Have you ever wondered, though, how your cells can survive for a few minutes without oxygen — or how your body manages when oxygen levels are lower than normal? It turns out that we have an innate system that detects when oxygen levels are low and turns on other genes that can help cells survive when precious O2 is not readily available.
The hypoxia-inducible factor-1 alpha (HIF1A) gene codes for a transcription factor, which regulates the rate at which certain other genes get transcribed.
Genes code for proteins; your cells need to know which genes need to be translated into the proteins needed at that instance. Basically, a transcription factor is like a switch that can turn on or off the transcription of genes. There are a couple of thousand different transcription factors in the human genome.
HIF-1a (hypoxia-inducible factor-1 alpha) responds to the amount of oxygen available to the cell. Hypoxia refers to a state where there isn’t enough oxygen available.
Thus, when there isn’t enough oxygen available at a cellular level, the HIF-1a transcription factor can change the transcription rate for the genes that code for proteins involved in oxygen and glucose transport. It switches on the genes needed for resolving the problems of low oxygen.
When oxygen levels are low (hypoxia), cells and tissues need to respond quickly. The use of oxygen normally occurs in the cellular process to make energy or ATP. (Yes, your cells have a backup route of anaerobic glycolysis, which works without oxygen, but this isn’t as efficient for making ATP.)
Thus, when oxygen levels drop, HIF-1a levels rise, kicking off a bunch of processes.
One way that HIF-1a helps your body respond to the lack of oxygen is by stimulating the growth of more blood vessels. It also stimulates the production of red blood cells by increasing erythropoietin (EPO) expression.
The first thing that comes to mind with low oxygen levels may be high altitudes. As you go up a mountain in Colorado – or in the Himalayas – oxygen levels decrease. Anyone who has flown out to Colorado to ski at Breckenridge (or worse, A-basin) knows how bad it feels if you don’t take time to adjust slowly to the altitude. (Spend that first night in Denver. Seriously…)
Another way that oxygen levels can drop in your body is during aerobic exercise. You know… like when it is New Year’s Day, and you decided to join the gym and run on the treadmill — for the first time in a year. Your muscles are suddenly using up oxygen faster than you can suck it in, leaving you gasping and feeling like you’re going to die.
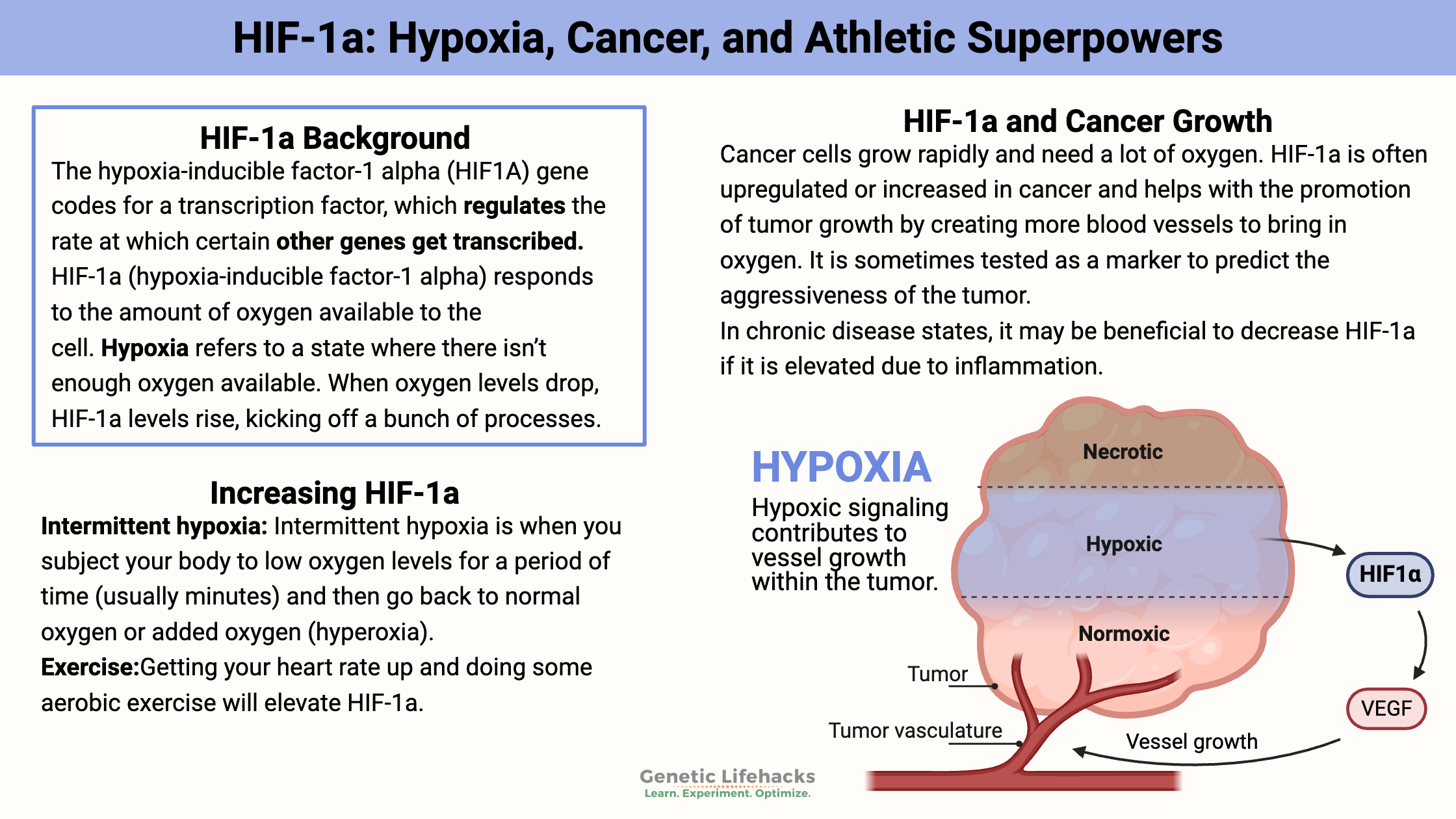
Cancer cells grow rapidly and need a lot of oxygen. Thus, they need more blood vessels to bring in the O2, and they need nutrients.
HIF-1a is very often upregulated or increased in cancer and helps promote tumor growth by creating more blood vessels to bring in oxygen. It is sometimes tested as a marker to predict the aggressiveness of the tumor.
Under normal oxygen levels, known as normoxia, cells produce HIF1a and then quickly degrade it using enzymes called prolyl hydroxylases (PHDs). These PHDs are oxygen sensors, altering HIF-1a and degrading it when there is plenty of oxygen present in the cell.[ref]
Under normal oxygen conditions (normoxia), HIF1A is rapidly degraded. However, in hypoxic conditions, the degradation is inhibited, leading to the accumulation of HIF1A in the cell. This protein then translocates to the nucleus, where it dimerizes with HIF1B and binds to hypoxia-responsive elements (HREs) in the DNA, activating the transcription of various target genes.
Researchers recently developed ways of inhibiting PHDs, thus allowing HIF-1A to be upregulated under normal oxygen conditions. These PHD (prolyl hydroxylase) inhibitors may be used for regenerating damaged tissue (increased blood vessel formation) or for treating specific types of anemia since increased HIF1A stimulates red blood cell production.
The flip side of encouraging HIF-1a under normoxia is that you really don’t want to encourage cancer growth. As a result, the use of PHD inhibitors only applies to specific conditions.
In addition to activation by hypoxia, the activation of HIF-1a also occurs from cytokines, growth factors, hormones, and cancer genes. For example, the growth factor IGF-1 can increase HIF1a. Estrogen can also increase HIF-1a, such as in the thickening of the endometrium each month for premenopausal women.[ref][ref][ref]
Interestingly, tamoxifen, a treatment for estrogen-positive breast cancer, works in part by reducing HIF-1a levels.[ref]
Cytokines are inflammatory molecules that the body produces as part of the immune response. They are signals to increase inflammation. Specifically, the cytokine TNF-alpha causes an increase in HIF-1a. [ref] This is one connection between inflammatory conditions, such as arthritis or atherosclerosis, and higher HIF-1a levels.[ref]
There are natural ways to both inhibit HIF1a and increase HIF1a. I’m listing both here – you can decide which way is best for your body right now.
Logically, if you are trying to increase HIF1a, you may not want to take an inhibitor of HIF1a at the same time. For example, if you are taking resveratrol (inhibitor) while also trying to increase HIF1a through breathwork, you may not reap as much benefit.
Intermittently raising HIF1a may have benefits for your body. Just like exercise causes stress on the body at the moment, the benefits afterward outweigh the short-term stress on the body. Similarly, intermittently raising HIF-1a levels could be of benefit in certain situations.
Intermittent hypoxia reduces fasting blood glucose levels and decreases LDL cholesterol in people with prediabetes. The study used middle-aged and older patients and subjected them to sessions of four cycles of 5 minutes of hypoxia followed by either normal oxygen or hyperoxia.[ref]
There is a saying for athletes, ‘live high, train low’, which uses the idea that changes in altitude and oxygen levels will affect performance. This is based on changes in HIF-1a, which causes an increase in EPO and red blood cells. Research does show that changing the oxygen content of the air – either through intermittent hypoxia or continuous hypoxia – can increase red blood cell production.[ref]
Timing and dose are vital with intermittent hypoxia therapy. Chronic intermittent hypoxia at night, or ‘sleep apnea’, has many negative consequences. In fact, sleep apnea is a risk factor for diabetes, obesity, and cardiovascular disease.[ref][ref] So, while a little intermittent hypoxia can be a good thing, chronic hypoxia every single night is bad.
So how can you do intermittent hypoxia therapy? There are athletic training facilities with expensive equipment to do it. And there are YouTube videos that have some odd DIY ideas. I’ll let you discover it on your own.
Ban, Hyun Seung, et al. “The Novel Hypoxia-Inducible Factor-1α Inhibitor IDF-11774 Regulates Cancer Metabolism, Thereby Suppressing Tumor Growth.” Cell Death & Disease, vol. 8, no. 6, June 2017, p. e2843. PubMed, https://doi.org/10.1038/cddis.2017.235.
Burián, Z., et al. “Selective Inhibition of HIF1α Expression by ZnSO4 Has Antitumoral Effects in Human Melanoma.” Pathology Oncology Research: POR, vol. 26, no. 2, Apr. 2020, pp. 673–79. PubMed, https://doi.org/10.1007/s12253-018-00573-1.
Cao, Zongxian, et al. “Trans-3,4,5’-Trihydroxystibene Inhibits Hypoxia-Inducible Factor 1alpha and Vascular Endothelial Growth Factor Expression in Human Ovarian Cancer Cells.” Clinical Cancer Research: An Official Journal of the American Association for Cancer Research, vol. 10, no. 15, Aug. 2004, pp. 5253–63. PubMed, https://doi.org/10.1158/1078-0432.CCR-03-0588.
Choudhury, Ryan. “Hypoxia and Hyperbaric Oxygen Therapy: A Review.” International Journal of General Medicine, vol. 11, Nov. 2018, pp. 431–42. PubMed Central, https://doi.org/10.2147/IJGM.S172460.
Chun, Y. S., et al. “Zinc Induces the Accumulation of Hypoxia-Inducible Factor (HIF)-1alpha, but Inhibits the Nuclear Translocation of HIF-1beta, Causing HIF-1 Inactivation.” Biochemical and Biophysical Research Communications, vol. 268, no. 2, Feb. 2000, pp. 652–56. PubMed, https://doi.org/10.1006/bbrc.2000.2180.
Cortes, Ernesto, et al. “Tamoxifen Mechanically Reprograms the Tumor Microenvironment via HIF‐1A and Reduces Cancer Cell Survival.” EMBO Reports, vol. 20, no. 1, Jan. 2019, p. e46557. PubMed Central, https://doi.org/10.15252/embr.201846557.
Dale, E. A., et al. “Unexpected Benefits of Intermittent Hypoxia: Enhanced Respiratory and Nonrespiratory Motor Function.” Physiology, vol. 29, no. 1, Jan. 2014, pp. 39–48. PubMed Central, https://doi.org/10.1152/physiol.00012.2013.
Doğan, Canan Sercan, et al. “Comparison of Athletic Performance of Turkish Ice Hockey Players with ACE I/D (Rs1799752), ACTN3 (Rs1815739), PPARA (Rs4253778) and HIF1A (Rs11549465) Polymorphisms.” Cellular and Molecular Biology (Noisy-Le-Grand, France), vol. 70, no. 10, Nov. 2024, pp. 142–53. PubMed, https://doi.org/10.14715/cmb/2024.70.10.19.
Fang, Jing, et al. “Apigenin Inhibits VEGF and HIF-1 Expression via PI3K/AKT/p70S6K1 and HDM2/P53 Pathways.” FASEB Journal: Official Publication of the Federation of American Societies for Experimental Biology, vol. 19, no. 3, Mar. 2005, pp. 342–53. PubMed, https://doi.org/10.1096/fj.04-2175com.
Fernández-Torres, Javier, et al. “Polymorphic Variation of Hypoxia Inducible Factor-1 A (HIF1A) Gene Might Contribute to the Development of Knee Osteoarthritis: A Pilot Study.” BMC Musculoskeletal Disorders, vol. 16, Aug. 2015, p. 218. PubMed, https://doi.org/10.1186/s12891-015-0678-z.
Ferreira, Marta, et al. “Hypoxia and Renal Cell Carcinoma: The Influence of HIF1A+1772C/T Functional Genetic Polymorphism on Prognosis.” Urologic Oncology, vol. 35, no. 8, Aug. 2017, p. 532.e25-532.e30. PubMed, https://doi.org/10.1016/j.urolonc.2017.04.002.
Fu, S. L., et al. “A Polymorphism in the 3’ Untranslated Region of Hypoxia-Inducible Factor-1 Alpha Confers an Increased Risk of Cervical Cancer in a Chinese Population.” Neoplasma, vol. 61, no. 1, 2014, pp. 63–69.
Gabbasov, Rashid T., et al. “The HIF1A Gene Pro582Ser Polymorphism in Russian Strength Athletes.” Journal of Strength and Conditioning Research, vol. 27, no. 8, Aug. 2013, pp. 2055–58. PubMed, https://doi.org/10.1519/JSC.0b013e31827f06ae.
Guo, Xu, et al. “SNP Rs2057482 in HIF1A Gene Predicts Clinical Outcome of Aggressive Hepatocellular Carcinoma Patients after Surgery.” Scientific Reports, vol. 5, June 2015, p. 11846. PubMed Central, https://doi.org/10.1038/srep11846.
Halliday, Matthew R., et al. “Intensive Treadmill Exercise Increases Expression of Hypoxia-Inducible Factor 1α and Its Downstream Transcript Targets: A Potential Role in Neuroplasticity.” Neuroreport, vol. 30, no. 9, June 2019, pp. 619–27. PubMed, https://doi.org/10.1097/WNR.0000000000001239.
Jain, Tanmay, et al. “Hypoxia Inducible Factor as a Therapeutic Target for Atherosclerosis.” Pharmacology & Therapeutics, vol. 183, Mar. 2018, pp. 22–33. PubMed, https://doi.org/10.1016/j.pharmthera.2017.09.003.
Kang, Min-Jung, et al. “Associations between Single Nucleotide Polymorphisms of MMP2, VEGF, and HIF1A Genes and the Risk of Developing Colorectal Cancer.” Anticancer Research, vol. 31, no. 2, Feb. 2011, pp. 575–84.
Kazi, Armina A., et al. “Estrogen Rapidly Activates the PI3K/AKT Pathway and Hypoxia-Inducible Factor 1 and Induces Vascular Endothelial Growth Factor A Expression in Luminal Epithelial Cells of the Rat Uterus.” Biology of Reproduction, vol. 81, no. 2, Aug. 2009, pp. 378–87. PubMed Central, https://doi.org/10.1095/biolreprod.109.076117.
Koistinen, P. O., et al. “EPO, Red Cells, and Serum Transferrin Receptor in Continuous and Intermittent Hypoxia.” Medicine and Science in Sports and Exercise, vol. 32, no. 4, Apr. 2000, pp. 800–04. PubMed, https://doi.org/10.1097/00005768-200004000-00012.
Konac, Ece, et al. “An Investigation of Relationships between Hypoxia-Inducible Factor-1 Alpha Gene Polymorphisms and Ovarian, Cervical and Endometrial Cancers.” Cancer Detection and Prevention, vol. 31, no. 2, 2007, pp. 102–09. PubMed, https://doi.org/10.1016/j.cdp.2007.01.001.
Kushwah, Neetu, et al. “Neuroprotective Role of Intermittent Hypobaric Hypoxia in Unpredictable Chronic Mild Stress Induced Depression in Rats.” PloS One, vol. 11, no. 2, 2016, p. e0149309. PubMed, https://doi.org/10.1371/journal.pone.0149309.
Larruskain, Jon, et al. “Genetic Variants and Hamstring Injury in Soccer: An Association and Validation Study.” Medicine and Science in Sports and Exercise, vol. 50, no. 2, Feb. 2018, pp. 361–68. PubMed, https://doi.org/10.1249/MSS.0000000000001434.
Lee, Chi Fung, et al. “Targeting NAD+ Metabolism as Interventions for Mitochondrial Disease.” Scientific Reports, vol. 9, Feb. 2019, p. 3073. PubMed Central, https://doi.org/10.1038/s41598-019-39419-4.
Li, Pu, et al. “Genetic Polymorphisms in HIF1A Are Associated with Prostate Cancer Risk in a Chinese Population.” Asian Journal of Andrology, vol. 14, no. 6, Nov. 2012, pp. 864–69. PubMed, https://doi.org/10.1038/aja.2012.101.
Li, Su-Hua, et al. “Interaction Between Hypoxia-Inducible Factor 1-Alpha Gene Polymorphism and Helicobacter Pylori Infection on Gastric Cancer in a Chinese Tibetan Population.” Biochemical Genetics, vol. 63, no. 3, June 2025, pp. 2363–72. PubMed, https://doi.org/10.1007/s10528-024-10776-8.
Li, Yujie, et al. “The Association between the Rs11549465 Polymorphism in the Hif-1α Gene and Cancer Risk: A Meta-Analysis.” International Journal of Clinical and Experimental Medicine, vol. 8, no. 2, 2015, pp. 1561–74.
———. “The Association between the Rs11549465 Polymorphism in the Hif-1α Gene and Cancer Risk: A Meta-Analysis.” International Journal of Clinical and Experimental Medicine, vol. 8, no. 2, Feb. 2015, pp. 1561–74. PubMed Central, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4402731/.
López-Reyes, Alberto, et al. “The HIF1A Rs2057482 Polymorphism Is Associated with Risk of Developing Premature Coronary Artery Disease and with Some Metabolic and Cardiovascular Risk Factors. The Genetics of Atherosclerotic Disease (GEA) Mexican Study.” Experimental and Molecular Pathology, vol. 96, no. 3, June 2014, pp. 405–10. PubMed, https://doi.org/10.1016/j.yexmp.2014.04.010.
Luo, Beibei, et al. “Hepatic PHD2/HIF-1α Axis Is Involved in Postexercise Systemic Energy Homeostasis.” FASEB Journal: Official Publication of the Federation of American Societies for Experimental Biology, vol. 32, no. 9, Sept. 2018, pp. 4670–80. PubMed, https://doi.org/10.1096/fj.201701139R.
Maybin, Jacqueline A., et al. “Hypoxia and Hypoxia Inducible Factor-1α Are Required for Normal Endometrial Repair during Menstruation.” Nature Communications, vol. 9, Jan. 2018, p. 295. PubMed Central, https://doi.org/10.1038/s41467-017-02375-6.
McGarry, Trudy, et al. “Hypoxia, Oxidative Stress and Inflammation.” Free Radical Biology and Medicine, Oxidative stress and altered redox signalling in autoimmune and connective tissue diseases, vol. 125, Sept. 2018, pp. 15–24. ScienceDirect, https://doi.org/10.1016/j.freeradbiomed.2018.03.042.
McPhee, J. S., et al. “HIF1A P582S Gene Association with Endurance Training Responses in Young Women.” European Journal of Applied Physiology, vol. 111, no. 9, Sept. 2011, pp. 2339–47. PubMed, https://doi.org/10.1007/s00421-011-1869-4.
Miyata, Toshio, et al. “Hypoxia. 1. Intracellular Sensors for Oxygen and Oxidative Stress: Novel Therapeutic Targets.” American Journal of Physiology. Cell Physiology, vol. 300, no. 2, Feb. 2011, pp. C226-231. PubMed, https://doi.org/10.1152/ajpcell.00430.2010.
Nagle, Dale G., and Yu-Dong Zhou. “Natural Product-Based Inhibitors of Hypoxia-Inducible Factor-1 (HIF-1).” Current Drug Targets, vol. 7, no. 3, Mar. 2006, pp. 355–69. PubMed Central, https://doi.org/10.2174/138945006776054979.
Nagy, Geza, et al. “Association of Hypoxia Inducible Factor-1 Alpha Gene Polymorphism with Both Type 1 and Type 2 Diabetes in a Caucasian (Hungarian) Sample.” BMC Medical Genetics, vol. 10, Aug. 2009, p. 79. PubMed, https://doi.org/10.1186/1471-2350-10-79.
———. “Association of Hypoxia Inducible Factor-1 Alpha Gene Polymorphism with Both Type 1 and Type 2 Diabetes in a Caucasian (Hungarian) Sample.” BMC Medical Genetics, vol. 10, Aug. 2009, p. 79. PubMed, https://doi.org/10.1186/1471-2350-10-79.
Ota, Hiroyo, et al. “Relationship Between Intermittent Hypoxia and Type 2 Diabetes in Sleep Apnea Syndrome.” International Journal of Molecular Sciences, vol. 20, no. 19, Sept. 2019, p. 4756. PubMed, https://doi.org/10.3390/ijms20194756.
Pavan, Aline Renata, et al. “Unraveling the Anticancer Effect of Curcumin and Resveratrol.” Nutrients, vol. 8, no. 11, Nov. 2016, p. 628. PubMed Central, https://doi.org/10.3390/nu8110628.
Peña-Villalobos, Isaac, et al. “Hyperbaric Oxygen Increases Stem Cell Proliferation, Angiogenesis and Wound-Healing Ability of WJ-MSCs in Diabetic Mice.” Frontiers in Physiology, vol. 9, July 2018. Frontiers, https://doi.org/10.3389/fphys.2018.00995.
Regueira, Tomas, et al. “Hypoxia Inducible Factor-1 Alpha Induction by Tumour Necrosis Factor-Alpha, but Not by Toll-like Receptor Agonists, Modulates Cellular Respiration in Cultured Human Hepatocytes.” Liver International: Official Journal of the International Association for the Study of the Liver, vol. 29, no. 10, Nov. 2009, pp. 1582–92. PubMed, https://doi.org/10.1111/j.1478-3231.2009.02109.x.
Ryan, Silke. “Mechanisms of Cardiovascular Disease in Obstructive Sleep Apnoea.” Journal of Thoracic Disease, vol. 10, no. Suppl 34, Dec. 2018, pp. S4201–11. PubMed, https://doi.org/10.21037/jtd.2018.08.56.
Serebrovska, Tetiana V., et al. “Intermittent Hypoxia/Hyperoxia Versus Intermittent Hypoxia/Normoxia: Comparative Study in Prediabetes.” High Altitude Medicine & Biology, vol. 20, no. 4, Dec. 2019, pp. 383–91. PubMed, https://doi.org/10.1089/ham.2019.0053.
Sunkari, Vivekananda Gupta, et al. “Hyperbaric Oxygen Therapy Activates Hypoxia-Inducible Factor 1 (HIF-1), Which Contributes to Improved Wound Healing in Diabetic Mice.” Wound Repair and Regeneration: Official Publication of the Wound Healing Society [and] the European Tissue Repair Society, vol. 23, no. 1, 2015, pp. 98–103. PubMed, https://doi.org/10.1111/wrr.12253.
Tin’kov, Aleksey N., and Valeriy A. Aksenov. “Effects of Intermittent Hypobaric Hypoxia on Blood Lipid Concentrations in Male Coronary Heart Disease Patients.” High Altitude Medicine & Biology, vol. 3, no. 3, 2002, pp. 277–82. PubMed, https://doi.org/10.1089/152702902320604250.
Vakhitova, Y. V., et al. “Molecular Mechanism Underlying the Action of Substituted Pro-Gly Dipeptide Noopept.” Acta Naturae, vol. 8, no. 1, 2016, pp. 82–89. PubMed Central, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4837574/.
Xie, Rui-Yan, et al. “Salidroside and FG-4592 Ameliorate High Glucose-Induced Glomerular Endothelial Cells Injury via HIF Upregulation.” Biomedicine & Pharmacotherapy = Biomedecine & Pharmacotherapie, vol. 118, Oct. 2019, p. 109175. PubMed, https://doi.org/10.1016/j.biopha.2019.109175.
Yang, Xi, et al. “HIF-1α 1772 C/T and 1790 G/A Polymorphisms Are Significantly Associated with Higher Cancer Risk: An Updated Meta-Analysis from 34 Case-Control Studies.” PLoS ONE, vol. 8, no. 11, Nov. 2013, p. e80396. PubMed Central, https://doi.org/10.1371/journal.pone.0080396.
Yu, Zhen-Gang, et al. “The Association of Genetic Polymorphisms of Hypoxia Inducible Factor-1 Alpha and Vascular Endothelial Growth Factor with Increased Risk of Chronic Obstructive Pulmonary Disease: A Case-Control Study.” The Kaohsiung Journal of Medical Sciences, vol. 33, no. 9, Sept. 2017, pp. 433–41. PubMed, https://doi.org/10.1016/j.kjms.2017.05.014.
Zhang, Shixiao, et al. “Chronic Intermittent Hybobaric Hypoxia Protects against Cerebral Ischemia via Modulation of mitoKATP.” Neuroscience Letters, vol. 635, Dec. 2016, pp. 8–16. PubMed, https://doi.org/10.1016/j.neulet.2016.10.025.