Currently, 10% of people age 65 or older have Alzheimer’s disease (AD), and the risk for Alzheimer’s disease increases considerably with age. It is a disease for which prevention needs to start decades before the symptoms appear.[ref]
One way to understand your Alzheimer’s risk – and to understand the mechanisms by which the disease occurs – is to look at genetic variants that increase the risk for Alzheimer’s. On the other hand, you can also learn a lot by looking at genetic variants that protect against Alzheimer’s.
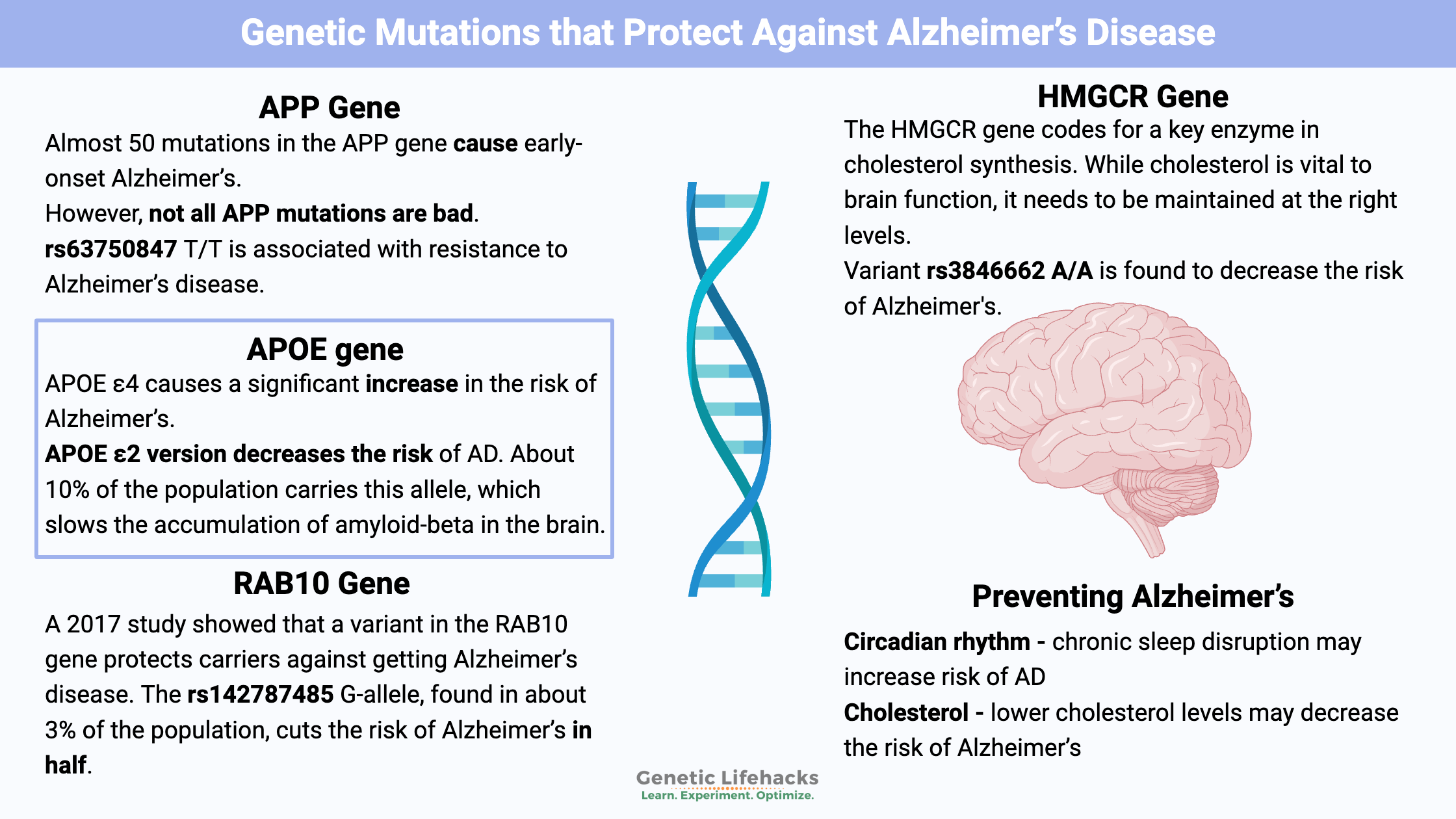
Genes combine with environmental exposure and lifestyle to determine your risk for Alzheimer’s disease. There are genetic variants that increase your risk for Alzheimer’s disease. Other variants decrease your risk for the disease.
Even if you don’t carry the protective variants below, a lot can be gleaned from understanding how the variants work and why they decrease the risk of Alzheimer’s. This seems to be a relatively new field of research, but it is interesting to note that several of these protective genetic variants are involved in either cholesterol or lipid metabolism in the brain.
Amyloid-β is a protein formed when enzymes cut the APP (amyloid precursor protein) protein into smaller fragments. Some of the protein fragments move out of the brain. But amyloid-β that isn’t cleared can build up around neurons, forming plaque.
There are currently no medications for curing Alzheimer’s disease, and research shows that preventative actions need to start decades before the onset of the disease.
Ali-Rahmani, Fatima, et al. “HFE Gene Variants, Iron, and Lipids: A Novel Connection in Alzheimer’s Disease.” Frontiers in Pharmacology, vol. 5, July 2014, p. 165. PubMed Central, https://doi.org/10.3389/fphar.2014.00165.
Allen, Mariet, et al. “Association of MAPT Haplotypes with Alzheimer’s Disease Risk and MAPT Brain Gene Expression Levels.” Alzheimer’s Research & Therapy, vol. 6, no. 4, 2014, p. 39. PubMed, https://doi.org/10.1186/alzrt268.
“Alzheimer’s Disease Facts and Figures.” Alzheimer’s Association, https://www.alz.org/alzheimers-dementia/facts-figures. Accessed 20 Feb. 2026.
Benn, Marianne, et al. “Low LDL Cholesterol, PCSK9 and HMGCR Genetic Variation, and Risk of Alzheimer’s Disease and Parkinson’s Disease: Mendelian Randomisation Study.” The BMJ, vol. 357, Apr. 2017, p. j1648. PubMed Central, https://doi.org/10.1136/bmj.j1648.
Bhattarai, Prabesh, et al. “Rare Genetic Variation in Fibronectin 1 (FN1) Protects against APOEε4 in Alzheimer’s Disease.” Acta Neuropathologica, vol. 147, no. 1, 2024, p. 70. PubMed Central, https://doi.org/10.1007/s00401-024-02721-1.
Bracher-Smith, Matthew, et al. “Whole Genome Analysis in APOE4 Homozygotes Identifies the DAB1-RELN Pathway in Alzheimer’s Disease Pathogenesis.” Neurobiology of Aging, vol. 119, Nov. 2022, pp. 67–76. PubMed Central, https://doi.org/10.1016/j.neurobiolaging.2022.07.009.
Corder, E. H., et al. “Protective Effect of Apolipoprotein E Type 2 Allele for Late Onset Alzheimer Disease.” Nature Genetics, vol. 7, no. 2, June 1994, pp. 180–84. PubMed, https://doi.org/10.1038/ng0694-180.
Dai, Qi-He, and Dao-Kai Gong. “Association of the Polymorphisms and Plasma Level of CHI3L1 with Alzheimer’s Disease in the Chinese Han Population: A Case-Control Study.” Neuropsychobiology, vol. 77, no. 1, 2019, pp. 29–37. PubMed, https://doi.org/10.1159/000492536.
English, Amber R., and Gia K. Voeltz. “Rab10 GTPase Regulates ER Dynamics and Morphology.” Nature Cell Biology, vol. 15, no. 2, Feb. 2013, pp. 169–78. www.nature.com, https://doi.org/10.1038/ncb2647.
Hampel, Harald, and Simone Lista. “Have We Learnt All We Need to Know from Genetic Studies – Is Genetics over in Alzheimer’s Disease?” Alzheimer’s Research & Therapy, vol. 5, no. 2, 2013, p. 11. PubMed, https://doi.org/10.1186/alzrt165.
Harold, Denise, et al. “Genome-Wide Association Study Identifies Variants at CLU and PICALM Associated with Alzheimer’s Disease.” Nature Genetics, vol. 41, no. 10, Oct. 2009, pp. 1088–93. www.nature.com, https://doi.org/10.1038/ng.440.
Jonsson, Thorlakur, et al. “A Mutation in APP Protects against Alzheimer’s Disease and Age-Related Cognitive Decline.” Nature, vol. 488, no. 7409, Aug. 2012, pp. 96–99. www.nature.com, https://doi.org/10.1038/nature11283.
Kim, Yeo Jin, et al. “Protective Effects of APOE E2 against Disease Progression in Subcortical Vascular Mild Cognitive Impairment Patients: A Three-Year Longitudinal Study.” Scientific Reports, vol. 7, no. 1, May 2017, p. 1910. www.nature.com, https://doi.org/10.1038/s41598-017-02046-y.
Lambert, Jean-Charles, et al. “Meta-Analysis of 74,046 Individuals Identifies 11 New Susceptibility Loci for Alzheimer’s Disease.” Nature Genetics, vol. 45, no. 12, Dec. 2013, pp. 1452–58. PubMed Central, https://doi.org/10.1038/ng.2802.
Lananna, Brian V., et al. “Chi3l1 /YKL-40 Is Controlled by the Astrocyte Circadian Clock and Regulates Neuroinflammation and Alzheimer’s Disease Pathogenesis.” Science Translational Medicine, vol. 12, no. 574, Dec. 2020, p. eaax3519. DOI.org (Crossref), https://doi.org/10.1126/scitranslmed.aax3519.
Leduc, V., et al. “HMGCR Is a Genetic Modifier for Risk, Age of Onset and MCI Conversion to Alzheimer’s Disease in a Three Cohorts Study.” Molecular Psychiatry, vol. 20, no. 7, July 2015, pp. 867–73. PubMed, https://doi.org/10.1038/mp.2014.81.
Leduc, Valerie, et al. “HMGCR Is a Genetic Modifier for Risk, Age of Onset and MCI Conversion to Alzheimer’s Disease in a Three Cohorts Study.” Molecular Psychiatry, vol. 20, no. 7, July 2015, pp. 867–73. PubMed Central, https://doi.org/10.1038/mp.2014.81.
Lemche, Erwin. “Early Life Stress and Epigenetics in Late-Onset Alzheimer’s Dementia: A Systematic Review.” Current Genomics, vol. 19, no. 7, Nov. 2018, pp. 522–602. PubMed Central, https://doi.org/10.2174/1389202919666171229145156.
Liu, Chia-Chen, et al. “APOE3 -Jacksonville (V236E) Variant Reduces Self-Aggregation and Risk of Dementia.” Science Translational Medicine, vol. 13, no. 613, Sept. 2021, p. eabc9375. DOI.org (Crossref), https://doi.org/10.1126/scitranslmed.abc9375.
Ridge, Perry G., et al. “Linkage, Whole Genome Sequence, and Biological Data Implicate Variants in RAB10 in Alzheimer’s Disease Resilience.” Genome Medicine, vol. 9, Nov. 2017, p. 100. PubMed Central, https://doi.org/10.1186/s13073-017-0486-1.
Ruiz, A., et al. “Follow-up of Loci from the International Genomics of Alzheimer’s Disease Project Identifies TRIP4 as a Novel Susceptibility Gene.” Translational Psychiatry, vol. 4, no. 2, Feb. 2014, p. e358. PubMed Central, https://doi.org/10.1038/tp.2014.2.
Shi, Yang, and David M. Holtzman. “Interplay between Innate Immunity and Alzheimer’s Disease: APOE and TREM2 in the Spotlight.” Nature Reviews. Immunology, vol. 18, no. 12, Dec. 2018, pp. 759–72. PubMed Central, https://doi.org/10.1038/s41577-018-0051-1.
Suri, Sana, et al. “The Forgotten APOE Allele: A Review of the Evidence and Suggested Mechanisms for the Protective Effect of APOE Ɛ2.” Neuroscience and Biobehavioral Reviews, vol. 37, no. 10 Pt 2, Dec. 2013, pp. 2878–86. PubMed, https://doi.org/10.1016/j.neubiorev.2013.10.010.
Tetzeli, Rick. “Could This Radical New Approach to Alzheimer’s Lead to a Breakthrough?” Fortune, https://fortune.com/longform/alzheimers-disease-cure-breakthrough/. Accessed 20 Feb. 2026.
Wright, Sage M., et al. “Association Study of Rs3846662 with Alzheimer’s Disease in a Population-Based Cohort: The Cache County Study.” Neurobiology of Aging, vol. 84, Dec. 2019, p. 242.e1-242.e6. PubMed, https://doi.org/10.1016/j.neurobiolaging.2019.03.004.