Key takeaways:
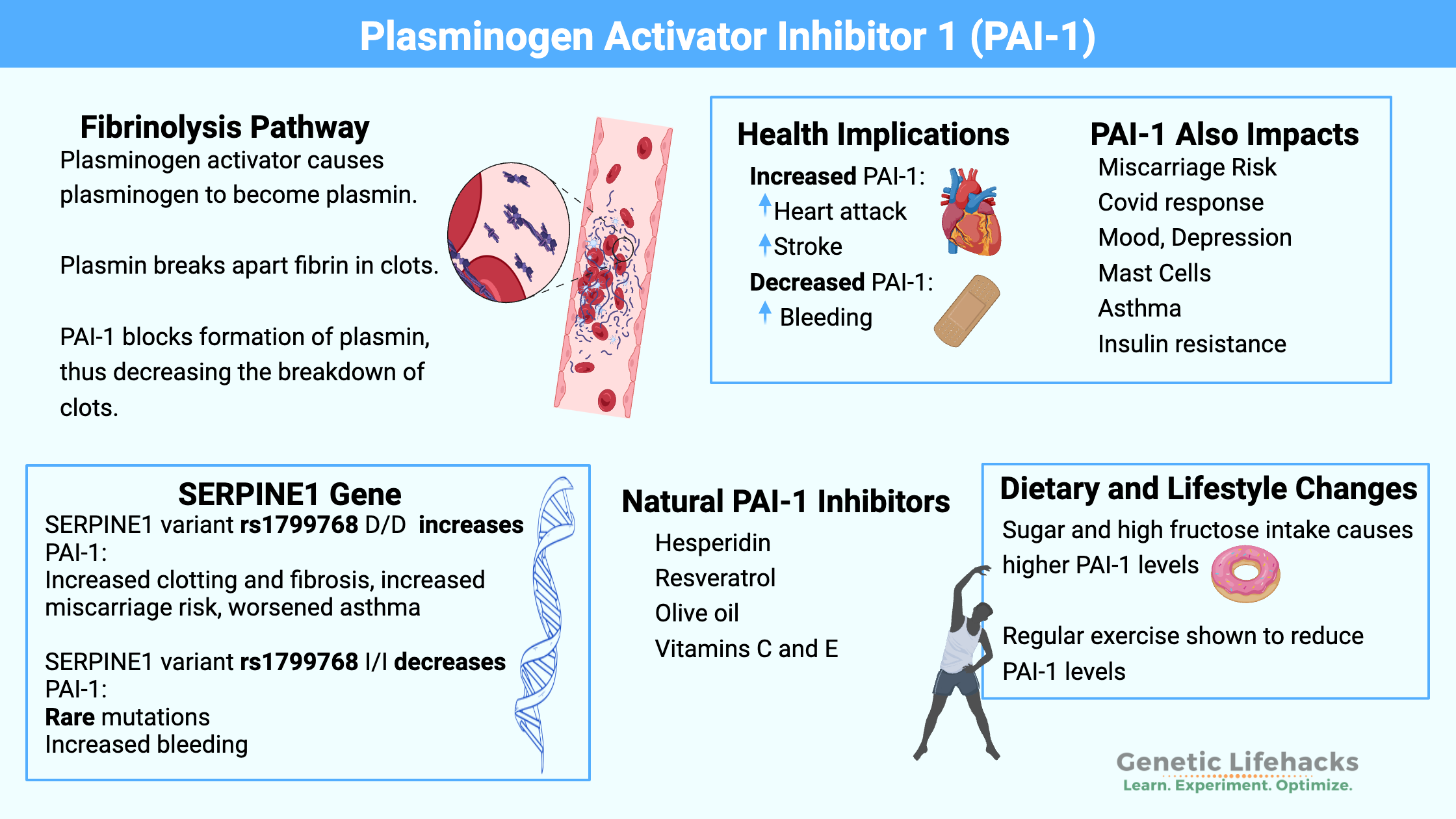
~ Plasminogen Activator Inhibitor (PAI-1) is an important enzyme in regulating the breakdown of blood clots and fibrosis.
~ Elevated PAI-1 levels are associated with an increased relative risk of heart attack, stroke, blood clots, tissue fibrosis, miscarriage, and more.
~ Genetic polymorphisms in the SERPINE1 gene can increase PAI-1 levels.
~ Lifestyle, diet, and natural supplements can help to reduce elevated PAI-1.
Members will see their genotype report below, plus additional solutions in the Lifehacks section. Consider joining today.
What is Plasminogen Activator Inhibitor-1 (PAI-1)?
PAI-1 is a serine protease inhibitor. Proteases are enzymes that break apart peptide bonds in proteins. Serine is an amino acid that is part of this type of protease enzyme.
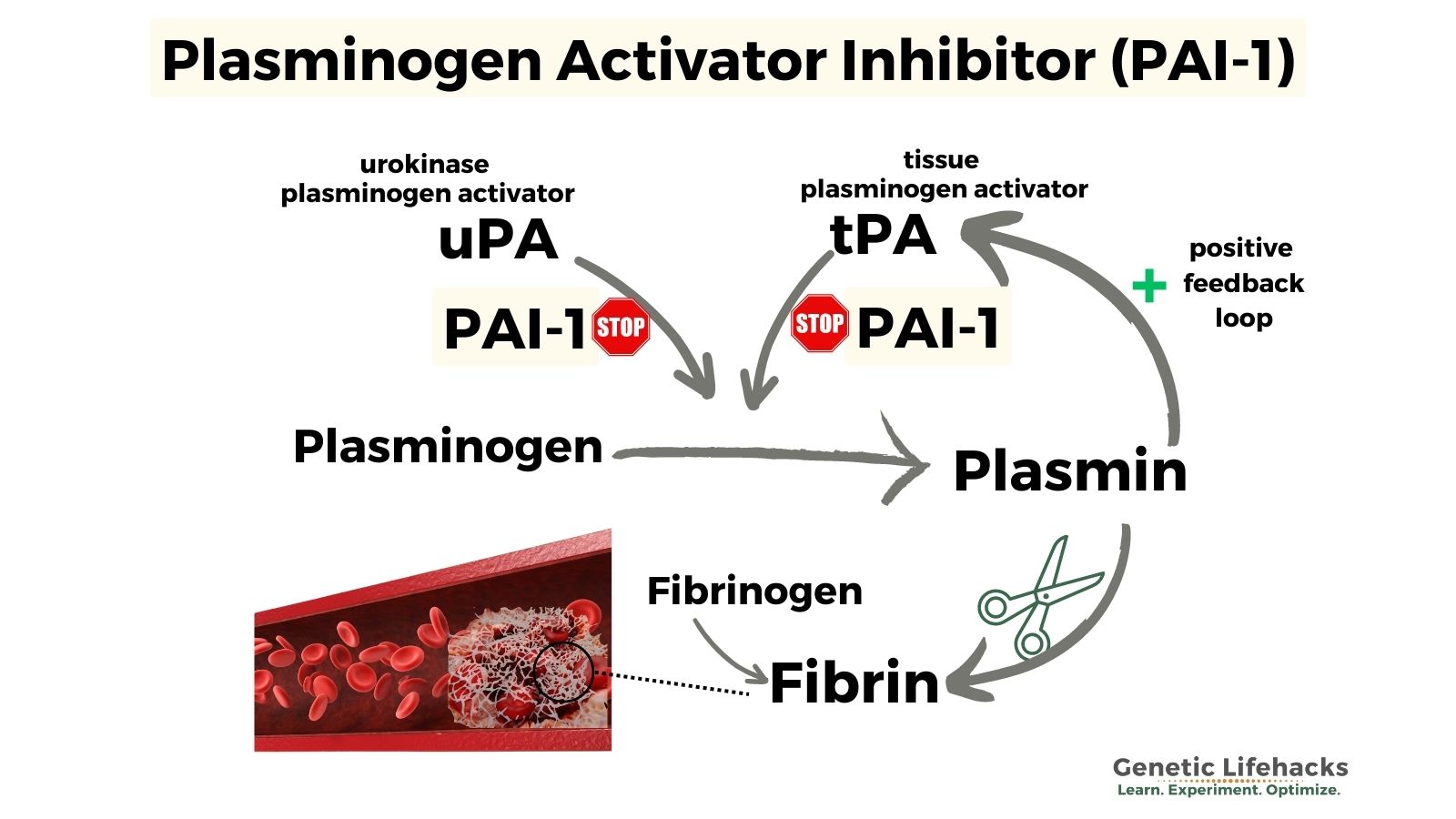
PAI-1 inhibits the action of tissue-type plasminogen activator (tPA). Tissue-type plasminogen activator is involved in degrading the fibrin network in clots through a process called fibrinolysis. In this way, tPA causes the breakdown of clots. Inhibiting tPA with PAI-1 therefore prevents too much fibrinolysis (too much clot breakdown). Tissue plasminogen activator is synthesized by endothelial cells that line blood vessels.
PAI-1 also inhibits urokinase plasminogen activator (uPA), which is the enzyme that cleaves plasminogen to form plasmin. [ref] uPA is synthesized in monocytes and macrophages, which are immune system cells. It is also synthesized in urinary epithelial cells.
By inhibiting the activation of plasminogen to plasmin, PAI-1 is part of the continual balancing act of breaking down and forming the fibrin network in clots.
In a nutshell, PAI-1 inhibits fibrinolysis, the process of breaking down clots.[ref] It is key to the delicate balance in clotting:
- Too little PAI-1 can increase the risk of hemorrhaging or excessive bleeding
- Too much PAI-1 can increase the risk of inappropriate blood clot formation
Background science: What is fibrinolysis?
Blood clots form in response to wounds or other inflammatory stimuli. Clots are formed from activated platelets, red blood cells, and cross-linked fibrin proteins that form a mesh. The fibrin mesh is made through a series of steps from fibrinogen, a protein that circulates in the blood.[ref]
Blood clots actively dissolve and reform in a continuous process. It is a tightly regulated balancing act between fibrin formation and fibrinolysis.
- Activation of plasminogen: Plasminogen is an inactive form of plasmin that circulates in the blood. Fibrinolysis begins when plasminogen is converted into plasmin, its active form. Tissue plasminogen activator (tPA) and urokinase plasminogen activator (uPA) are the enzymes that convert plasminogen into plasmin, in order to break down fibrin. tPA is released from the endothelial cells in response to the initiation of the clotting process (thrombin).
- Active plasmin: Plasmin is an enzyme that cleaves fibrin into fibrin degradation products. D-dimer is one of these fibrin degradation products and is usually measured to see if someone has a clot.
- Feedback loop: In a positive feedback loop, plasmin activates tPA and uPA, leading to further activation of plasminogen to plasmin and increasing the breakdown of the clot.
- Inhibition of plasmin: This is where PAI-1 comes in – it inhibits the activation of plasminogen into plasmin and acts as a brake on the system. There is also another protein, alpha-2-antiplasmin, which can inhibit the active plasmin.
Feedback loops, regulation of the processes — when everything is in balance, the clot forms quickly, is constantly remodeled, and then dissolves away when it is no longer needed.
Too little fibrinolysis is called hypofibrinolysis. This can lead to a thrombosis (clot) that can travel to the heart or lungs and be fatal. Hypofibrinolysis can be caused by autoantibodies to tPA or uPA or by genetic variants that increase PAI-1.
Plasmin Activator Inhibitor family:
PAI-1 is responsible for about 60% of the PA inhibitory activity in the plasma, but there are two other enzymes, PAI-2 and PAI-3, which are involved in PA inhibition under certain circumstances (pregnancy, tumors).
PAI-1 levels are associated with cardiovascular disease, metabolic disease, cancer, tissue fibrosis in inflammation, neurodegenerative disease, and mood disorders.[ref]
Let’s take a look at what all PAI-1 levels impact:
Blood clots, heart attacks, and strokes:
Myocardial infarction: Researchers have known for three decades that certain variants in the gene that codes for PAI-1 cause an increased risk of heart attack due to blood clots. The variants increase the amount of PAI-1 that is made by the body, and so there is less degredation of the clot.[ref][ref]
Blood clots: PAI-1 levels are often elevated in people with deep vein thrombosis (DVT).[ref][ref] Similarly, the risk of venous thromboembolism is increased with higher levels of PAI-1.[ref]
Atherosclerosis: Atherosclerotic plaque formation is associated with higher PAI-1 expression in the local plaque area. Not all studies agree on whether higher circulating levels of PAI-1 are involved in the development of atherosclerosis, and there are likely other factors involved.[ref]
Stroke: Ischemic strokes are caused by a blood clot in the brain that reduces blood flow to an area. Genetic variants that increase PAI-1 levels are linked to an increased risk of stroke.[ref] One drug that people who have an ischemic stroke receive is recombinant tPA to increase plasmin levels to dissolve the clot.
Heart valve stenosis:
In a study of aortic valves undergoing replacement, researchers found that fibrosis in the valves correlated with higher levels of PAI-1 as well as higher levels of LDL.[ref]
PAI-1 and tissue Fibrosis:
Fibrosis occurs when too much extracellular matrix is deposited, such as in a scar that forms during wound healing.
While we usually think of scarring and wounds in terms of a cut on the skin, fibrosis can also occur when there is an insult or dysfunction in an organ and PAI-1 helps control the breakdown of fibrin. For example, lung fibrosis can occur due to smoking or asbestos exposure[ref].
PAI-1 balances the formation of fibrin by uPA and tPA. Normally, when there is a wound, levels of PAI-1 are somewhat elevated to inhibit uPA and tPA and speed wound healing.[ref]
In general, if PAI-1 levels are too high, there may be excessive accumulation of extracellular matrix proteins and collagen. When PAI-1 levels are too low in the heart, spontaneous fibrosis can also occur.[ref]
Animal studies show that overexpression of PAI-1 causes an increase in lung fibrosis, such as in hypersensitivity pneumonitis, idiopathic pulmonary fibrosis, silicosis, and cystic fibrosis.[ref]
In the brain: PAI-1, neurotransmitters, and mood
Plasminogen activator inhibitor-1 is found in areas of the brain associated with mood (prefrontal cortex, amygdala, hippocampus) as well as in the bloodstream of the brain. Studies show that depressed patients have higher plasma levels of PAI-1. Animal studies show that chronic, unpredictable mild stress causes an increase in PAI-1 in the brain. BDNF is a neurotrophic factor that interacts with PAI-1. The precursor form, proBDNF, is cleaved by plasminogen activator (PA) to form the mature form of BDNF. BDNF helps the brain form new synaptic connections and maintain plasticity. [ref]
Animal research shows that decreasing PAI-1 levels causes depressive-like behaviors. The results showed that PAI-1 plays a role in depression beyond simply inhibiting PA, which could decrease BDNF. Instead, the researchers found that a lack of PAI-1 impairs the metabolism of serotonin and dopamine.[ref]
Stress response:
When you are stressed, cortisol is released. The increase in cortisol causes a number of different genes to be expressed or blocked. One effect of increased cortisol is to increase the production of tissue plasminogen activator (tPA) and PAI-1. In the brain, however, the stress response shifts so that there is an increase in PAI-1 compared to tPA. Researchers have found that this increased ratio of PAI-1 to tPA is part of the negative effect of stress on cognitive function. Blocking pro-BDNF to BDNF is thought to be part of the stress-related effects on memory and cognitive function.[ref]
PAI-1 and cancer:
PAI-1 is thought to have a function beyond its anti-fibrinolytic effects when it comes to cancer. Most research suggests it has a pro-tumor promotion role.[ref]
In pancreatic tumors that express PAI-1, there is an increased risk of blood clots (venous thromboembolism).[ref]
Mast cells, asthma, and PAI-1:
Mast cells induce PAI-1 expression in endothelial cells.[ref] In asthma, mast cell activation causes the release of histamine and proteases that contribute to asthma symptoms.[ref] Mast cells also play a role in tissue fibrosis in asthma, and PAI-1 is thought to be part of this pathway.[ref] More PAI-1 leads to fibrosis.
Researchers have found that genetic variants that increase PAI-1 levels are associated with more airflow obstruction in asthma patients, along with lower forced expiratory volume (FEV1).[ref]
COVID-19 and PAI-1:
For the original strain of SARS-CoV-2, patients with severe symptoms had higher PAI-1 levels than those with milder disease. Elevated initial levels were also found to be predictive of disease severity.[ref]
Another study also found that PAI-1 and its cofactor vitronectin were elevated in Covid patients when compared to other non-Covid respiratory infections and to healthy individuals. Additionally, the study found that there was increased clot fiber length and branching with decreased clot degradation due to the high PAI-1 activity.[ref]
A cell study in lung epithelial cells showed that the spike protein in combination with TLR2 (toll-like receptor 2, an immune system molecule) “enhances PAI-1 expression, potentially affecting the local coagulation cascade.”[ref]
I mentioned above that stress and cortisol increase PAI-1 expression. Similarly, blocking the cortisol receptor (MR) prevents the increase in PAI-1 in response to the spike protein.[ref]
A study of 100 people found that the Covid mRNA vaccination did not increase PAI-1 compared to a control group (who had previously been vaccinated with the adenovirus Covid vaccine). [ref]
However, another study showed that the S1 spike protein alone “stimulated robust production of PAI-1 by human pulmonary microvascular endothelial cells (HPMECs).”[ref]
Senescence, aging, and plasminogen activator inhibitor:
Cells are constantly being replaced in the body. For example, intestinal epithelial cells have a life span of 3-5 days. Heart muscle cells and brain cells, on the other hand, have very slow or no turnover.
Cellular senescence is the state in which a cell can no longer replicate and regenerate. Instead, it stops dividing and releases inflammatory signals so that it can be eliminated by the immune system. This is an essential check to prevent excessive growth (e.g., cancer).
As we age, the number of senescent cells overwhelms the immune system’s ability to eliminate them, leading to chronic low-grade inflammation.
PAI-1 levels increase with age, and levels correlate with the number of senescent cells. In 2006, PAI-1 was identified as a driver of cellular senescence. Research studies over the last decade show that PAI-1 is an essential part of cellular senescence and that inhibiting PAI-1 can reduce senescence in certain situations. [ref]
Endothelial nitric oxide synthase:
PAI-1 has recently been found to bind to endothelial nitric oxide and inhibit its production and bioavailability.[ref] Endothelial nitric oxide is a potent vasodilator, which in turn means that it lowers blood pressure.
While the research here is still quite new, it is important to keep in mind that PAI-1 has functions other than inhibiting tPA and uPA.
Insulin resistance:
One reason for cardiovascular complications in type 2 diabetes may be elevated PAI-1 levels. Insulin resistance causes an increase in PAI-1 production.[ref][ref]
PAI-1 levels are also elevated in some people with obesity.[ref] PAI-1 is made in the liver, and fatty liver may be the reason why PAI-1 is elevated in insulin resistance and obesity.[ref][ref]
PAI-1 and pregnancy:
During pregnancy, PAI-1 levels naturally increase, which increases coagulation and clotting factors. PAI-1 levels increase gradually in a healthy pregnancy, reaching a maximum level at 32-40 weeks. Levels return to normal a month or two after delivery.
Women with higher-than-normal PAI-1 levels have an increased relative risk of miscarriage, pre-eclampsia, and gestational diabetes.[ref]
PAI-1 4G/5G Polymorphism:
A common variant in PAI-1 is called 4G/5G due to a repeated section in the promoter region of the SERPINE1 gene, which codes for PAI-1. The PAI-1 5G allele is the more common allele, however, the 4G variant is found in around 35-40% of the population depending on the ancestry group. It is linked to two different rs ids, rs1799768 (below in the genotype report) and rs1799889.
Some fertility websites link the PAI-1 4G allele to an increased relative risk of recurrent miscarriages. However, not all studies agree on whether the PAI-1 4G allele is a statistical risk in recurrent miscarriages.
An analysis of studies on PAI-1 4G/5G for placenta-mediated pregnancy complications showed that in 32 out of 59 studies, there was no increased risk of complications.[ref]
So there may be an association, but it’s not seen in all studies. It could be due to interactions with other genetic variants that are common in certain ancestry groups, or it could be an interaction with an environmental or dietary factor.
Before taking any medical action, talk to your fertility doctor about how the variant may affect your specific situation.
Can you determine PAI-1 4G / 5G polymorphism from your 23andMe or Ancestry raw data?
A PAI-1 4G marker SNP is available in 23andMe version 4 data, which you can see below in the genotype report if you have that version of raw data. To know which version of 23andMe raw data you have, look at the filename of your downloaded file. At the end of the filename, it will say v4_full.txt for the version 4 data. If it says “v5”, you have the newer version 5 data file that doesn’t appear to have the PAI-1 4G information. (Here’s how to download 23andMe data)
However, there are other genetic polymorphisms that also affect PAI-1 levels, and these variants are included in 23andMe v5 and AncestryDNA raw data.
PAI-1 Genotype report:
The SERPINE1 gene encodes plasminogen activator inhibitor-1 (PAI-1).
Access this content:
An active subscription is required to access this content.
Lifehacks:
If you are trying to reduce fibrosis or clotting, inhibiting PAI-1 may be helpful. However, keep in mind that this could also cause increased bleeding risk. Please talk with your doctor or pharmacist about drug interactions if you are taking any medications before starting a supplement.
Dietary and Lifestyle Interventions:
Which type of fat should I eat?
A cell study showed that saturated fats (palmitic acid and stearic acid) had no effect on PAI-1 expression. However, omega-6 polyunsaturated fats and omega-3 EPA caused a significant increase in PAI-1.[ref] Another study showed that both stearic acid (saturated fat) and oleic acid (omega-9 in olive oil) had no effect on PAI-1 levels. Other studies back this up, showing that saturated fat does not cause an increase in PAI-1, and that monounsaturated and polyunsaturated fats both increase PAI-1 at least a little bit.[ref][ref] Not all studies agree here. One study of both Japanese and American men found that those with higher serum levels of linoleic and arachidonic acid (omega-6 oils) had lower PAI-1 levels.[ref]
My takeaway: Doctors often recommend cutting out saturated fat for cardiovascular disease, but cutting down on saturated fat may not be effective for people with genetically higher PAI-1, if that is the cause of their heart problems.
Cut the sugar and soda:
One dietary interaction that seems more straightforward is that PAI-1 levels decrease on a low-glycemic-index diet (compared to a high-glycemic-index diet).[ref] Another study suggests that high fructose intake causes higher PAI-1 levels, along with fatty liver disease.[ref]
Regular exercise:
In people with PAI-1 4G/4G, regular exercise helped to reduce PAI-1 levels by 36%.[ref] Another clinical trial in people with fatty liver disease showed that exercise reduced PAI-1 levels significantly.[ref]
Natural PAI-1 inhibitors:
Access this content:
An active subscription is required to access this content.
Related articles:
Nattokinase and Lumbrokinase: Natural Supplements for Clotting and Heart Health
References: