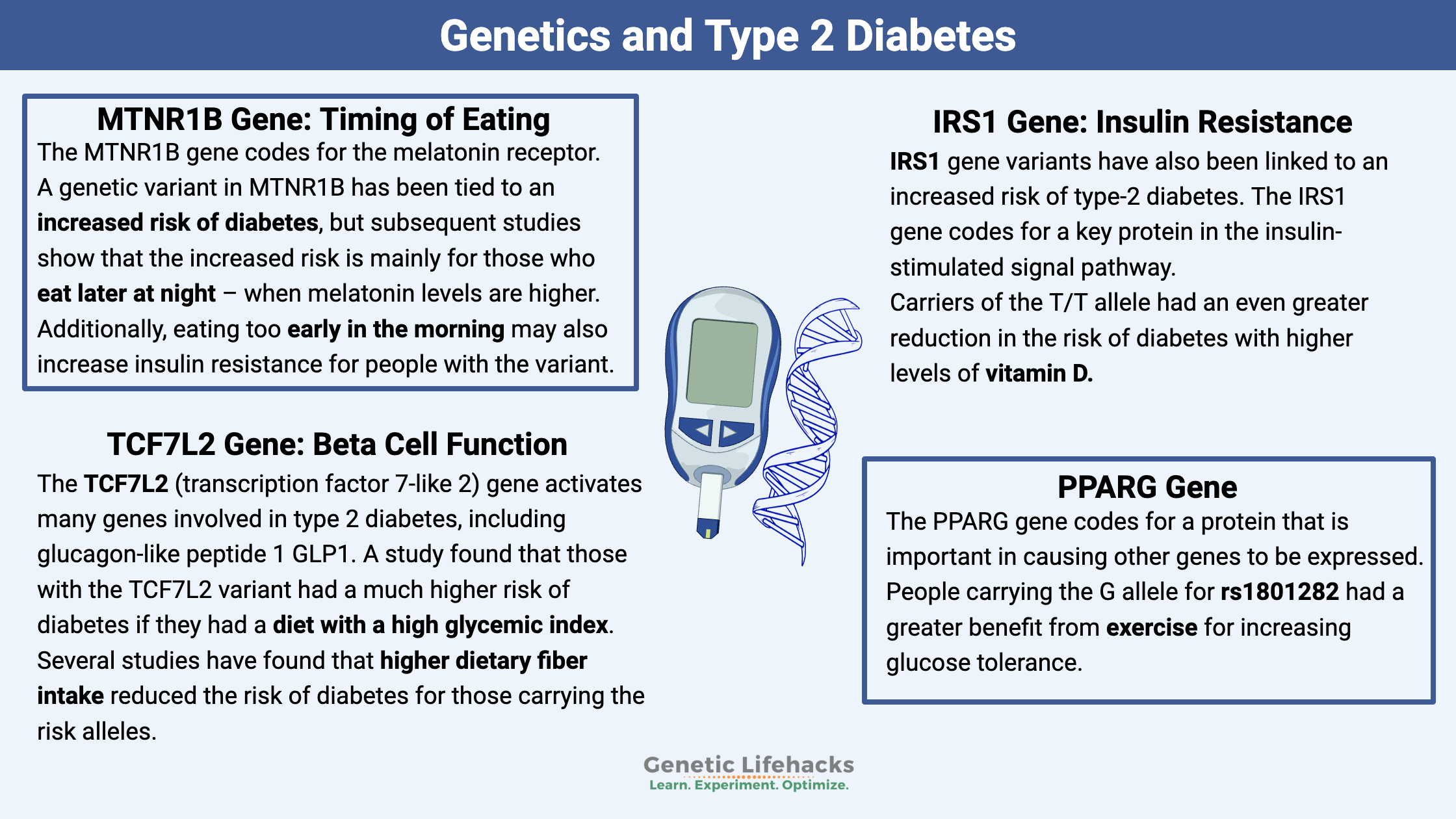
Key takeaways:
~ For most people, type 2 diabetes is due to a combination of lifestyle and genetic risk factors.
~ Knowing your root cause can help you prioritize lifestyle changes that may work to prevent, manage, or even reverse type 2 diabetes.
~ Targeting the right pathway may help you to prevent or reverse pre-diabetes or diabetes.
Genetic variants increase susceptibility to diabetes:
While diet is important, genetics plays a big role in diabetes. Studies on twins show that the genetic component of type 2 diabetes is estimated to be between 46-70%.[ref] Thus, for type 2 diabetes, genetic susceptibility interacts with what you eat, when you eat, and vitamin or mineral deficiencies.
Diabetes is a term applied when your blood sugar levels are higher than normal, or you inappropriately respond to foods. Your body regulates blood glucose levels through the release of insulin.
With diabetes, your body can be resistant to the effects of insulin — or — you may not produce enough insulin.
How can you use this information?
Just as there are multiple ways to have high blood glucose levels, there are multiple genetic variants that cause increased susceptibility to diabetes.
By knowing which genetic variants you carry, you can know which pathways are likely to be causing your high blood glucose levels. This can help you personalize your approach to either managing or reversing your type 2 diabetes.
If you don’t have diabetes, understanding your genetic susceptibility can help you target the right pathways for preventing elevated blood glucose levels. If you know where your genetic weakness lies, you can target that pathway to get the most benefit.
Diabetes Genotype Report:
This genotype report shows you your genotype for the variants, along with the specific interventions that research shows are beneficial.
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
Intermittent Fasting: Benefits from Changing Gene Expression
References:
Baig, Sonia, et al. “Heredity of Type 2 Diabetes Confers Increased Susceptibility to Oxidative Stress and Inflammation.” BMJ Open Diabetes Research & Care, vol. 8, no. 1, Jan. 2020, p. e000945. PubMed Central, https://doi.org/10.1136/bmjdrc-2019-000945.
Caro-Gomez, María Antonieta, et al. “Association of Native American Ancestry and Common Variants in ACE, ADIPOR2, MTNR1B, GCK, TCF7L2 and FTO Genes with Glycemic Traits in Colombian Population.” Gene, vol. 677, Nov. 2018, pp. 198–210. PubMed, https://doi.org/10.1016/j.gene.2018.07.066.
Cauchi, Stéphane, et al. “Transcription Factor TCF7L2 Genetic Study in the French Population: Expression in Human Beta-Cells and Adipose Tissue and Strong Association with Type 2 Diabetes.” Diabetes, vol. 55, no. 10, Oct. 2006, pp. 2903–08. PubMed, https://doi.org/10.2337/db06-0474.
Ding, Weiyue, et al. “Meta-Analysis of Association between TCF7L2 Polymorphism Rs7903146 and Type 2 Diabetes Mellitus.” BMC Medical Genetics, vol. 19, Mar. 2018, p. 38. PubMed Central, https://doi.org/10.1186/s12881-018-0553-5.
Eriksen, Rebeca, et al. “Gene‐diet Quality Interactions on Haemoglobin A1c and Type 2 Diabetes Risk: The Airwave Health Monitoring Study.” Endocrinology, Diabetes & Metabolism, vol. 2, no. 4, July 2019, p. e00074. PubMed Central, https://doi.org/10.1002/edm2.74.
Florez, Jose C., et al. “TCF7L2 Polymorphisms and Progression to Diabetes in the Diabetes Prevention Program.” The New England Journal of Medicine, vol. 355, no. 3, July 2006, pp. 241–50. PubMed, https://doi.org/10.1056/NEJMoa062418.
Gautier, Alain, et al. “Effects of Genetic Susceptibility for Type 2 Diabetes on the Evolution of Glucose Homeostasis Traits Before and After Diabetes Diagnosis.” Diabetes, vol. 60, no. 10, Oct. 2011, pp. 2654–63. PubMed Central, https://doi.org/10.2337/db10-1442.
“Glycemic Index Chart: GI Ratings for Hundreds of Foods.” University Health News, 22 June 2020, https://universityhealthnews.com/daily/nutrition/glycemic-index-chart/.
Hindy, G., et al. “Role of TCF7L2 Risk Variant and Dietary Fibre Intake on Incident Type 2 Diabetes.” Diabetologia, vol. 55, no. 10, 2012, pp. 2646–54. PubMed Central, https://doi.org/10.1007/s00125-012-2634-x.
Hosseini-Esfahani, Firoozeh, et al. “Some Dietary Factors Can Modulate the Effect of the Zinc Transporters 8 Polymorphism on the Risk of Metabolic Syndrome.” Scientific Reports, vol. 7, no. 1, May 2017, p. 1649. PubMed, https://doi.org/10.1038/s41598-017-01762-9.
Kommoju, Uma Jyothi, et al. “No Detectable Association of IGF2BP2 and SLC30A8 Genes with Type 2 Diabetes in the Population of Hyderabad, India.” Meta Gene, vol. 1, Dec. 2013, pp. 15–23. PubMed, https://doi.org/10.1016/j.mgene.2013.09.003.
Lane, Jacqueline M., et al. “Impact of Common Diabetes Risk Variant in MTNR1B on Sleep, Circadian, and Melatonin Physiology.” Diabetes, vol. 65, no. 6, June 2016, pp. 1741–51. PubMed Central, https://doi.org/10.2337/db15-0999.
Langenberg, C., et al. “Common Genetic Variation in the Melatonin Receptor 1B Gene (MTNR1B) Is Associated with Decreased Early-Phase Insulin Response.” Diabetologia, vol. 52, no. 8, Aug. 2009, pp. 1537–42. PubMed, https://doi.org/10.1007/s00125-009-1392-x.
Li, Qiuyan, et al. “Associations between Two Single-Nucleotide Polymorphisms (Rs1801278 and Rs2943641) of Insulin Receptor Substrate 1 Gene and Type 2 Diabetes Susceptibility: A Meta-Analysis.” Endocrine, vol. 51, no. 1, Jan. 2016, pp. 52–62. Springer Link, https://doi.org/10.1007/s12020-015-0770-z.
Liu, Chen, et al. “MTNR1B Rs10830963 Is Associated with Fasting Plasma Glucose, HbA1C and Impaired Beta-Cell Function in Chinese Hans from Shanghai.” BMC Medical Genetics, vol. 11, Apr. 2010, p. 59. PubMed Central, https://doi.org/10.1186/1471-2350-11-59.
Lopez-Minguez, Jesus, et al. “Late Dinner Impairs Glucose Tolerance in MTNR1B Risk Allele Carriers: A Randomized, Cross-over Study.” Clinical Nutrition, vol. 37, no. 4, Aug. 2018, pp. 1133–40. ScienceDirect, https://doi.org/10.1016/j.clnu.2017.04.003.
Lyssenko, Valeriya, et al. “Common Variant in MTNR1B Associated with Increased Risk of Type 2 Diabetes and Impaired Early Insulin Secretion.” Nature Genetics, vol. 41, no. 1, Jan. 2009, pp. 82–88. PubMed, https://doi.org/10.1038/ng.288.
Mahmutovic, Lejla, et al. “Association of IRS1 Genetic Variants with Glucose Control and Insulin Resistance in Type 2 Diabetic Patients from Bosnia and Herzegovina.” Drug Metabolism and Personalized Therapy, vol. 34, no. 1, Mar. 2019. PubMed, https://doi.org/10.1515/dmpt-2018-0031.
Marín, Carmen, et al. “The Insulin Sensitivity Response Is Determined by the Interaction between the G972R Polymorphism of the Insulin Receptor Substrate 1 Gene and Dietary Fat.” Molecular Nutrition & Food Research, vol. 55, no. 2, Feb. 2011, pp. 328–35. PubMed, https://doi.org/10.1002/mnfr.201000235.
Maruthur, Nisa M., and Braxton D. Mitchell. “Zinc–Rs13266634 and the Arrival of Diabetes Pharmacogenetics: The ‘Zinc Mystique.’” Diabetes, vol. 63, no. 5, May 2014, pp. 1463–64. PubMed Central, https://doi.org/10.2337/db14-0151.
Patel, Chirag J., et al. “Systematic Identification of Interaction Effects between Genome- and Environment-Wide Associations in Type 2 Diabetes Mellitus.” Human Genetics, vol. 132, no. 5, 2013, pp. 495–508. PubMed Central, https://doi.org/10.1007/s00439-012-1258-z.
Pearson, Ewan R., et al. “Variation in TCF7L2 Influences Therapeutic Response to Sulfonylureas: A GoDARTs Study.” Diabetes, vol. 56, no. 8, Aug. 2007, pp. 2178–82. PubMed, https://doi.org/10.2337/db07-0440.
Peschke, Elmar, et al. “Melatonin and Pancreatic Islets: Interrelationships between Melatonin, Insulin and Glucagon.” International Journal of Molecular Sciences, vol. 14, no. 4, Mar. 2013, pp. 6981–7015. PubMed Central, https://doi.org/10.3390/ijms14046981.
Qi, Qibin, et al. “Insulin Receptor Substrate 1 (IRS1) Gene Variation Modifies Insulin Resistance Response to Weight-Loss Diets in a Two-Year Randomized Trial.” Circulation, vol. 124, no. 5, Aug. 2011, pp. 563–71. PubMed Central, https://doi.org/10.1161/CIRCULATIONAHA.111.025767.
Rosta, Klara, et al. “Association Study with 77 SNPs Confirms the Robust Role for the Rs10830963/G of MTNR1B Variant and Identifies Two Novel Associations in Gestational Diabetes Mellitus Development.” PLoS ONE, vol. 12, no. 1, Jan. 2017, p. e0169781. PubMed Central, https://doi.org/10.1371/journal.pone.0169781.
Rung, Johan, et al. “Genetic Variant near IRS1 Is Associated with Type 2 Diabetes, Insulin Resistance and Hyperinsulinemia.” Nature Genetics, vol. 41, no. 10, Oct. 2009, pp. 1110–15. PubMed, https://doi.org/10.1038/ng.443.
Schäfer, S. A., et al. “A Common Genetic Variant in WFS1 Determines Impaired Glucagon-like Peptide-1-Induced Insulin Secretion.” Diabetologia, vol. 52, no. 6, June 2009, pp. 1075–82. PubMed, https://doi.org/10.1007/s00125-009-1344-5.
Stolerman, E. S., et al. “TCF7L2 Variants Are Associated with Increased Proinsulin/Insulin Ratios but Not Obesity Traits in the Framingham Heart Study.” Diabetologia, vol. 52, no. 4, Apr. 2009, pp. 614–20. PubMed, https://doi.org/10.1007/s00125-009-1266-2.
Tabara, Yasuharu, et al. “Replication Study of Candidate Genes Associated with Type 2 Diabetes Based on Genome-Wide Screening.” Diabetes, vol. 58, no. 2, Feb. 2009, pp. 493–98. PubMed, https://doi.org/10.2337/db07-1785.
Thorsby, P. M., et al. “Comparison of Genetic Risk in Three Candidate Genes (TCF7L2, PPARG, KCNJ11) with Traditional Risk Factors for Type 2 Diabetes in a Population-Based Study–the HUNT Study.” Scandinavian Journal of Clinical and Laboratory Investigation, vol. 69, no. 2, 2009, pp. 282–87. PubMed, https://doi.org/10.1080/00365510802538188.
Tian, Lili, et al. “Curcumin Represses Mouse 3T3-L1 Cell Adipogenic Differentiation via Inhibiting MiR-17-5p and Stimulating the Wnt Signalling Pathway Effector Tcf7l2.” Cell Death & Disease, vol. 8, no. 1, Jan. 2017, p. e2559. PubMed Central, https://doi.org/10.1038/cddis.2016.455.
Tong, Yu, et al. “Association between TCF7L2 Gene Polymorphisms and Susceptibility to Type 2 Diabetes Mellitus: A Large Human Genome Epidemiology (HuGE) Review and Meta-Analysis.” BMC Medical Genetics, vol. 10, Feb. 2009, p. 15. PubMed Central, https://doi.org/10.1186/1471-2350-10-15.
Trasino, Steven E., and Lorraine J. Gudas. “Vitamin A: A Missing Link in Diabetes?” Diabetes Management (London, England), vol. 5, no. 5, 2015, pp. 359–67. PubMed Central, https://doi.org/10.2217/dmt.15.30.
van der Kroef, Sabrina, et al. “Association between the Rs7903146 Polymorphism in the TCF7L2 Gene and Parameters Derived with Continuous Glucose Monitoring in Individuals without Diabetes.” PLoS ONE, vol. 11, no. 2, Feb. 2016, p. e0149992. PubMed Central, https://doi.org/10.1371/journal.pone.0149992.
Xu, Kuanfeng, et al. “Association between Rs13266634 C/T Polymorphisms of Solute Carrier Family 30 Member 8 (SLC30A8) and Type 2 Diabetes, Impaired Glucose Tolerance, Type 1 Diabetes–a Meta-Analysis.” Diabetes Research and Clinical Practice, vol. 91, no. 2, Feb. 2011, pp. 195–202. PubMed, https://doi.org/10.1016/j.diabres.2010.11.012.
Zheng, Ju-Sheng, et al. “Circulating 25-Hydroxyvitamin D, IRS1 Variant Rs2943641, and Insulin Resistance: Replication of a Gene-Nutrient Interaction in 4 Populations of Different Ancestries.” Clinical Chemistry, vol. 60, no. 1, Jan. 2014, pp. 186–96. PubMed, https://doi.org/10.1373/clinchem.2013.215251.
“Zinc.” Linus Pauling Institute, 23 Apr. 2014, https://lpi.oregonstate.edu/mic/minerals/zinc.
https://diabetes.diabetesjournals.org/content/early/2018/01/08/db17-0318. Accessed 6 Apr. 2022.
Debbie Moon is a biologist, engineer, author, and the founder of Genetic Lifehacks where she has helped thousands of members understand how to apply genetics to their diet, lifestyle, and health decisions. With more than 10 years of experience translating complex genetic research into practical health strategies, Debbie holds a BS in engineering from Colorado School of Mines and an MSc in biological sciences from Clemson University. She combines an engineering mindset with a biological systems approach to explain how genetic differences impact your optimal health.