Once toxins are initially broken down in the body, they must be converted to a water-soluble molecule in order to be excreted. Genetic variants in the NAT genes significantly influence the cancer risk due to toxicants, including cigarette smoke.
NATs: N-acetyltransferase
N-acetyltransferase is a phase II detoxification enzyme getting rid of several types of toxicants.
This enzyme helps your body eliminate aromatic amines, drugs, cigarette smoke, and carcinogens. Basically, it makes specific toxins more water soluble so that they can be excreted through a process called acetylation.
There are several common genetic variants that can classify a person as a slow, intermediate, or rapid acetylator. The rapid acetylator phenotype is considered to be the ancestral type and is the most common type in Asian and African populations; Caucasian people are actually slightly more likely to be intermediate or slow acetylators.[ref]
Geneticists think that the slow NAT acetylation genotypes arose around 10,000 years ago with the shift to farming and herding. Population groups that are traditionally more hunter/gatherers are more likely to have fast NAT2.[ref]
Historically, N-acetyltransferase was first recognized in the 1950s to play a role in the metabolism of tuberculosis drugs. A significant percentage of people were found to be poor acetylators resulting in deleterious side effects of the tuberculosis drug isoniazid.
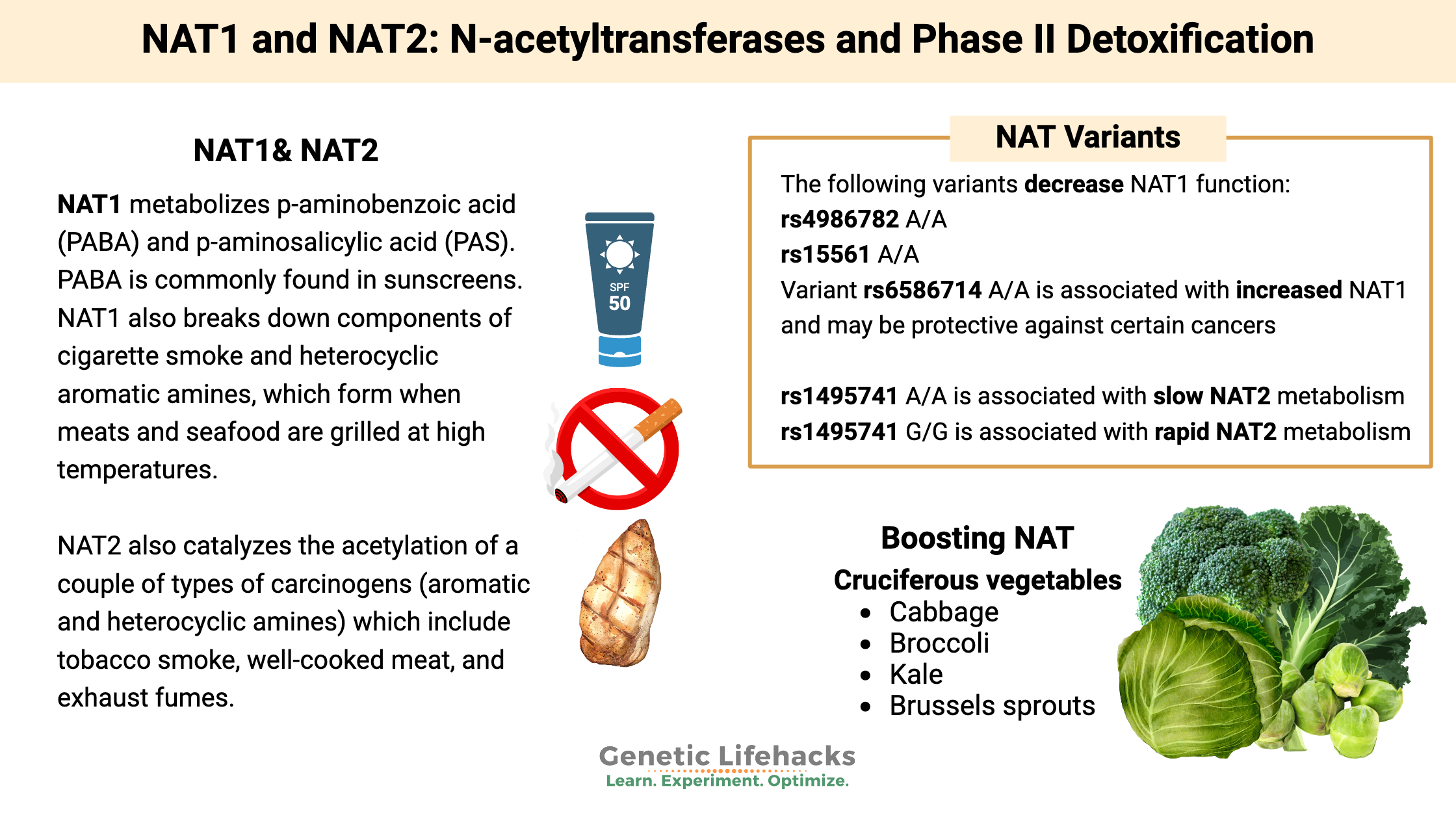
NAT1:
NAT1 metabolizes p-aminobenzoic acid (PABA) and p-aminosalicylic acid (PAS).[ref] PABA is commonly found in sunscreens, and PAS is used as an antibiotic for tuberculosis.
NAT1 also breaks down components of cigarette smoke and heterocyclic aromatic amines, which form when meats and seafood are grilled at high temperatures.
Studies link NAT1 genetic variants to an increased risk of bladder, colon, breast, lung, prostate, and pancreatic cancers.[ref]
Smoking is not good for anyone, but it is even riskier for those with NAT1 slow acetylator variants. NAT1 slow and intermediate acetylators had been shown to have a significantly higher risk for esophageal cancer and lung cancer in smokers.[ref]
Research shows that NAT1 slow acetylators have more DNA adducts formed with heterocyclic aromatic amines (carcinogens from meat being cooked at high temps). DNA adducts are segments of DNA that are bound to a carcinogen and are often a precursor for cancerous cells.[ref] Another study shows that red meat consumption for slow and intermediate acetylators increases the risk of esophageal cancer.[ref]
NAT1 is also involved in folate metabolism, and folate may be a co-enzyme for NAT1 hydrolysis of acetyl-Coenzyme A.[ref] Rs15561 -A/A (below) has been associated with cleft lip (especially if the mother smokes) and spina bifida. Note that it is the baby’s genotype, not the mother’s, that is being studied, so this is another really good reason not to smoke when pregnant.[ref] Both of those conditions are linked to folate metabolism and the methylation cycle.
NAT2:
NAT2 also catalyzes the acetylation of a couple of types of carcinogens (aromatic and heterocyclic amines) which include tobacco smoke, well-cooked meat, and exhaust fumes. NAT2 has been studied extensively and is thought to play a role in the risk of several types of cancers.
NAT2 is found mainly in the liver and intestines, in contrast with NAT1, which is found throughout the body.[ref]
NAT2 slow acetylators:
Low NAT2 function due to genetic variants causes a slow acetylator phenotype. NAT2 is important in the detoxification of xenobiotics, which are chemicals that come into your body such as acrylamide or particulates from smoke or smog. This is important when exposed to carcinogens, but it also may play a role in the metabolism and side effects of certain drugs. Many of these studies on drug interactions with NAT2 are on tuberculosis drugs. In addition, for transplant patients on immunosuppressants, there can be significant drug interactions with certain antibiotics.[ref][ref]
NAT1 Genotype Report:
Members: Log in to see your data below.
Not a member? Join here.
Why is this section is now only for members? Here’s why…
Access this content:
An active subscription is required to access this content.
Lifehacks for NAT1 and NAT2:
Stop smoking:
If you need a reason not to smoke, being a slow or intermediate acetylator is a really good reason never to pick up a cigarette. Cigarette smoke contains various carcinogens, including aromatic and heterocyclic amines. When inhaled, these carcinogens can undergo metabolic activation by NAT1 and NAT2 enzymes, potentially leading to the formation of DNA adducts and increased cancer risk.
Many studies over the past two decades have shown an interaction between cigarette smokers with low NAT2 activity and bladder cancer. The increased risk ranges from 2 to 6-fold, depending on the study population group.[ref][ref]
The risk of bladder cancer is increased even more for people with GSTM1 null genotypes. Smokers who were GSTM1 null and low NAT2 were at a 20-fold increased risk of bladder cancer.[ref]
GSTM1 gene:
Check your genetic data for rs366631 (23andMe v4 only):
- A/A: deletion (null) GSTM1 gene. 2x increased risk of breast cancer[ref], increased susceptibility to lung cancer in smokers[ref], increased risk of prostate cancer in Caucasians[ref] (common genotype in many population groups)
- A/G: GSTM1 present
- G/G: GSTM1 present
Members: Your genotype for rs366631 is —.
Breast cancer risk is also increased in people who have low NAT2 activity genotypes. The increased risk includes both smokers and women exposed to second-hand smoke.[ref][ref] Again, the risk is even higher in women with both low NAT2 and GSTM1 null genotypes. For these women, long-term smoking increased the risk of breast cancer by 6-fold.[ref]
Lung cancer risk is also increased in people with low or intermediate NAT status who also smoke.[ref]
Dietary changes:
Diet may interact with the NAT1 and NAT2 genotypes.
Grilled meat is linked to an increase in specific types of colon cancer (p53 mutations) in people with fast acetylation genotypes.[ref] Overall, though, the increased colon cancer risk from grilled meat doesn’t depend on NAT2 status.[ref]
Eating more cruciferous vegetables:
Eating more cruciferous vegetables (cabbage, broccoli, kale, Brussels sprouts) decreases the risk of bladder cancer, regardless of NAT2 status in smokers. Cruciferous vegetables contain isothiocyanate, which has been shown to decrease cancer risk[ref] ITC is also available as a supplement if you hate eating cruciferous vegetables.
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
Detoxifying Phthalates: Genes and Diet
Phthalates are a type of chemical used as plasticizers to make plastics more pliable. There has been a lot of research on the endocrine-disrupting effects of phthalates. Your genetic variants may impact whether phthalates are a problem for you.
CYP2C19 – Metabolizing medications
Several popular drugs, such as omeprazole, esomeprazole, and clopidogrel, are metabolized by the CYP2C19 enzyme. There are important genetic variants in the CYP2C19 gene that changes how fast or slowly these drugs are broken down. Learn how these variants might affect their efficacy.
References: