Key takeaways:
- NAD+ is a coenzyme in every living cell that powers mitochondrial energy production, DNA repair, immune function, and cellular stress response
- Levels decline 50% or more by age 60 due to increased consumption by PARP (DNA repair), CD38 (inflammation), and decreased production
- This decline drives multiple hallmarks of aging: fatigue, cognitive decline, metabolic dysfunction, and increased disease risk
- Genetic variants impact the body’s production of NAD+ and related genes, such as sirtuins, PARPs, and CD38, which interact to promote healthy aging.
- Nicotinamide riboside (NR) and nicotinamide mononucleotide (NMN) are available as supplements and increase NAD+ levels.
What is NAD+?
Imagine if you could turn back your cellular clock. Scientists have discovered that declining levels of a single molecule—NAD+—may be responsible for many hallmarks of aging. The good news? You can boost it. Here’s what you need to know…
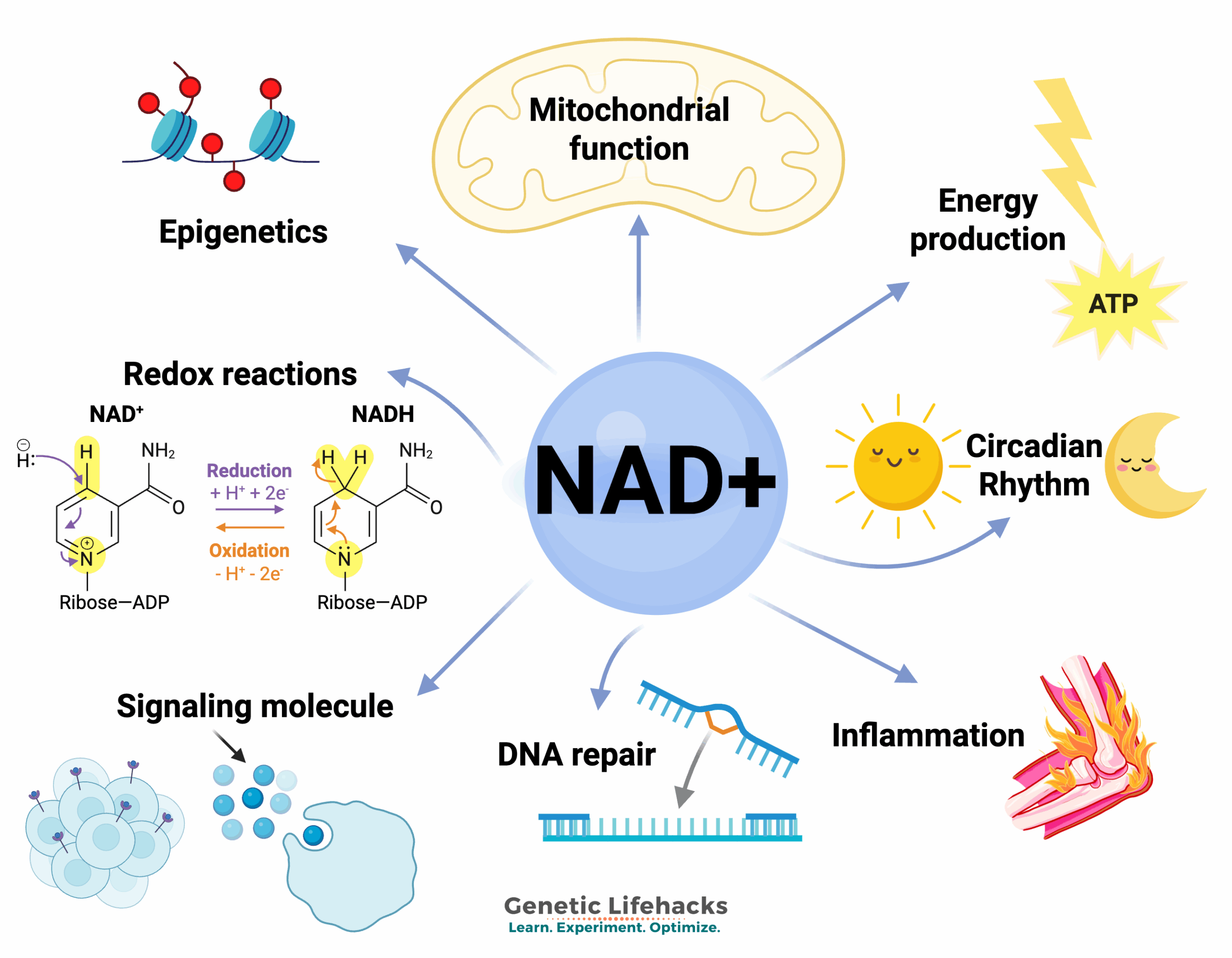
NAD+ (nicotinamide adenine dinucleotide) is an important molecule that all plants and animals produce and use in cellular processes, including ATP production. It is a niacin derivative necessary for multiple cellular functions — a molecule essential for life.
NAD+ is used in cells as an integral part of ATP (energy) production, powering the immune response, cell signaling, gene expression, repairing DNA, and maintaining telomeres.
In this article, we will look at:
- NAD+ in mitochondrial energy production
- Reactions that consume NAD+
- How NAD+ is synthesized in the body
- The decline in NAD+ levels as we age
- NR and NMN supplements to boost NAD+
- Genetic variants that influence your NAD+ pathways
- Solutions and lifehacks
Let’s start with the essential role of NAD+ in ATP production, and then look at reactions that deplete it.
NAD+ in cellular energy production:
In cellular metabolism, NAD+ is an essential part of energy production, shuttling electrons in redox reactions. When we eat food, it’s converted into the components needed by the cells to produce ATP for energy. For example, carbohydrates break down into glucose, which can then be directly used in cells to produce ATP, the molecule your body uses for energy storage.
During cellular energy production, the majority of ATP production comes from processes in the mitochondria (the powerhouse of the cell). This is where NAD+ is essential.
There are two ways ATP is synthesized in the mitochondria, and both involve NAD+:
- Within the Krebs cycle (a.k.a. citric acid cycle), electrons shuttle between NAD+ and NADH.
- Within the inner membrane of the mitochondria, the electron transport chain uses NAD+ for the transfer of electrons. This is your body’s main way of producing energy when enough oxygen is present.
Here’s what this looks like:
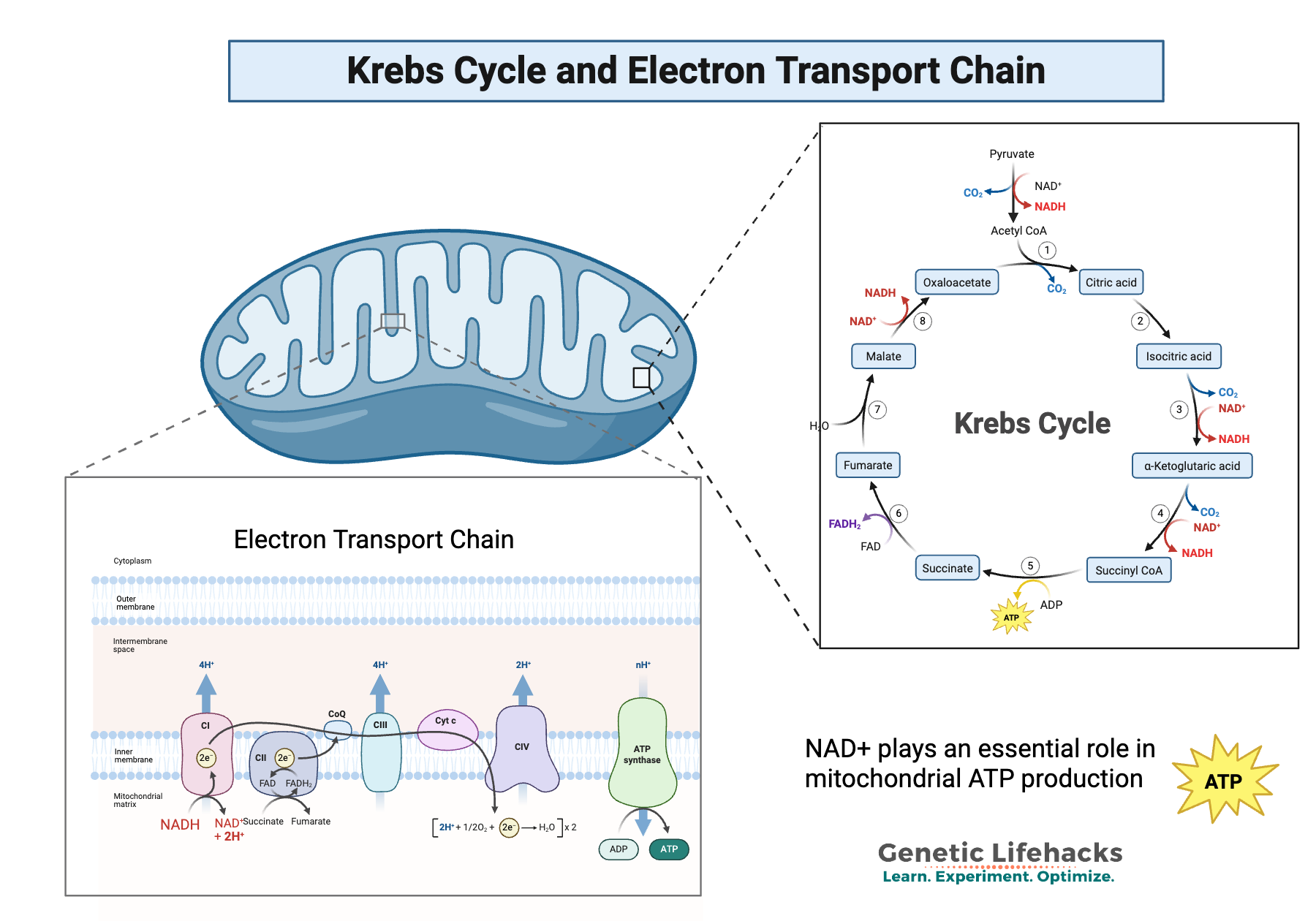
Keep in mind that in redox reactions, the ratio between the oxidized and reduced forms (NAD+/NADH) changes, but the NAD is not consumed. The total NAD+/NADH amount essentially stays the same in these mitochondrial processes for ATP production.
Other roles of NAD+: Reactions that consume NAD+
Beyond mitochondrial ATP production, NAD+ is also utilized in numerous other reactions in the body.
ADP-ribose is derived from NAD+ and acts as a versatile tag, attaching to proteins and nucleic acids to control processes like DNA repair, immunity, and gene regulation. It can form single units or long chains called poly(ADP-ribose) (PAR) to orchestrate cellular activities, especially in response to stress. ADP-ribose transfer reactions consume NAD+.
ADP-ribose transfer reactions include:
- The repair of DNA
- The maintenance of telomeres
- PARP and CD38/CD157 reactions
CD38/CD157: Immune system using up NAD+
Two very similar and related enzymes, CD157 and CD38, consume a lot of NAD+ in the body. CD157 acts both as an enzyme and as a cellular receptor on immune cells. It is important in immune system reactions, including in the brain. Research ties variants in CD157 to Parkinson’s disease and REM sleep disorder. Studies also find that CD157 variants impact autism risk related to the regulation of brain development.[ref] CD38 is a very similar molecule to CD157 and acts similarly, but in different cell types. Both molecules use up NAD+ by acting as enzymes that catalyze the degradation of NAD+ in order to use the metabolites as messenger molecules. CD157 and CD38 also play an important role in the immune system and are found as surface receptors on immune cells. Both are also involved in the immune response and neuroinflammation in the brain.[ref] (More on CD38 and CD157 in the Genotype report section and Lifehacks section)
Sirtuins:
Sirtuins are a family of proteins (SIRT1 through SIRT7) essential for turning on and off the translation of genes within a cell. For instance, SIRT1 is linked to nutrient sensing and insulin resistance, and its enhancement can help resist obesity-related problems. Other sirtuins, like SIRT6, play roles in metabolic regulation and mitochondrial function. All sirtuins depend on NAD+ for their activity and utilize ADP-ribose in their reactions.
Cell signaling processes:
Additionally, NAD+ is involved in cell signaling processes both within and outside of cells. NAD+ exists in distinct pools (cytosolic, mitochondrial, nuclear), with each being important for cell signaling. Its levels fluctuate with diet, exercise, and circadian rhythms, acting as a metabolic sensor.[ref]
Synthesizing NAD+ in the body:
Your body makes NAD+ in two ways: the de novo pathway (i.e. from scratch) or the salvage pathway.
De novo synthesis of NAD+
Precursors of NAD+ include different forms of niacin (vitamin B3) and tryptophan, which can be used to synthesize niacin. Forms of niacin, whether from food or supplements, include:
- nicotinamide (also called niacinamide)
- nicotinic acid, which is also called niacin, and causes flushing in larger amounts
Tryptophan is an essential amino acid. Your body can also convert tryptophan into niacin through the kynurenine pathway. However, this pathway to form niacin isn’t usually enough to meet the body’s needs, and thus, you also need to get niacin from food. What happens if you don’t get enough niacin from food? A lack of niacin causes a disease state known as pellagra. Symptoms of pellagra include dementia, diarrhea, and a skin rash.
In addition to using niacin from tryptophan, another way cells create more NAD+ is by converting nicotinic acid. The first step in converting nicotinic acid to NA mononucleotide (NAM) uses the NAPRT enzyme coded for by the NAPRT gene.[ref][ref]
NR (Nicotinamide Riboside) and NMN (Nicotinamide Mononucleotide):
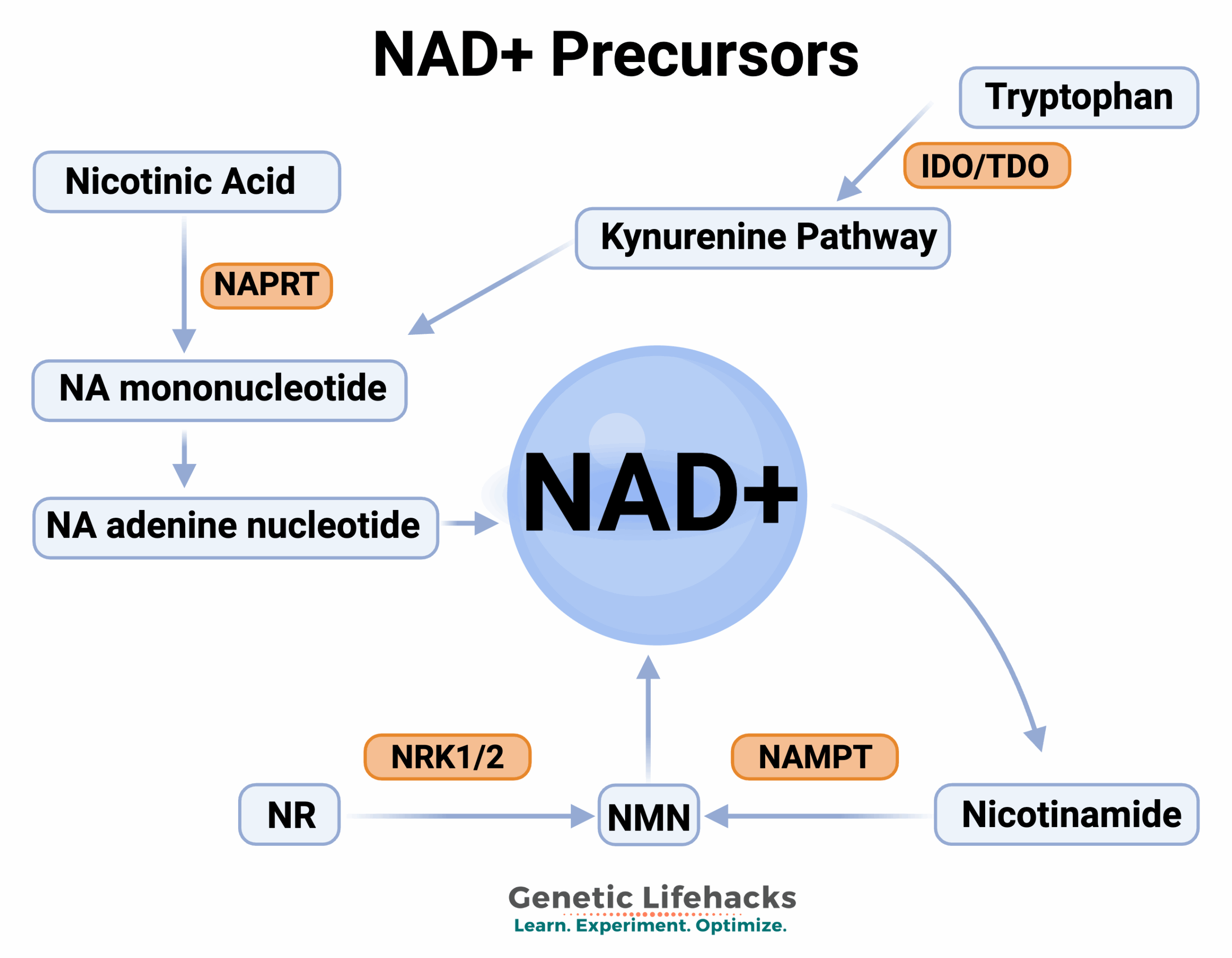
Both nicotinamide riboside (NR) and nicotinamide mononucleotide (NMN) are essential in creating and recycling NAD+. Plus, both are available as supplements. Let’s look at how they come into play in the NAD+ pathway – and why supplemental NR or NMN may be important in aging.
NMN, synthesized from nicotinamide (niacinamide) and PRPP (5’-phosphoribosyl-pyrophosphate), uses the enzyme NAMPT.[ref] (See NAMPT in the Genotype report.)
Nicotinamide riboside (NR) is another precursor of NAD+ and an intermediate in the NAD salvage pathway. It can be found at low levels in foods, particularly in milk, and it is available as a supplement.
NAD Salvage Pathway
NAD+ doesn’t have to be synthesized continually from the precursors — it can be recycled through the NAD Salvage Pathways. Reusing the components of NAD+, specifically nicotinamide, is your body’s main way of having enough NAD+ available in all cells. This salvage pathway is how NAD+ is synthesized from supplemental NR and NMN.
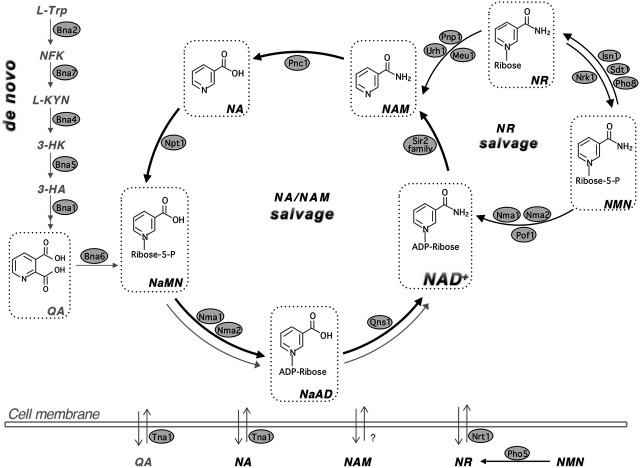
from PMC8171187, CC 4.0
This salvage process hinges on a key enzyme, NAMPT (Nicotinamide phosphoribosyltransferase), which converts nicotinamide to NMN.
NAMPT is the rate-limiting enzyme in the synthesis of NAD+ through the conversion of nicotinamide to NMN. It exists in intracellular (iNAMPT) and extracellular (eNAMPT, also known as visfatin) forms. The intracellular version drives NAD+ production. Note that NAMPT is often overexpressed in tumors and helps to drive cancer growth.[ref]
Balancing levels through excretion:
Levels of NAD+ are carefully controlled in the body. Excess nicotinamide that isn’t recycled in the salvage pathway is broken down and eventually excreted. The NNMT enzyme uses a methyl group to metabolize excess nicotinamide into metabolites that are eventually excreted in the urine. Excess levels of these metabolites, called 4py and 2py, are linked to a possible increase in the risk of atherosclerosis, pointing to excess niacin or nicotinamide not always being benign.[ref] It’s also important to note here that excess niacin or nicotinamide reduces the availability of methyl groups, which could be a problem for someone with methylation cycle genetic constraints.
Related article: Niacin and atherosclerosis
The decline of NAD+ in aging:
Let’s switch gears here and look at how and why NAD+ is important in the big picture of long-term health. Simply put, NAD+ levels decline with age, which is thought to be at the heart of many age-related diseases (e.g. heart disease, neurodegenerative diseases).[ref]
First, keep in mind that NAD+ is important in DNA repair, which prevents cell death or cancer caused by mutations that aren’t repaired.[ref] Mitochondrial energy decrease in aging is another big part of why everything goes downhill. The availability of NAD+ is key to mitochondrial energy production. Decreased NAD+ levels cause impaired neuronal plasticity (cognitive decline), poor cellular stress response, and increased cellular senescence.
Let’s take a look at some of the causes of NAD+ declines in aging:
| Enzyme/protein | Main role | How it uses NAD+ | Aging / disease relevance |
|---|---|---|---|
| PARP1 | DNA damage sensing and repair | Transfers ADP‑ribose units from NAD+ to proteins and DNA, consuming large amounts of NAD+. | Chronically activated by accumulated DNA damage, contributing to NAD+ depletion, impaired ATP production, and cell death in cardiovascular and neurodegenerative disease. |
| CD38 | Immune signaling, calcium regulation | Hydrolyzes NAD+ to generate signaling metabolites such as cyclic ADP‑ribose. | Upregulated by inflammation and senescence, driving age‑related NAD+ decline and inhibiting sirtuin activity. |
| CD157 (BST1) | Immune and neuroimmune signaling | Similar NAD‑glycohydrolase and base‑exchange activities, consuming NAD+. | Variants associate with Parkinson’s and autism; NR supplementation can rescue oxytocin‑linked behavioral phenotypes in CD157‑deficient mice. |
| Sirtuins (SIRT1–7) | Epigenetic regulation, metabolism, stress resistance | Use NAD+ as a co‑substrate for deacetylation and related reactions. | Reduced sirtuin activity with low NAD+ contributes to insulin resistance, fatty liver, telomere dysfunction, and accelerated aging phenotypes. |
How CD38 and inflammation result in NAD+ depletion in aging:
CD38 is a multifunctional enzyme that is key to the decline of NAD+ in aging by depleting levels. Increased inflammation causes increased CD38, which uses up more NAD+, causing a parallel decline in NAD+ levels. Increased cellular senescence in aging also causes increased CD38, with a subsequent decrease in NAD+.[ref]
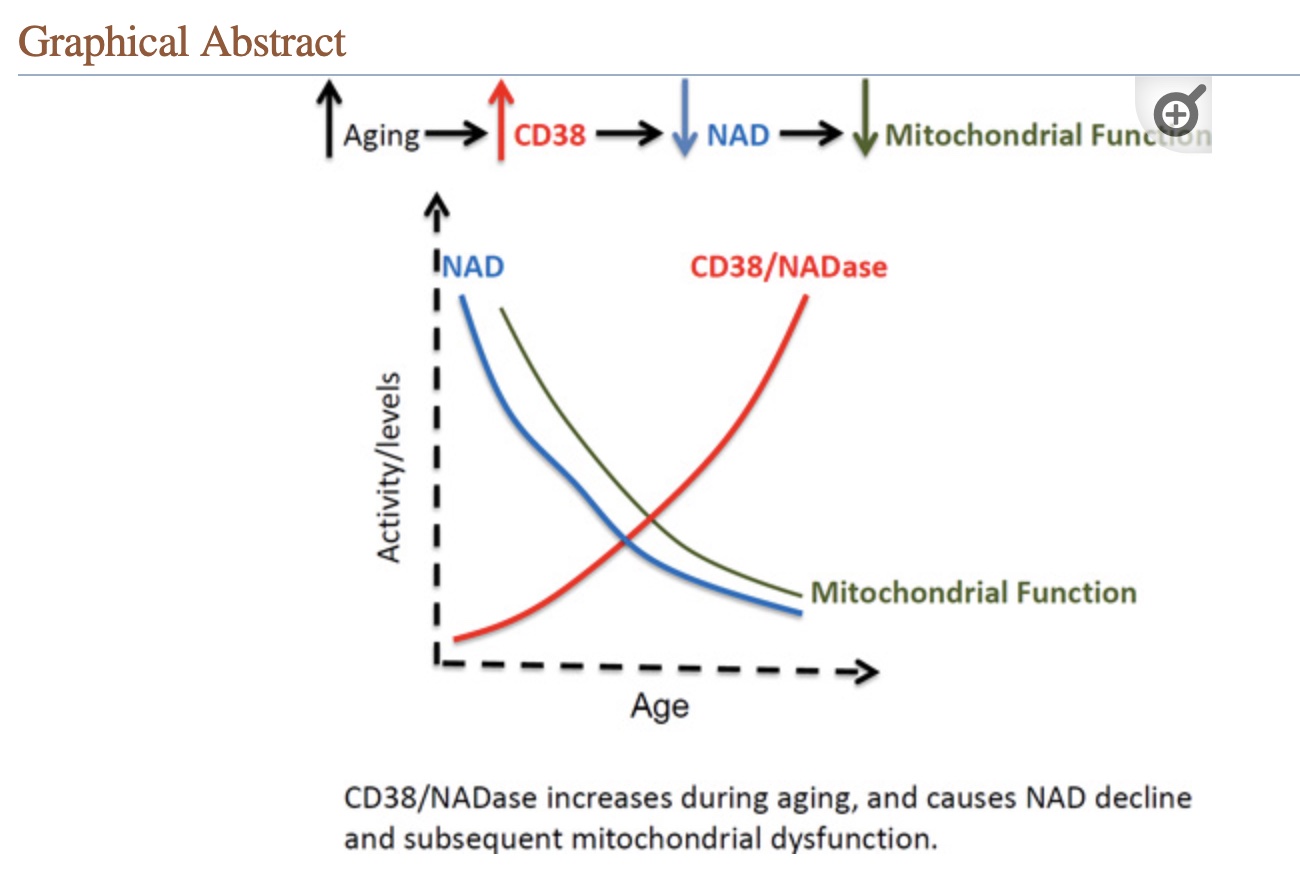
T cells, immune cells, endothelial cells, and smooth muscle cells all can produce CD38, and it is upregulated by proinflammatory cytokines, cellular senescence, steroid hormones, and in response to bacterial infections.[ref]
CD38 does a lot, including:[ref]
- regulating calcium ions and neurotransmitter release
- activation of T cells
- inhibits sirtuins both by reducing NAD levels and by generating a niacin metabolite that feeds back to inhibit SIRTs
- regulates the resolution of inflammatory response and tolerance of self (role in autoimmune diseases)
- limits NAD availability in bacterial infections (bacteria can use NAD for energy)
Nicotinamide Riboside, NAD+ pathway, and Covid: Why Covid hits older adults hard
NAD+ levels interact with the immune response in Covid patients in multiple ways:
- When someone is ill with COVID-19, the PARP genes are overexpressed, and NAD levels decrease.
- Researchers find that in COVID-19, PARPs are increased, the NAD+ salvage pathway is activated, and NAD biosynthesis (e.g., niacin creation) is decreased. [ref] [ref]
- CD38 levels are broadly upregulated in Covid patients, which further depletes NAD+ levels.[ref]
Sirtuins, NAD+, and Aging:
Let’s take a look at how sirtuins interact with NAD+ and aging in more detail.
Sirtuins are a family of genes (SIRT1 through SIRT7) involved in regulating gene expression. Sirtuins cause the DNA in the cell nucleus to be either accessible or inaccessible for a gene to be transcribed. The ability for the regulation of genes to be transcribed into proteins is fundamental to cell function. Every cell contains the same DNA in the nucleus. The differences between a liver cell and a muscle cell are due to the regulation of which genes are transcribed. Thus, disrupting the sirtuins can lead to mucked-up cell function and the symptoms of aging.
Lifespan extension studies:
Initial studies on the sirtuin genes in yeast showed that adding additional copies of the SIRT gene increased lifespan by 30%. This discovery kicked off decades of research showing how important sirtuins are in human healthspan.[ref]
Functions of the SIRTs:
- SIRT1 encodes the sirtuin 1 protein. It involves sensing nutrient availability and is thus linked to problems with insulin resistance. Studies show that animals with insulin resistance have decreased SIRT1 levels. When researchers increase SIRT1 in animals, they are resistant to the problems of obesity and insulin resistance that a high-fat diet induces in them.[ref][ref][ref]
- SIRT2 codes for the sirtuin 2 protein, which arranges the chromosomes for cell division in mitosis.
- SIRT3, 4, and 5, found in the mitochondria, are important for oxidative stress and fat metabolism.[ref]
- SIRT6 is important in gene expression for metabolic regulation, telomere maintenance, and mitochondrial respiration. Reducing Sirt6 in the liver causes animals to develop fatty liver disease, and knocking out Sirt6 altogether causes animals to die within a few weeks due to severely accelerated aging.[ref]
Sirtuins use NAD+ to complete their cellular activity, and through that, the NAD+ levels may be a sensor for how much energy is available in an organism.[ref] (More on the SIRTs in the Genotype section below)
PARPs and NAD+ in aging:
Another family of enzymes that utilizes NAD+ in its reactions is the PARPs, which stand for poly(ADP-ribose) polymerase. PARPs are important in DNA repair and genomic stability. They detect broken DNA and signal for it to be repaired. Additionally, when DNA isn’t able to be repaired, cell death is initiated. Again, these are vital cellular functions, especially in aging.[ref]
PARP1 uses up a lot of NAD+ in the process, causing a decrease in ATP production for the cell. When a cell hasn’t replicated the DNA properly, the DNA damage signaling response is enacted.[ref] Cell death is necessary in the right context, but excessive cell death, especially in the brain, is not good.
Excessive DNA breakage can lead to a lot of PARP activation, thus depleting NAD+.
What causes DNA breakage? Reactive oxygen species (oxidative stress), lipid peroxidation, radiation, and a lot of different environmental toxicants. DNA damage occurs all the time in the normal course of cell replication, but in aging, oxidative stress causes an increase in PPAR1 and a subsequent decrease in NAD+. PARP1 can initiate cellular repair for single-strand DNA breaks. This is important for longevity.
Inhibiting PARP is a way to mitigate the decreased NAD+ and ATP levels and decrease cell death. It doesn’t fix the cause (DNA breakage), but it puts a band-aid on the downstream effects of PARP activation. Atherosclerosis and congestive heart failure are two diseases in which PARP inhibitors might be used. The inflammation within the vascular cells causes PARP1 activation and the subsequent decrease in NAD+ and cellular energy. Inhibiting PARP then slows the inflammatory response and preserves the ATP and NAD+ in the heart cells.[ref][ref]
So if decreased NAD+ causes much of the pathophysiology found in aging, what can we do about it?
Research studies on supplemental NR and NMN:
Nicotinamide riboside (NR) and nicotinamide mononucleotide (NMN), two components of the NAD+ salvage pathway, are readily available as supplements and used to increase NAD+ levels.
Clinical trials (in humans) using NR and NMN:
Decreased Inflammation:
A study of ‘aged men’ looked at the effects of supplementing with 1,000 mg of NR per day for three weeks. The results showed an elevation of NAD+ in the muscles and a decrease in inflammatory cytokine levels.[ref]
Heart health:
A study that included 30 middle-aged and older men and women looked at the effect of NR vs placebo for six weeks. Oral NR supplementation (1,000mg/day ) raised NAD+ levels by 60% compared to placebo. NR lowered blood pressure and aortic stiffness (a little). Notably, participants who had stage one hypertension to begin with had a 10-point drop in systolic blood pressure.[ref]
A small clinical trial in patients with heart failure showed that oral nicotinamide riboside decreased proinflammatory cytokines. The researchers concluded that NR may improve mitochondrial respiration and attenuate inflammation in heart failure.[ref]
Brain health:
A study of NR supplementation (500 mg, 2x /day, 6 weeks) in older adults showed increased NAD+ levels in neuronal vesicles and decreased neuroinflammatory markers.[ref]
Reduced inflammation in COPD:
A placebo-controlled clinical trial using NR (1g / twice a day) in COPD patients showed reduced inflammatory markers in the lung sputum within 6 weeks.[ref]
Skeletal muscle metabolism:
A recent study found: “NR supplementation of 1000 mg/d for 6 wk in healthy overweight or obese men and women increased skeletal muscle NAD+ metabolites, affected skeletal muscle acetylcarnitine metabolism, and induced minor changes in body composition and sleeping metabolic rate.”[ref]
Fighting off infections:
A clinical trial found that people recovered more quickly from COVID-19 when given supplemental NR, N-acetyl-cysteine, and l-carnitine. The phase II and phase III clinical trials found that recovery was 3-4 days faster with the supplement.[ref]
Part of the Alzheimer’s stack:
An exciting phase II clinical trial using a combination of natural supplements showed improved cognitive function in people diagnosed with Alzheimer’s disease. The combination included 1 g of nicotinamide riboside, 2.5 g NAC, 3.7 g l-carnitine, and 12g of l-serine.[ref]
Insulin secretion regulation:
A small study using 250 mg of NMN daily, before breakfast, found that it helped to attenuate insulin secretion after eating sugar.[ref]
Premature aging prevention:
A study in people with Werner syndrome, a genetic disease that causes premature aging, showed that nicotinamide riboside could prevent some of the premature aging, including helping with kidney function.[ref]
Sleep quality in older adults:
A 2024 clinical trial showed that 250 mg/day of NMN improved sleep quality in older adults after 12 weeks.[ref]
Animal studies on NR and NMN:
A number of animal studies show the efficacy of raising NAD+ levels with NR or NMN. These types of studies are important for understanding the mechanism through which increasing NAD+ levels works.
- Reverses Alzheimer’s in animals: In a mouse model of Alzheimer’s disease, NMN shows the restoration of mitochondrial function in the brain. The oxygen consumption deficits in the brain mitochondria, found in Alzheimer’s, showed a reversal.[ref][ref] In addition, inhibiting NAMPT using a compound called P7C3-A20 also reverses advanced Alzheimer’s in animal models.[ref]
- Restores Fertility: Several studies have shown that NMN or NAD+ precursors restore fertility at the end of an animal’s normal reproductive age. It seems to do this through rejuvenating egg quality.[ref][ref]
- Counteracts hemorrhagic shock: In a rodent model of hemorrhagic shock, those receiving NMN had less inflammation, better cellular metabolism, and increased survival in hemorrhagic shock.[ref]
- Decreases signs of aging: Nicotinamide riboside (NR) was fed to old mice for three months. The NR decreased several of the signs of aging in mice, such as altered fat mass, cholesterol levels, and liver enzymes. [ref]
- Reverses fatty liver disease: Quite a few studies show that NR can reverse fatty liver disease.[ref][ref][ref]
- Improves cognitive function: Another mouse study showed that NR could improve cognitive function in a mouse and reduce inflammatory markers in the brain.[ref]
- Protects against hearing loss: NR was shown to protect mice from age-related noise-induced hearing loss by increasing SIRT3 expression.[ref]
- Prevents retinal degeneration: NR helps to prevent retinal degeneration and inflammation in the retina. [ref]
- Improves mitochondrial function: A mouse study also found that NMN could dampen the DNA damage response and improve mitochondrial function.[ref]
- Increased lifespan: A small increase in lifespan (about 4%) has been shown in mice fed NR starting at old age.[ref]
- Restored SIRT1 levels: Middle-aged mice fed NMN showed increased Sirt1 levels, similar to younger mice.[ref]
CD157 and oxytocin:
Interestingly, researchers have found in animal studies that CD157 is important in oxytocin levels in the brain. The study was specifically looking at how low oxytocin relates to autism spectrum behaviors. Giving NR to mice that lack CD157/BST1 corrected the behavioral deficits in the animals.[ref] A recent study in children with autism found that nicotinamide riboside levels (along with 7 other metabolites) are lower in autism spectrum disorder compared to healthy controls.[ref]
Safety and side effects of NR and NMN supplements:
The first question is always – is it safe? So far, studies show that NR and NMN are safe for most people. Keep in mind that these studies last months to years, so the long-term effects of taking NR or NMN are based just on animal studies. Talk with your doctor if you have questions about supplements.
- A study looked at the safety of nicotinamide riboside (TruNiagen brand) taken by healthy men and women for 8 weeks, in doses ranging from 100 to 1000 mg. All doses increased NAD+ metabolites within two weeks, which was dose-dependent. Most importantly, there were no differences in adverse events between the NR groups and the placebo group. [ref]
- A 6-month randomized, double-blind, placebo-controlled clinical trial with NR plus pterostilbene found that it was safe and well-tolerated. While it didn’t cure NAFLD (fatty liver) in six months, there was a significant improvement in liver enzymes, ALT and GGT, in the NR group compared to placebo. [ref]
- Another trial found that 2,000 mg/day of NR in obese, sedentary men aged 40 – 70 was safe and well-tolerated in a 12-week study. [ref] Note that 2,000 mg/day is a higher dose than most supplements, which usually are in the 100-300 mg range.
- In a safety trial for people with Parkinson’s disease, the patients took 1,500 mg of nicotinamide riboside daily. “NR therapy was well tolerated with no moderate or severe adverse events, and no significant difference in mild adverse events.”[ref]
Does NR or NMN promote atherosclerotic plaque?
A 2024 study made headlines with evidence that niacin increases atherosclerotic plaque for some people with specific genetic variants. The study focused more on dietary niacin than other sources; however, a 2025 study in mice also showed that higher doses of NR increased atherosclerotic plaque through the same pathway.[ref]
While more studies are needed, I suggest reading through the article on Niacin and Atherosclerosis and checking your genetic variants.
Does NR or NMN mess up methylation?
Some health gurus on the internet have theorized that taking NR or NMN will mess up your methylation cycle. While it makes sense in theory, this study in adults supplementing with nicotinamide riboside noted that NR did not mess up methylation.[ref]
Does NR or NMN actually boost NAD+?
Several clinical trials show that both supplements will increase NAD+ levels.
- A clinical trial examined the effects of NR on healthy volunteers for 9 days. The study participants took 250 mg for the first two days and then were titrated up to 1000 mg. On day 9, NAD+ levels had increased by 100%. No side effects were reported for the NR supplement. Interestingly, most of the individual response curves were similar in percentage increase, but a couple of participants had a much bigger response.[ref]
- Another clinical trial looked at NMN to see if it increased NAD+ levels. The results showed that 300, 600, and 900 mg doses significantly increased NAD+ levels. It was safe and well tolerated; subjective health improved statistically in the group taking NMN compared to the placebo group.[ref]
Now let’s switch gears and look at how your genetic variants may influence your personal need for NAD+.
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
HPA Axis Dysfunction: Understanding Cortisol and Genetic Interactions
References:
Airhart, Sophia E., et al. “An Open-Label, Non-Randomized Study of the Pharmacokinetics of the Nutritional Supplement Nicotinamide Riboside (NR) and Its Effects on Blood NAD+ Levels in Healthy Volunteers.” PLoS ONE, vol. 12, no. 12, Dec. 2017, p. e0186459. PubMed Central, https://doi.org/10.1371/journal.pone.0186459.
Altay, Ozlem, et al. “Combined Metabolic Activators Accelerates Recovery in Mild-to-Moderate COVID-19.” Advanced Science (Weinheim, Baden-Wurttemberg, Germany), vol. 8, no. 17, Sept. 2021, p. e2101222. PubMed, https://doi.org/10.1002/advs.202101222.
Amano, Hisayuki, et al. “Telomere Dysfunction Induces Sirtuin Repression That Drives Telomere-Dependent Disease.” Cell Metabolism, vol. 29, no. 6, June 2019, pp. 1274-1290.e9. PubMed, https://doi.org/10.1016/j.cmet.2019.03.001.
Bertoldo, Michael J., et al. “NAD+ Repletion Rescues Female Fertility during Reproductive Aging.” Cell Reports, vol. 30, no. 6, Feb. 2020, pp. 1670-1681.e7. www.cell.com, https://doi.org/10.1016/j.celrep.2020.01.058.
Brown, Kevin D., et al. “Activation of SIRT3 by the NAD⁺ Precursor Nicotinamide Riboside Protects from Noise-Induced Hearing Loss.” Cell Metabolism, vol. 20, no. 6, Dec. 2014, pp. 1059–68. PubMed, https://doi.org/10.1016/j.cmet.2014.11.003.
Cantó, Carles, et al. “NAD+ Metabolism and the Control of Energy Homeostasis – a Balancing Act between Mitochondria and the Nucleus.” Cell Metabolism, vol. 22, no. 1, July 2015, pp. 31–53. PubMed Central, https://doi.org/10.1016/j.cmet.2015.05.023.
Chaubey, Kalyani, et al. “Pharmacologic Reversal of Advanced Alzheimer’s Disease in Mice and Identification of Potential Therapeutic Nodes in Human Brain.” Cell Reports Medicine, Dec. 2025, p. 102535. DOI.org (Crossref), https://doi.org/10.1016/j.xcrm.2025.102535.
Clement, James, et al. “The Plasma NAD+ Metabolome Is Dysregulated in ‘Normal’ Aging.” Rejuvenation Research, vol. 22, no. 2, Apr. 2019, pp. 121–30. liebertpub.com (Atypon), https://doi.org/10.1089/rej.2018.2077.
Conze, Dietrich, et al. “Safety and Metabolism of Long-Term Administration of NIAGEN (Nicotinamide Riboside Chloride) in a Randomized, Double-Blind, Placebo-Controlled Clinical Trial of Healthy Overweight Adults.” Scientific Reports, vol. 9, no. 1, July 2019, p. 9772. PubMed, https://doi.org/10.1038/s41598-019-46120-z.
Dollerup, Ole L., et al. “A Randomized Placebo-Controlled Clinical Trial of Nicotinamide Riboside in Obese Men: Safety, Insulin-Sensitivity, and Lipid-Mobilizing Effects.” The American Journal of Clinical Nutrition, vol. 108, no. 2, Aug. 2018, pp. 343–53. PubMed, https://doi.org/10.1093/ajcn/nqy132.
Duarte-Pereira, Sara, et al. “NAMPT and NAPRT1: Novel Polymorphisms and Distribution of Variants between Normal Tissues and Tumor Samples.” Scientific Reports, vol. 4, Sept. 2014, p. 6311. PubMed Central, https://doi.org/10.1038/srep06311.
Ear, Po Hien, et al. “Maternal Nicotinamide Riboside Enhances Postpartum Weight Loss, Juvenile Offspring Development, and Neurogenesis of Adult Offspring.” Cell Reports, vol. 26, no. 4, Jan. 2019, pp. 969-983.e4. ScienceDirect, https://doi.org/10.1016/j.celrep.2019.01.007.
Elhassan, Yasir S., et al. “Nicotinamide Riboside Augments the Aged Human Skeletal Muscle NAD+ Metabolome and Induces Transcriptomic and Anti-Inflammatory Signatures.” Cell Reports, vol. 28, no. 7, Aug. 2019, pp. 1717-1728.e6. PubMed, https://doi.org/10.1016/j.celrep.2019.07.043.
Gariani, Karim, et al. “Eliciting the Mitochondrial Unfolded Protein Response by Nicotinamide Adenine Dinucleotide Repletion Reverses Fatty Liver Disease in Mice.” Hepatology (Baltimore, Md.), vol. 63, no. 4, Apr. 2016, pp. 1190–204. PubMed Central, https://doi.org/10.1002/hep.28245.
Garten, Antje, et al. “Physiological and Pathophysiological Roles of NAMPT and NAD Metabolism.” Nature Reviews Endocrinology, vol. 11, no. 9, Sept. 2015, pp. 535–46. www.nature.com, https://doi.org/10.1038/nrendo.2015.117.
Gerasimenko, Maria, et al. “Nicotinamide Riboside Supplementation Corrects Deficits in Oxytocin, Sociability and Anxiety of CD157 Mutants in a Mouse Model of Autism Spectrum Disorder.” Scientific Reports, vol. 10, 2020. www.ncbi.nlm.nih.gov, https://doi.org/10.1038/s41598-019-57236-7.
Guan, Yi, et al. “Nicotinamide Mononucleotide, an NAD+ Precursor, Rescues Age-Associated Susceptibility to AKI in a Sirtuin 1–Dependent Manner.” Journal of the American Society of Nephrology, vol. 28, no. 8, Aug. 2017, pp. 2337–52. jasn.asnjournals.org, https://doi.org/10.1681/ASN.2016040385.
Han, Xue, et al. “Nicotinamide Riboside Exerts Protective Effect against Aging-Induced NAFLD-like Hepatic Dysfunction in Mice.” PeerJ, vol. 7, 2019, p. e7568. PubMed, https://doi.org/10.7717/peerj.7568.
Henning, Robert J., et al. “Poly(ADP-Ribose) Polymerase (PARP) and PARP Inhibitors: Mechanisms of Action and Role in Cardiovascular Disorders.” Cardiovascular Toxicology, vol. 18, no. 6, Dec. 2018, pp. 493–506. PubMed, https://doi.org/10.1007/s12012-018-9462-2.
Hong, Guangliang, et al. “Administration of Nicotinamide Riboside Prevents Oxidative Stress and Organ Injury in Sepsis.” Free Radical Biology & Medicine, vol. 123, Aug. 2018, pp. 125–37. PubMed, https://doi.org/10.1016/j.freeradbiomed.2018.05.073.
Kane, Alice E., and David A. Sinclair. “Sirtuins and NAD+ in the Development and Treatment of Metabolic and Cardiovascular Diseases.” Circulation Research, vol. 123, no. 7, Sept. 2018, pp. 868–85. PubMed Central, https://doi.org/10.1161/CIRCRESAHA.118.312498.
Kang, Dae-Wook, et al. “Distinct Fecal and Plasma Metabolites in Children with Autism Spectrum Disorders and Their Modulation after Microbiota Transfer Therapy.” MSphere, vol. 5, no. 5, Oct. 2020, pp. e00314-20. PubMed, https://doi.org/10.1128/mSphere.00314-20.
Lee, Hee Jae, and Soo Jin Yang. “Supplementation with Nicotinamide Riboside Reduces Brain Inflammation and Improves Cognitive Function in Diabetic Mice.” International Journal of Molecular Sciences, vol. 20, no. 17, Aug. 2019, p. E4196. PubMed, https://doi.org/10.3390/ijms20174196.
Long, Aaron N., et al. “Effect of Nicotinamide Mononucleotide on Brain Mitochondrial Respiratory Deficits in an Alzheimer’s Disease-Relevant Murine Model.” BMC Neurology, vol. 15, Mar. 2015, p. 19. PubMed Central, https://doi.org/10.1186/s12883-015-0272-x.
Lopatina, Olga L., et al. “CD157 and Brain Immune System in (Patho)Physiological Conditions: Focus on Brain Plasticity.” Frontiers in Immunology, vol. 11, 2020. www.ncbi.nlm.nih.gov, https://doi.org/10.3389/fimmu.2020.585294.
Martens, Christopher R., et al. “Chronic Nicotinamide Riboside Supplementation Is Well-Tolerated and Elevates NAD+ in Healthy Middle-Aged and Older Adults.” Nature Communications, vol. 9, no. 1, Mar. 2018, p. 1286. www.nature.com, https://doi.org/10.1038/s41467-018-03421-7.
Masri, Selma, et al. “Partitioning Circadian Transcription by SIRT6 Leads to Segregated Control of Cellular Metabolism.” Cell, vol. 158, no. 3, July 2014, pp. 659–72. PubMed Central, https://doi.org/10.1016/j.cell.2014.06.050.
Murata, Michael M., et al. “NAD+ Consumption by PARP1 in Response to DNA Damage Triggers Metabolic Shift Critical for Damaged Cell Survival.” Molecular Biology of the Cell, vol. 30, no. 20, Sept. 2019, pp. 2584–97. PubMed, https://doi.org/10.1091/mbc.E18-10-0650.
Nevoral, Jan, et al. “Epigenetic and Non-Epigenetic Mode of SIRT1 Action during Oocyte Meiosis Progression.” Journal of Animal Science and Biotechnology, vol. 10, Aug. 2019, p. 67. PubMed Central, https://doi.org/10.1186/s40104-019-0372-3.
Pfluger, Paul T., et al. “Sirt1 Protects against High-Fat Diet-Induced Metabolic Damage.” Proceedings of the National Academy of Sciences, vol. 105, no. 28, July 2008, pp. 9793–98. www.pnas.org, https://doi.org/10.1073/pnas.0802917105.
Remie, Carlijn M. E., et al. “Nicotinamide Riboside Supplementation Alters Body Composition and Skeletal Muscle Acetylcarnitine Concentrations in Healthy Obese Humans.” The American Journal of Clinical Nutrition, vol. 112, no. 2, Aug. 2020, pp. 413–26. PubMed, https://doi.org/10.1093/ajcn/nqaa072.
Salic, Kanita, et al. “Combined Treatment with L-Carnitine and Nicotinamide Riboside Improves Hepatic Metabolism and Attenuates Obesity and Liver Steatosis.” International Journal of Molecular Sciences, vol. 20, no. 18, Sept. 2019, p. E4359. PubMed, https://doi.org/10.3390/ijms20184359.
Sims, Carrie A., et al. “Nicotinamide Mononucleotide Preserves Mitochondrial Function and Increases Survival in Hemorrhagic Shock.” JCI Insight, vol. 3, no. 17, p. e120182. PubMed Central, https://doi.org/10.1172/jci.insight.120182. Accessed 15 Dec. 2021.
Sun, Cheng, et al. “SIRT1 Improves Insulin Sensitivity under Insulin-Resistant Conditions by Repressing PTP1B.” Cell Metabolism, vol. 6, no. 4, Oct. 2007, pp. 307–19. www.cell.com, https://doi.org/10.1016/j.cmet.2007.08.014.
Yaku, Keisuke, et al. “BST1 Regulates Nicotinamide Riboside Metabolism via Its Glycohydrolase and Base-Exchange Activities.” Nature Communications, vol. 12, 2021. www.ncbi.nlm.nih.gov, https://doi.org/10.1038/s41467-021-27080-3.
Zhang, Hongbo, et al. “NAD+ Repletion Improves Mitochondrial and Stem Cell Function and Enhances Life Span in Mice.” Science, vol. 352, no. 6292, June 2016, pp. 1436–43. science.org (Atypon), https://doi.org/10.1126/science.aaf2693.
Zhang, Xian, et al. “Systemic Treatment With Nicotinamide Riboside Is Protective in a Mouse Model of Light-Induced Retinal Degeneration.” Investigative Ophthalmology & Visual Science, vol. 61, no. 10, Aug. 2020. www.ncbi.nlm.nih.gov, https://doi.org/10.1167/iovs.61.10.47.
Zhou, Bo, et al. “Boosting NAD Level Suppresses Inflammatory Activation of PBMCs in Heart Failure.” The Journal of Clinical Investigation, vol. 130, no. 11, Nov. 2020, pp. 6054–63. PubMed, https://doi.org/10.1172/JCI138538.
https://academic.oup.com/edrv/article/31/2/194/2354747#59027139. Accessed 15 Dec. 2021.
https://www.genecards.org/cgi-bin/carddisp.pl?gene=NAPRT. Accessed 15 Dec. 2021.

