June 2006, p. 51. PubMed, https://doi.org/10.1186/1471-2350-7-51.
Guo, Yue, et al. “Klotho Protects the Heart from Hyperglycemia-Induced Injury by Inactivating ROS and NF-κB-Mediated Inflammation Both in Vitro and in Vivo.” Biochimica et Biophysica Acta (BBA) – Molecular Basis of Disease, vol. 1864, no. 1, Jan. 2018, pp. 238–51. ScienceDirect, https://doi.org/10.1016/j.bbadis.2017.09.029.
Hajare, Aditya Dipakrao, et al. “Klotho Antiaging Protein: Molecular Mechanisms and Therapeutic Potential in Diseases.” Molecular Biomedicine, vol. 6, no. 1, Mar. 2025, p. 19. Springer Link, https://doi.org/10.1186/s43556-025-00253-y.
Hao, Qiukui, et al. “G-395A Polymorphism in the Promoter Region of the KLOTHO Gene Associates with Reduced Cognitive Impairment among the Oldest Old.” Age (Dordrecht, Netherlands), vol. 38, no. 1, Feb. 2016, p. 7. PubMed, https://doi.org/10.1007/s11357-015-9869-7.
Invidia, Laura, et al. “The Frequency of Klotho KL-VS Polymorphism in a Large Italian Population, from Young Subjects to Centenarians, Suggests the Presence of Specific Time Windows for Its Effect.” Biogerontology, vol. 11, no. 1, Feb. 2010, pp. 67–73. PubMed, https://doi.org/10.1007/s10522-009-9229-z.
Janić, Miodrag, et al. “Expression of Longevity Genes Induced by a Low-Dose Fluvastatin and Valsartan Combination with the Potential to Prevent/Treat ‘Aging-Related Disorders.’” International Journal of Molecular Sciences, vol. 20, no. 8, Apr. 2019, p. 1844. PubMed, https://doi.org/10.3390/ijms20081844.
Jurado-Fasoli, Lucas, et al. “Alcohol Consumption and S-Klotho Plasma Levels in Sedentary Healthy Middle-Aged Adults: A Cross Sectional Study.” Drug and Alcohol Dependence, vol. 194, Jan. 2019, pp. 107–11. PubMed, https://doi.org/10.1016/j.drugalcdep.2018.09.024.
Kim, Sang Soo, et al. “Decreased Plasma α-Klotho Predict Progression of Nephropathy with Type 2 Diabetic Patients.” Journal of Diabetes and Its Complications, vol. 30, no. 5, July 2016, pp. 887–92. ScienceDirect, https://doi.org/10.1016/j.jdiacomp.2016.03.006.
Kurosu, Hiroshi, et al. “Suppression of Aging in Mice by the Hormone Klotho.” Science (New York, N.Y.), vol. 309, no. 5742, Sept. 2005, pp. 1829–33. PubMed Central, https://doi.org/10.1126/science.1112766.
Liu, Bu-Hui, et al. “[Molecular mechanisms of mycelium of Cordyceps sinensis ameliorating renal tubular epithelial cells aging induced by D-galactose via inhibiting autophagy-related AMPK/ULK1 signaling activation].” Zhongguo Zhong Yao Za Zhi = Zhongguo Zhongyao Zazhi = China Journal of Chinese Materia Medica, vol. 44, no. 6, Mar. 2019, pp. 1258–65. PubMed, https://doi.org/10.19540/j.cnki.cjcmm.20181205.001.
Mendoza-Carrera, Francisco, et al. “Association of KLOTHO Gene Variants with Metabolic and Renal Function Parameters in Mexican Patients Living with Type 2 Diabetes.” Journal of Diabetes and Metabolic Disorders, vol. 23, no. 1, June 2024, pp. 1125–31. PubMed, https://doi.org/10.1007/s40200-024-01398-3.
Mengel-From, Jonas, et al. “Genetic Variants in KLOTHO Associate With Cognitive Function in the Oldest Old Group.” The Journals of Gerontology Series A: Biological Sciences and Medical Sciences, vol. 71, no. 9, Sept. 2016, pp. 1151–59. PubMed Central, https://doi.org/10.1093/gerona/glv163.
———. “Genetic Variants in KLOTHO Associate With Cognitive Function in the Oldest Old Group.” The Journals of Gerontology Series A: Biological Sciences and Medical Sciences, vol. 71, no. 9, Sept. 2016, pp. 1151–59. PubMed Central, https://doi.org/10.1093/gerona/glv163.
Nagai, Taku, et al. “Cognition Impairment in the Genetic Model of Aging Klotho Gene Mutant Mice: A Role of Oxidative Stress.” FASEB Journal: Official Publication of the Federation of American Societies for Experimental Biology, vol. 17, no. 1, Jan. 2003, pp. 50–52. PubMed, https://doi.org/10.1096/fj.02-0448fje.
Nie, Fang, et al. “Serum Klotho Protein Levels and Their Correlations with the Progression of Type 2 Diabetes Mellitus.” Journal of Diabetes and Its Complications, vol. 31, no. 3, Mar. 2017, pp. 594–98. ScienceDirect, https://doi.org/10.1016/j.jdiacomp.2016.11.008.
Oguro, Ryosuke, et al. “Association of Carotid Atherosclerosis with Genetic Polymorphisms of the Klotho Gene in Patients with Hypertension.” Geriatrics & Gerontology International, vol. 10, no. 4, Oct. 2010, pp. 311–18. PubMed, https://doi.org/10.1111/j.1447-0594.2010.00612.x.
Olejnik, Agnieszka, et al. “The Biological Role of Klotho Protein in the Development of Cardiovascular Diseases.” BioMed Research International, vol. 2018, Dec. 2018, p. 5171945. PubMed Central, https://doi.org/10.1155/2018/5171945.
Rafikova, Ekaterina, et al. “Open Genes—a New Comprehensive Database of Human Genes Associated with Aging and Longevity.” Nucleic Acids Research, vol. 52, no. D1, Sept. 2023, pp. D950–62. PubMed Central, https://doi.org/10.1093/nar/gkad712.
Ramez, Maral, et al. “The Greater Effect of High-Intensity Interval Training versus Moderate-Intensity Continuous Training on Cardioprotection against Ischemia-Reperfusion Injury through Klotho Levels and Attenuate of Myocardial TRPC6 Expression.” BMC Cardiovascular Disorders, vol. 19, no. 1, May 2019, p. 118. PubMed, https://doi.org/10.1186/s12872-019-1090-7.
Semba, Richard D., et al. “Plasma Klotho and Mortality Risk in Older Community-Dwelling Adults.” The Journals of Gerontology Series A: Biological Sciences and Medical Sciences, vol. 66A, no. 7, July 2011, pp. 794–800. PubMed Central, https://doi.org/10.1093/gerona/glr058.
Shaker, Joseph L., and Leonard Deftos. “Calcium and Phosphate Homeostasis.” Endotext, edited by Kenneth R. Feingold et al., MDText.com, Inc., 2000. PubMed, http://www.ncbi.nlm.nih.gov/books/NBK279023/.
Shin, Eun-Joo, et al. “Melatonin Attenuates Memory Impairment Induced by Klotho Gene Deficiency Via Interactive Signaling Between MT2 Receptor, ERK, and Nrf2-Related Antioxidant Potential.” International Journal of Neuropsychopharmacology, vol. 18, no. 6, Jan. 2015, p. pyu105. PubMed Central, https://doi.org/10.1093/ijnp/pyu105.
Son, Dong Ok, et al. “Combined Effect of GABA and Glucagon-like Peptide-1 Receptor Agonist on Cytokine-Induced Apoptosis in Pancreatic β-Cell Line and Isolated Human Islets.” Journal of Diabetes, vol. 11, no. 7, July 2019, pp. 563–72. PubMed, https://doi.org/10.1111/1753-0407.12881.
Tang, Rong, et al. “[Effect of cordyceps sinensis extract on Klotho expression and apoptosis in renal tubular epithelial cells induced by angiotensin II].” Zhong Nan Da Xue Xue Bao. Yi Xue Ban = Journal of Central South University. Medical Sciences, vol. 34, no. 4, Apr. 2009, pp. 300–07.
Xu, Chen, et al. “Klotho Gene Polymorphism of Rs3752472 Is Associated with the Risk of Urinary Calculi in the Population of Han Nationality in Eastern China.” Gene, vol. 526, no. 2, Sept. 2013, pp. 494–97. PubMed, https://doi.org/10.1016/j.gene.2013.06.001.
Yokoyama, Jennifer S., et al. “Variation in Longevity Gene KLOTHO Is Associated with Greater Cortical Volumes.” Annals of Clinical and Translational Neurology, vol. 2, no. 3, Mar. 2015, pp. 215–30. DOI.org (Crossref), https://doi.org/10.1002/acn3.161.
Yokoyama, Serina, et al. “A Klotho Gene Single Nucleotide Polymorphism Is Associated with the Onset of Stroke and Plasma Klotho Concentration.” Aging, vol. 11, no. 1, Dec. 2018, pp. 104–14. PubMed, https://doi.org/10.18632/aging.101728.
Yoshikawa, Ryouhei, et al. “The Age-Related Changes of Dietary Phosphate Responsiveness in Plasma 1,25-Dihydroxyvitamin D Levels and Renal Cyp27b1 and Cyp24a1 Gene Expression Is Associated with Renal α-Klotho Gene Expression in Mice.” Journal of Clinical Biochemistry and Nutrition, vol. 62, no. 1, Jan. 2018, pp. 68–74. PubMed Central, https://doi.org/10.3164/jcbn.17-20.
Zhou, Donghao, et al. “Uncontrolled Hypertension Increases Risk of All-Cause and Cardiovascular Disease Mortality in US Adults: The NHANES III Linked Mortality Study.” Scientific Reports, vol. 8, no. 1, June 2018, p. 9418. www.nature.com, https://doi.org/10.1038/s41598-018-27377-2.
Zhu, Zewu, et al. “Klotho Gene Polymorphisms Are Associated with Healthy Aging and Longevity: Evidence from a Meta-Analysis.” Mechanisms of Ageing and Development, vol. 178, Mar. 2019, pp. 33–40. PubMed, https://doi.org/10.1016/j.mad.2018.12.003.