Key takeaways:
~ Osteoporosis is a degenerative bone disease that increases the risk of fractures.
~ Genetics plays a key role in osteoporosis with the hereditary component estimated to be 50-80%
~ Understanding your genetic susceptibility can lead you to targeted, personalized solutions for osteoporosis.
Osteoporosis, Bone Degeneration, and Aging:
Osteoporosis is a degenerative bone disease facing many of us as we age. Affecting about 54 million people in the US currently, this is a disease that is estimated to affect 50% of women and 25% of men in their lifetime.
Your bones are constantly breaking down and reforming. When the bone cells dissolve the bone matrix, it is called resorption, and the new bone deposits are called formation.
Bone is a dynamic structure, and bone remodeling occurs throughout your life. It is a tightly controlled process where bone is resorbed and then reformed in a balanced way.[ref] Osteoporosis is caused by a chronic excess of bone resorption relative to the formation of new bone. This leads to bone loss and a deterioration of the architecture of the bone.
Here are the types of cells involved in bone formation and breakdown:
- Osteoclasts are cells in the bone that control the breakdown or resorption of bone.
- Osteoblasts are the cells that control bone building or formation.
- Osteocytes are cells encased by mineralized bone that also regulate bone formation.
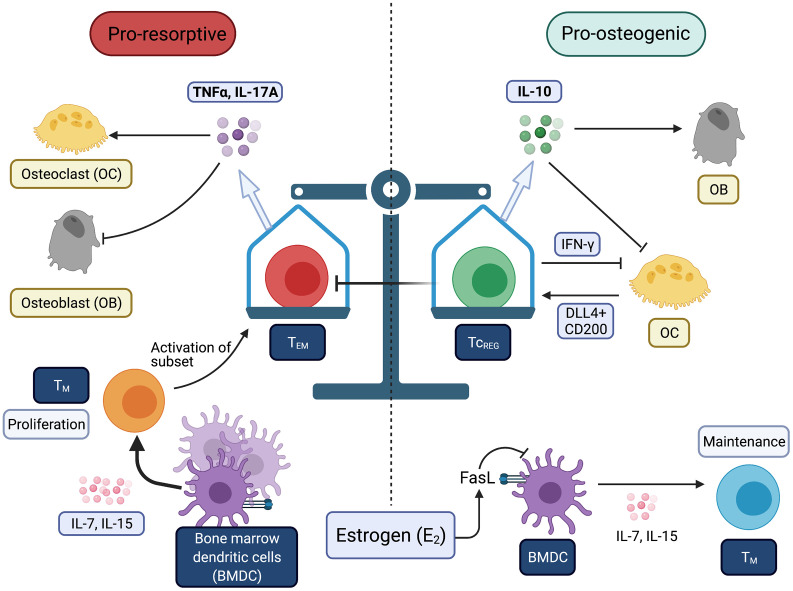
Both men and women lose bone mass in aging, but the loss is often worse for post-menopausal women due to the decline in estrogen signaling. The loss of bone mass increases the risk of fractures. The risk of fractures – such as breaking a hip – is a real worry with osteoporosis.
Genetics and Osteoporosis:
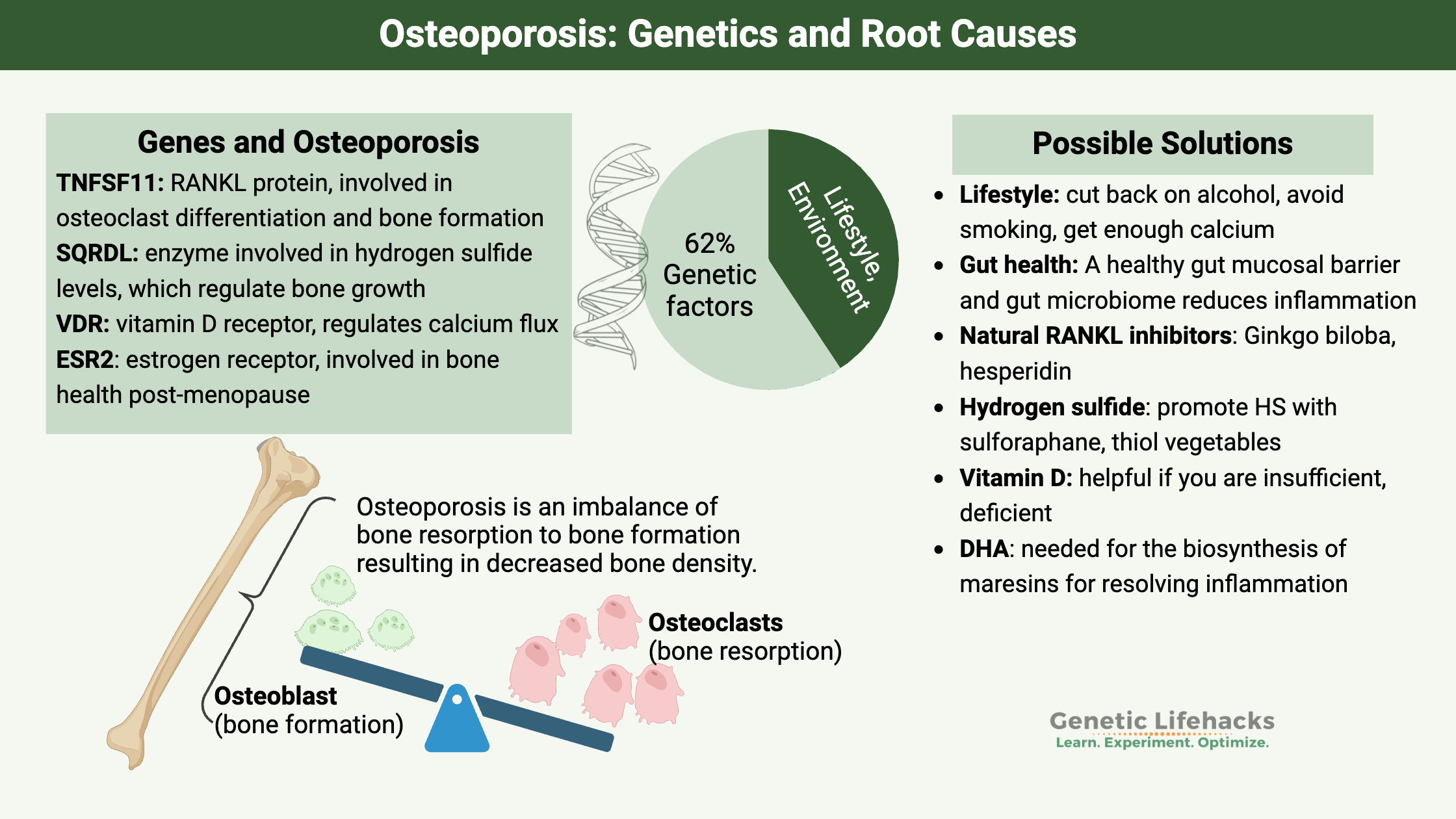
Osteoporosis is caused by a decrease in bone mineral density, and twin studies have shown that the hereditary aspect of bone mineral density (BMD) is between 50 and 80%.[ref] Specifically for osteoporosis, it is estimated that the causes of osteoporosis are about 62% genetic and 38% environmental.[ref] Understanding this genetic risk can help you with targeting the right pathway to prevent osteoporosis.
Within the system of resorption and re-formation of bones lies a balance of the immune response. When you have excess inflammation, the balance is tipped towards having more of the bone resorbed (broken down) by the osteoclasts.
IL-17A (interleukin 17A) and TNF-alpha are proinflammatory cytokines that act to increase bone destruction (pro-resorptive). Chronic, low-grade inflammation is common in aging, and it may be exacerbated by estrogen loss post-menopause.[ref]
Microplastics and nanoplastics in the bone:
A 2024 study showed that microplastics can be incorporated into bone stem cells, causing accelerated senescence which leads to excess RANKL production.[ref] While this is an animal study showing the mechanism of action, studies in humans also show microplastics accumulating in the bone marrow.[ref]
Related article: Microplastics Research Roundup
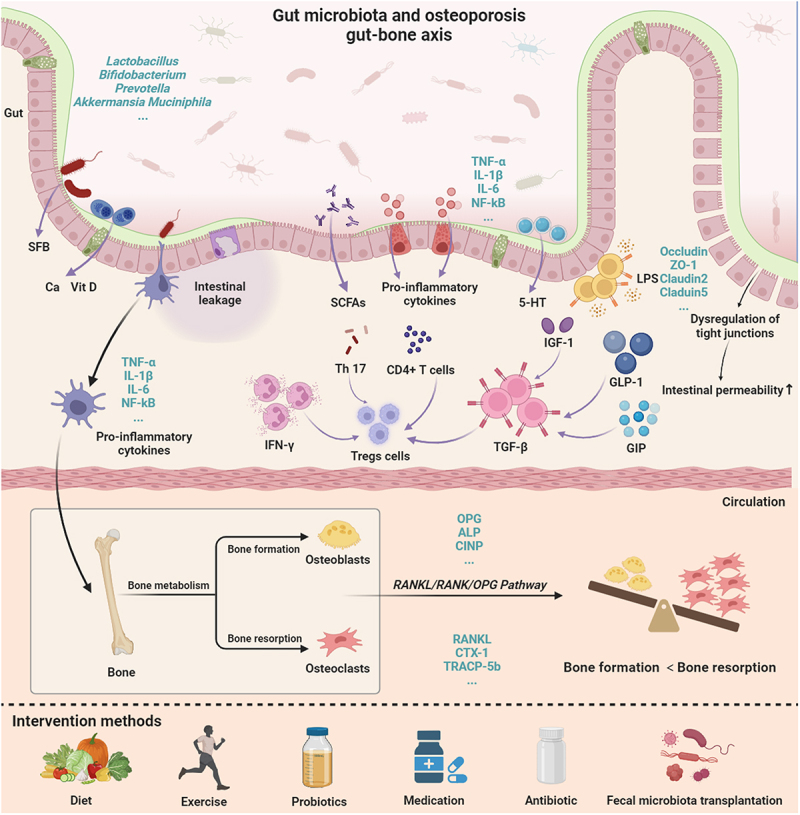
Gut microbiome and osteoporosis:
The gut microbiome can also affect bone health and inflammation in the body. When gut barrier function is impaired, microbes in the gut activate your immune system and increase inflammation in the body.[ref] New research shows that the gut microbiome directly affects bone metabolism and osteoporosis. Animal studies show that probiotics, such as Lactobacillus rhamnosus GG are protective against bone loss in a post-menopausal animal model. This was due to modulating the immune response and decreasing TNF-alpha and IL-17.[ref][ref][ref]
What lifestyle factors affect osteoporosis?
In addition to BMD, other risk factors include things like muscle strength, femoral neck geometry, nutrient deficiencies, and age at menopause. Those can also be partially due to genetics as well as caused by lifestyle choices (exercising, foods that you eat, etc.).
Environmental factors, of course, play a role in osteoporosis as well. Some non-genetic risk factors for osteoporosis include alcohol, smoking, age, and poor nutrition.[ref]
Other risk factors include:[ref][ref]
- taking oral glucocorticoids
- alcohol intake of 3 units or more a day
- low body mass index (less than 19)
- low estrogen in women, low testosterone in men
- excess thyroid hormones
- low vitamin D and calcium intake
Genes involved in Osteoporosis:
TNFSF11 gene:
TNFSF11 encodes tumor necrosis factor (ligand) 11, also called RANKL, which is a protein that is involved in immune response, osteoclast differentiation, and bone remodeling. One of the newer pharmaceutical options for osteoporosis is a drug known as a RANKL inhibitor. RANKL is the pathway for bone resorption, so blocking the resorption is thought to build stronger bones.
TNFSF11 is the gene that encodes the RANKL protein. In addition to bone resorption, RANKL plays an immune system role in other tissues such as the thymus, liver, colon, and more. Disruption of this gene in mouse models leads to severe osteoporosis as well as immune system problems.
LRP5:
The LRP5 gene codes for a protein involved in skeletal homeostasis. Several polymorphisms in LRP5 have been associated with lower bone mineral density. There are rare loss-of-function mutations causing severe osteoporosis and other, also rare, gain-of-function mutations that cause very dense bones.[ref]
SQRDL (sulfide quinone reductase-like) gene:
Sulfide quinone reductase is an enzyme that regulates the hydrogen sulfide levels in the cell. This enzyme is important in bone health because the bone marrow mesenchymal stem cells regulate their osteogenic differentiation by producing hydrogen sulfide. Researchers have created mice with low hydrogen sulfide and found that low HS caused osteoporosis. Moreover, giving the mice hydrogen sulfide can reverse bone loss.[ref] Notably, several other recent studies also point to hydrogen sulfide as a regulator of bone formation.[ref]
VDR gene:
Vitamin D is important in both calcium absorption and in regulating bone cell functions, and the VDR gene encodes the vitamin D receptor. There are several significant genetic variants in the VDR gene that have been studied for their association with osteoporosis.
ESR2 gene:
Estrogen deficiency is also a risk factor for osteoporosis. A polymorphism in the estrogen receptor 2 gene has also been associated with osteoporosis in postmenopausal women.
TGFB1 (transforming growth factor-beta 1) gene:
The TGFB1 gene codes for a protein that regulates cell growth, maturation, and cell death. A 2015 meta-analysis looked at the association between the TGFB1 polymorphisms and postmenopausal osteoporosis risk. The results showed an increased risk, but only for Asian women and not for Caucasian women.
Specialized pro-resolving lipid mediators (SPMs):
The resolution of inflammation is an active process that utilizes lipid mediators derived from DHA and EPA (omega-3 fatty acids found in fish oil). Specifically, maresin is a lipid mediator that is important in the resolution of inflammation and also in bone health. Decreased maresin 1 levels are associated with an increased risk of osteoporosis.[ref] Genetic variants in the maresin receptor increase the risk of osteoporosis also.
Osteoporosis Genotype Report:
Access this content:
An active subscription is required to access this content.
Lifehacks for osteoporosis:
Lifestyle and environmental factors that influence osteoporosis risk that you can control include:
- cutting back on alcohol
- never smoking
- get enough calcium in your diet
- exercise regularly
- get vitamin D through supplements or sunshine
- eating a healthy diet that promotes a healthy gut microbiome[ref]
Natural RANKL Inhibitors
Access this content:
An active subscription is required to access this content.
Need help with reversing or preventing osteoporosis?
Check out what Jana Davis (Genetic Lifehacks PRO member) has to say about osteoporosis. She is a functional nutritionist who specializes in bone health.
Related Articles and Topics:
Serotonin 2A receptor: Psychedelic response and Alzheimer’s disease
Vitamin K: Bone Strength, Blood Clots, Calcification, and Genetics
References: