Key takeaways:
- Your circadian clock is controlled by a set of core genes — CLOCK, BMAL1, PER, and CRY — that rise and fall in a 24-hour cycle. In people with depression, this cycle can be off kilter.
- Genetic variants in these clock genes can increase your risk for depression, anxiety, and seasonal affective disorder.
- Nighttime light exposure increases depression risk by 30%, while daytime light exposure reduces it by 20%, based on data from over 86,000 people.
- Antidepressants like SSRIs, ketamine, and lithium all act on the circadian system, and your clock gene variants may predict which treatments work best for you.
- The most effective intervention is also the simplest: get bright light during the day and keep it dark at night.
Note: This article is for informational purposes and does not constitute medical advice. If you are experiencing symptoms of depression, please consult a qualified healthcare provider.
Members will see their genotype report below, plus additional solutions in the Lifehacks section. Join today.
Genetics, Circadian Rhythm, and Depression:
In the brains of some people with depression, the molecular clock is broken. Core circadian genes that should rise and fall in a daily rhythm are desynchronized – the rhythm shifted in timing, weakened in amplitude, and out of step with each other. This finding, first demonstrated clearly in 2013 and reinforced by increasingly detailed research since, has reshaped how scientists understand mood disorders.
Researchers use genetics to understand the biological pathways that drive mood disorders. By looking at genetic variants that are more common in people with depression, researchers can pinpoint the root cause of depression for individuals.
Genetic studies show that circadian rhythm disruption can be an underlying cause of mood disorders, at least for at least a portion of people dealing with depression. And understanding your genes can help you to tailor your environment to promote mental health. Essentially, a loss of synchronization between your internal circadian rhythm and external cues (e.g. artificial light at night) is at the heart of depression or anxiety — for some people.
It’s not just a sleep issue, so stick with me here. Normally, your body temperature, cortisol levels, melatonin, and thyroid-stimulating hormone all rise and fall in a specific daily rhythm. Research shows that people with depression often have shifted daily rhythms — out of sync with the core circadian clock — for body temperature, cortisol, TSH, and melatonin.
Genetic variants in circadian rhythm genes combine with environmental factors — light exposure, sleep habits, work schedules, seasonal changes — to shape mood disorders. Understanding how your genes interact with the environment can help prioritize the right personalized solutions for you.
Definitions to keep us on track:
Before we get into the science, here are several types of depression that will be referenced:[ref]
- Dysthymia is a type of depression with a long-term loss of interest in life, low energy, low appetite, hopelessness, and poor concentration.
- Bipolar disorder, also called manic-depressive disorder, is characterized by episodes of depression (low energy, loss of motivation) and episodes of mania (high energy, reduced sleep, possibly out of touch with reality).
- Major depressive disorder (MDD) is a period of depressed mood, decreased motivation, and cognitive impairment. It is one of the most common psychiatric disorders, affecting an estimated 21 million adults in the United States alone.
Background Science: Core Circadian Rhythm Genes
Before we get into depression and your genetic variants, let’s look at how your circadian rhythm works.
At the core of biology for animals (and plants) are specific genes that code for a molecular circadian clock.
For millions of years, one environmental factor has been never-changing: the sun comes up in the morning and sets at night, creating a 24-hour rhythm that never changes. Electric lights — and more specifically, LED lights rich in blue wavelengths — have disrupted that ancient pattern in ways our biology hasn’t caught up with.
Our biological molecular clock, called the circadian clock, sets the rhythm of a vast portion of our genome over the course of about 24 hours. Research suggests that roughly 40–50% of protein-coding genes show circadian expression patterns, meaning they turn on and off in daily cycles. All of this is driven primarily by exposure to light in the blue wavelengths.
The molecular circadian clock is located in the hypothalamus, in a region called the suprachiasmatic nucleus (SCN). It operates through a feedback loop involving several key genetic players. During the day, CLOCK and BMAL1 proteins rise, activating cellular events that need to occur in daytime — and also switching on the genes for CRY and PER. Then at night, CRY1/2 and PER1/2/3 proteins accumulate, which feed back to inhibit CLOCK and BMAL1, signaling nighttime processes.
This negative feedback loop takes approximately 24 hours to complete, forming the basis of the circadian rhythm.
Example: Your body produces enzymes during the day to break down the foods and toxins you consume or are exposed to. At night, cellular maintenance processes take place, getting you ready for the next day.
Here’s a quick visual for you showing the Circadian Clock Genes:

Sleep and Circadian Rhythm:
One of the first things that people think about in regard to circadian rhythm is the sleep/wake cycle. We naturally sleep and wake on about the same schedule every day – or we would if it weren’t for alarm clocks, work, and social engagements.
Sleep is essential for good mental health, but it is just one aspect of the circadian rhythm. The clock also governs hormone secretion, body temperature cycles, immune activity, and cognitive function. All of these are important in mood and depressive disorders.
Setting the circadian clock.
For humans, the average circadian clock runs about 15 minutes longer than 24 hours. But we aren’t all average, and the circadian period varies between a few minutes less than 24 hours to about a half-hour longer.[ref].
To keep us on track, the body has a built-in clock setting mechanism: exposure to light. These external clues for setting the clock are called zeitgebers (German for time giver).[ref]
Imagine if your clock ran at about 24.5 hours instead of 24. Within a week, you would be about seven hours out of sync.
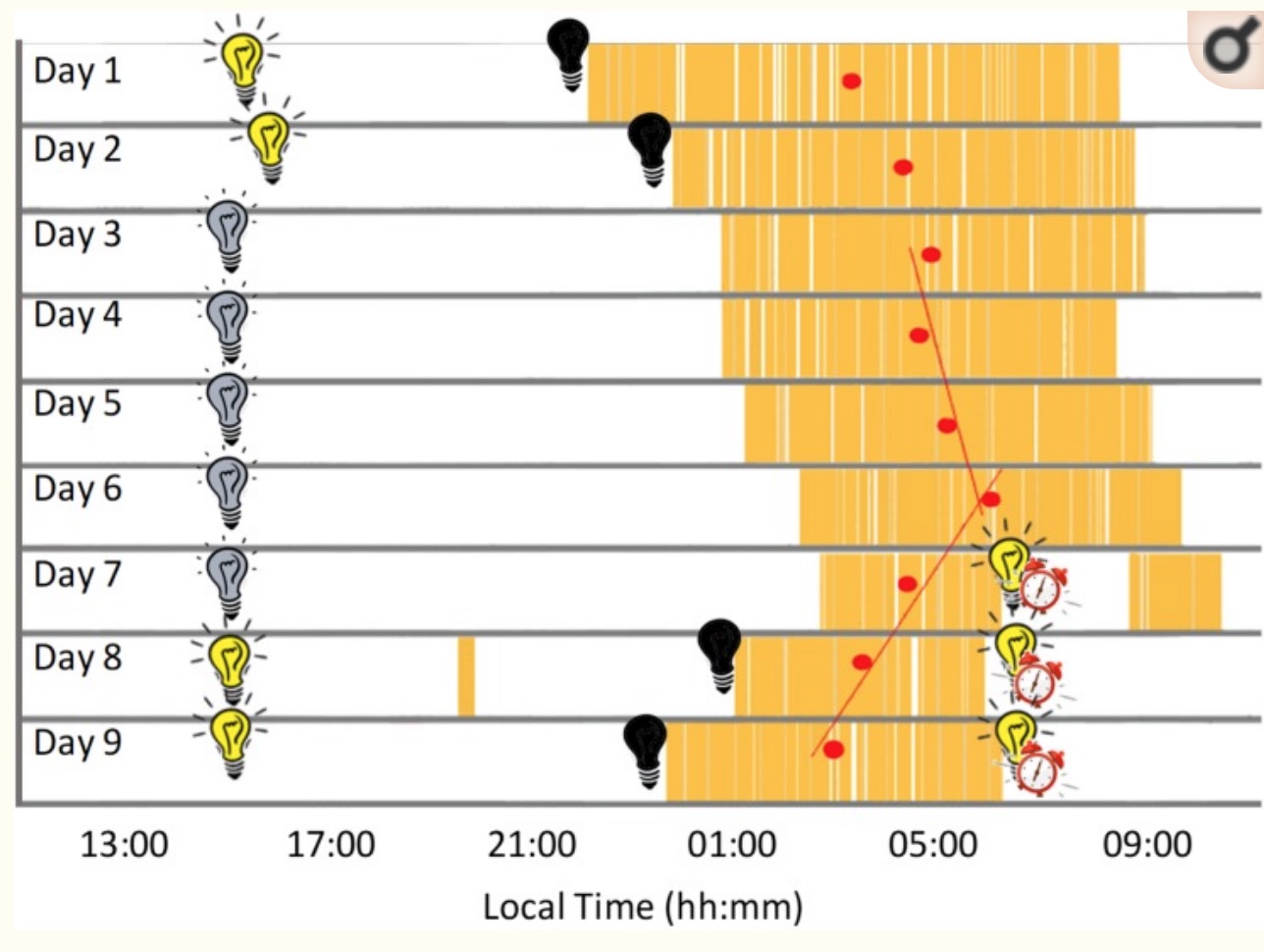
Researchers have demonstrated this in isolation studies. When participants are kept in caves or bunkers without access to outside light, their sleep-wake cycles drift. In one study, a participant stayed in a bunker for 9 days. During the first two days and last two days, he had lights available to turn on when he wanted. During days 3 through 7, only a dim light was constantly on. Sleep timing and sleep duration shifted dramatically throughout the week, illustrating how quickly the clock drifts without proper light cues. As you can see, sleep timing (in yellow) and sleep duration changed a lot throughout the week. [ref]
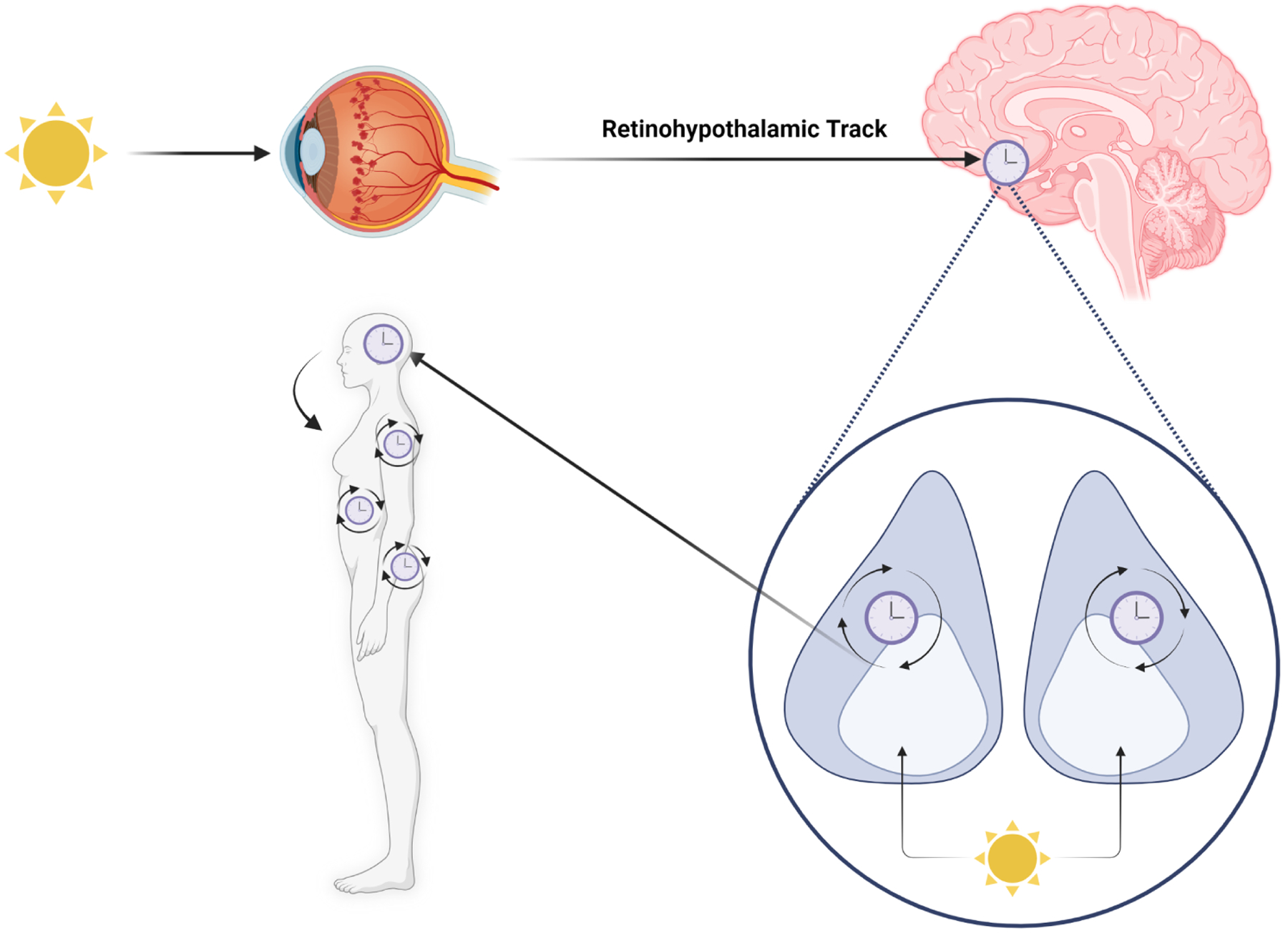
How Light Reaches the Clock: ipRGCs and Melanopsin
Recent research has provided a much more detailed picture of how light information reaches the master circadian clock in the hypothalamus. A specialized class of neurons in the retina called intrinsically photosensitive retinal ganglion cells (ipRGCs) use a photopigment called melanopsin to detect environmental light levels. These retinal cells don’t contribute to image formation; instead, they send a signal based on blue light activation directly to the SCN, synchronizing the body’s internal timekeeping with the external solar cycle. These ipRGCs work together with the classical photoreceptors (rods and cones) in setting the circadian rhythm.[ref]
How does melatonin come into play here?
Often referred to as the ‘sleep hormone’, melatonin levels rise at night when it is dark. It is a key molecule in circadian rhythm signaling that does much more than promote sleep.
Animal studies specifically show that melatonin is a key part of regulating the core clock genes. Without melatonin, the PER1 gene doesn’t rise and fall rhythmically.[ref]
Melatonin is produced by the pineal gland in large amounts at night, triggered by the absence of light signals through the ipRGC pathway. Exposure to artificial light at night significantly decreases overnight melatonin levels.
During the day, melatonin levels should be low – suppressed by bright, full-spectrum sunlight.
In people with depression, research shows that melatonin levels are often higher than they should be during the day. This is likely because indoor lighting is too dim to fully suppress melatonin production in some individuals, particularly those who are less sensitive to light’s melatonin-suppressing effect. It’s a kind of hyposensitivity that leaves them in a perpetual state of circadian ambiguity due to not having enough of a change in melatonin levels.[ref]
Master clock and peripheral clocks:
Your body’s core circadian rhythm is controlled in the part of the hypothalamus called the suprachiasmatic nucleus (SCN), but other tissues in the body also have a circadian rhythm, termed peripheral clocks. For example, your liver has a circadian rhythm that is somewhat separate from the core circadian clock in the SCN. While independent, it does get synchronized with the core circadian clock. Everything needs to work together for the majority of the time.[ref] Melatonin is one way the body synchronizes the central and peripheral clocks.
BMAL1: A core clock gene with broad effects
A recent large-scale genetic study showed just how broadly core clock genes influence health. Using UK Biobank data from approximately 500,000 participants, researchers looked at genetic variation in the BMAL1 gene across multiple ancestry groups and found associations with BMI, anhedonia, blood pressure, major depressive disorder, neuroticism, and risk-taking behavior.
Notably, the psychiatric and heart-related associations seem to be driven by distinct variants rather than shared genetic variants, suggesting BMAL1 plays multiple independent roles across body systems.[ref]
The rhythm of depression and anxiety:
Over the decades, researchers have come up with multiple theories about why circadian rhythm matters so much in depression and anxiety disorders.
The consensus is that mood disorders may reflect a loss of synchronization between the external environment’s rhythms and an individual’s internal rhythms — a mismatch with light and circadian rhythm. When this misalignment reaches a tipping point, it can lead to major problems such as depression, anxiety, or other psychiatric conditions.[ref]
When you are out of sync with your environment, it messes up your thoughts, sleep, body temp, emotions, and cognitive function.
Normally, your body temperature, cortisol levels, melatonin, and thyroid-stimulating hormone all rise and fall in a specific daily rhythm. The clinical reality of circadian desynchronization goes well beyond feeling “off.” People with depression often have shifted daily rhythms that are out of sync with the core circadian clock for their body temperature, cortisol, TSH, and melatonin production.[ref][ref]
Solid, overwhelming evidence: Light exposure affects mental health
A 2023 study using data from over 86,000 UK Biobank participants who wore wrist-mounted light sensors found that greater night-time light exposure was associated with increased risk for major depressive disorder, generalized anxiety disorder, PTSD, psychosis, bipolar disorder, and self-harm behavior. People exposed to high amounts of light at night had a 30% increase in depression risk. The flip-side was also true — those exposed to high amounts of daytime light saw a roughly 20% reduction in depression risk. The researchers took into account demographics, physical activity, sleep, shift work, and cardiometabolic health, and the light exposure findings still held true.[ref]
A 2025 meta-analysis of 19 studies covering over 556,000 participants showed that light at night was associated with increased odds of depression. The strongest associations were seen for indoor light measured at the bedside or wrist, rather than streetlights from satellite data.[ref]
These studies show that light at night (and lack of light during the day) carries real mental health consequences. So let’s take a look at the studies on specific genetic variants that play a role here.
Circadian rhythm genes in mental illness:
Neuropsychiatric diseases such as schizophrenia and bipolar disorder are tightly linked to the disruption of the core circadian rhythm genes. For example, in people with schizophrenia, cell studies show that there is a loss of circadian rhythm for the CRY1 and PER1 genes. Research also shows that there is a disruption in the CLOCK gene expression.[ref]
Researchers can manipulate the core circadian genes in animals – deleting genes or altering rhythms to see what happens. These studies give insight into what each circadian gene does.
CRY proteins: Too little or too much causes problems
Altering CRY protein levels in animals gives two different ends of the spectrum:
- When researchers knock out the CRY1/2 gene, the animals become anxious and are no longer active at the right time. Depression-like symptoms were less affected, but the animals show altered dopamine-related reward responses[ref]
- On the other hand, in animals that exhibited stress-induced depression-like helplessness, the researchers found higher than normal CRY expression. CRY was found to be regulating midbrain dopamine levels, which may play a role in depression.[ref]
In healthy people without depression, CRY2 levels rise and fall rhythmically over the course of 24 hours, and sleep deprivation causes a big increase in CRY2 levels. Strikingly, in people with depression, this pattern doesn’t happen: there is less of a CRY2 rhythm and no response to sleep deprivation.[ref]
CLOCK gene disruption in depressed brains:
In major depressive disorder, there is a disruption of the phase and amplitude of the core circadian clock genes. A landmark study in 2013 compared circadian gene expression in the brains of people with depression to a control group. The results showed that depressed people had weaker circadian clock gene patterns with shifted timing and “disrupted phase relationships between individual circadian genes.”[ref] While researchers had known for decades that there was a relationship between sleep, depression, and circadian rhythm, this study clearly showed that people with depression had a desynchronization in the core circadian clock genes.
In young people with depression, a delay in the circadian phase (later onset of melatonin and later drop in core body temperature) correlates with increased severity of depression.[ref] It is important to note here that not all of the young people with depression had a delayed circadian phase–only about 40% did.
PER protein and brain connectivity in adolescents:
A 2025 study of adolescents with major depressive disorder found reduced levels of CRY and PER proteins compared to a healthy control group. The depressed teens also showed reduced functional connectivity between the SCN and the orbitofrontal cortex — correlated to PER levels.[ref] Another study showed that there was an additive effect of multiple PER3 genetic variants (SNPs) in the risk of depression.[ref]
NPAS2 gene variants in the circadian-depression pathways:
A 2025 study involving 257 MDD patients found that variants in NPAS2 (a gene related to CLOCK) moderate the effect of an evening chronotype through sleep disturbances and depression. Specific NPAS2 variants strengthened the association between eveningness and poor sleep, and the overall mediation pathway from circadian preference to depression severity. [ref]
Circadian rhythm and seasonal depression (SAD):
Seasonal depression affects a lot of people living in higher latitudes in the winter months. It is defined as a period of fatigue, depression, and hopelessness – occurring with the changing of the seasons (usually late fall/winter).
Research shows that people with seasonal affective disorder (SAD) have a delay of about 1.5 hours in their melatonin production, compared to healthy controls. Along with the delay in melatonin, their body temperature rhythm was also delayed. Resetting this with bright light therapy in the morning shifted the melatonin rhythm back to normal and reversed depressive symptoms.[ref]
Depression medications that act on the circadian system:
Many common antidepressant medications impact circadian rhythm — sometimes intentionally, sometimes as an important side effect. Understanding these circadian effects may help explain why a given medication works well for some people and not others.
- Citalopram is a commonly prescribed SSRI. It actually makes people more sensitive to light during the daytime. Thus, if you are stuck in lower-level indoor lighting, it makes you more sensitive to the light and suppresses melatonin during the day by almost 50% more. The researchers note that this may be why citalopram works well for some people and doesn’t work (or has the opposite effect) for others.[ref]
- A large-scale study by 23andMe on bupropion found that the efficacy of the drug depended on a genetic variant involved in circadian rhythm pathways.[ref]
- For treatment-resistant depression, there are two methods that doctors can use that may immediately change mood: sleep deprivation (resetting of the clock) and ketamine. Low doses of IV ketamine are very effective for some people with depression. It is not FDA-approved for this usage in the US at this time, but some clinics offer it. Ketamine has recently been shown to act on depression via altering BDNF and increasing slow-wave sleep. It also alters the expression of PER and CRY.[ref][ref] Esketamine is a nasal spray formulation that is now FDA-approved to be administered in certified healthcare settings for depression.
- Fluoxetine has been shown in animal studies to normalize ‘light-induced’ circadian disruption. It does this through increased PER2 and PER3 levels.[ref]
- Agomelatine is a synthetic version of melatonin that is prescribed as an atypical antidepressant.[ref]
Circadian Rhythm Genotype Report:
The following circadian gene variants are linked with depression.
Access this content:
An active subscription is required to access this content.
Lifehacks for optimizing circadian rhythm:
Let me reiterate: this information is for educational purposes. If you are under a doctor’s care for a mood disorder, please check with your doctor before making any changes.
1) Shifting the Circadian Clock with light
Research shows that bright light in the morning advances the circadian clock, while exposure to light in the late evening or at night delays the clock. Even intermittent bright light exposures can shift the circadian phase.[ref]
Bright light in the morning
- A clinical trial of bright light therapy (7,000 lux for 1 hour in the morning after waking) showed that it was twice as effective as a placebo. The study group was composed of pregnant women, and almost 70% of the women attained remission using bright light therapy.[ref]
- In a randomized clinical trial of bright light therapy (7500 lux, 1 hour after rising) for older adults (age 60+), the results showed that light therapy improved mood, increased melatonin levels overnight, and enhanced sleep efficiency.[ref]
Practical idea: If you don’t have access to a light therapy device, try getting up a little earlier than normal and going outside. Eat breakfast, have coffee, read the news, walk the dog… Get in the habit of starting your day with a half-hour in the bright light.
Go outside during the day:
Have you ever gone hiking and found that you are ready to sleep earlier and generally sleep harder after being outside all day? Research shows that for each additional hour spent outside over the baseline, sleep is advanced by ~30 minutes.[ref]
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
Is Inflammation Causing Your Depression and Anxiety? The Science Behind the Link
Problems sleeping? Your genes can point to individualized solutions
References:
Burns, Angus C., et al. “Day and Night Light Exposure Are Associated with Psychiatric Disorders: An Objective Light Study in >85,000 People.” Nature Mental Health, vol. 1, no. 11, Nov. 2023, pp. 853–62. www.nature.com, https://doi.org/10.1038/s44220-023-00135-8.
Gardner, Wilf, et al. “The mPFC Molecular Clock Mediates the Effects of Sleep Deprivation on Depression-like Behavior and Regulates Sleep Consolidation and Homeostasis.” Molecular Psychiatry, vol. 31, no. 3, Mar. 2026, pp. 1530–45. www.nature.com, https://doi.org/10.1038/s41380-025-03276-7.
Huang, Qinte, et al. “NPAS2 Gene Variants Modulate the Circadian Preference-Depression Link in Major Depressive Disorder: A Mediation Role of Sleep and Somatic Symptoms.” Chronobiology International, vol. 42, no. 11, Nov. 2025, pp. 1566–76. DOI.org (Crossref), https://doi.org/10.1080/07420528.2025.2558107.
Guo, Qianru, et al. “The Relationship between the Circadian Protein PER and Adolescent Depression: The Mediating Effect of Aberrant Functional Connectivity of Suprachiasmatic Nucleus-Orbitofrontal Cortex.” Journal of Psychiatric Research, vol. 190, Oct. 2025, pp. 225–34. ScienceDirect, https://doi.org/10.1016/j.jpsychires.2025.08.001.
Li, Xiaomeng, et al. “Light at Night Exposure and Risk of Depression: A Meta-Analysis of Observational Studies.” Journal of Global Health, vol. 15, p. 04304. PubMed Central, https://doi.org/10.7189/jogh.15.04304.

