Key takeaways:
- The SOD1 gene encodes an enzyme important in reducing oxidative stress and protecting your cells from damage.
- SOD1 uses copper and zinc in neutralizing free radicals, such as superoxide radicals, turning them into less reactive molecules that the body can clear out.
- This helps to prevent damage to DNA and other proteins in cells.
- Genetic variants in SOD1 can change how well this antioxidant system works. These variants may increase the risk of several chronic diseases.
SOD1: Superoxide Dismutase 1
Our body has built-in antioxidants that fight against cellular stress. The superoxide dismutase (SOD1) enzyme fights against oxidative stress in your cells, constantly battling to balance out oxidants with antioxidants.
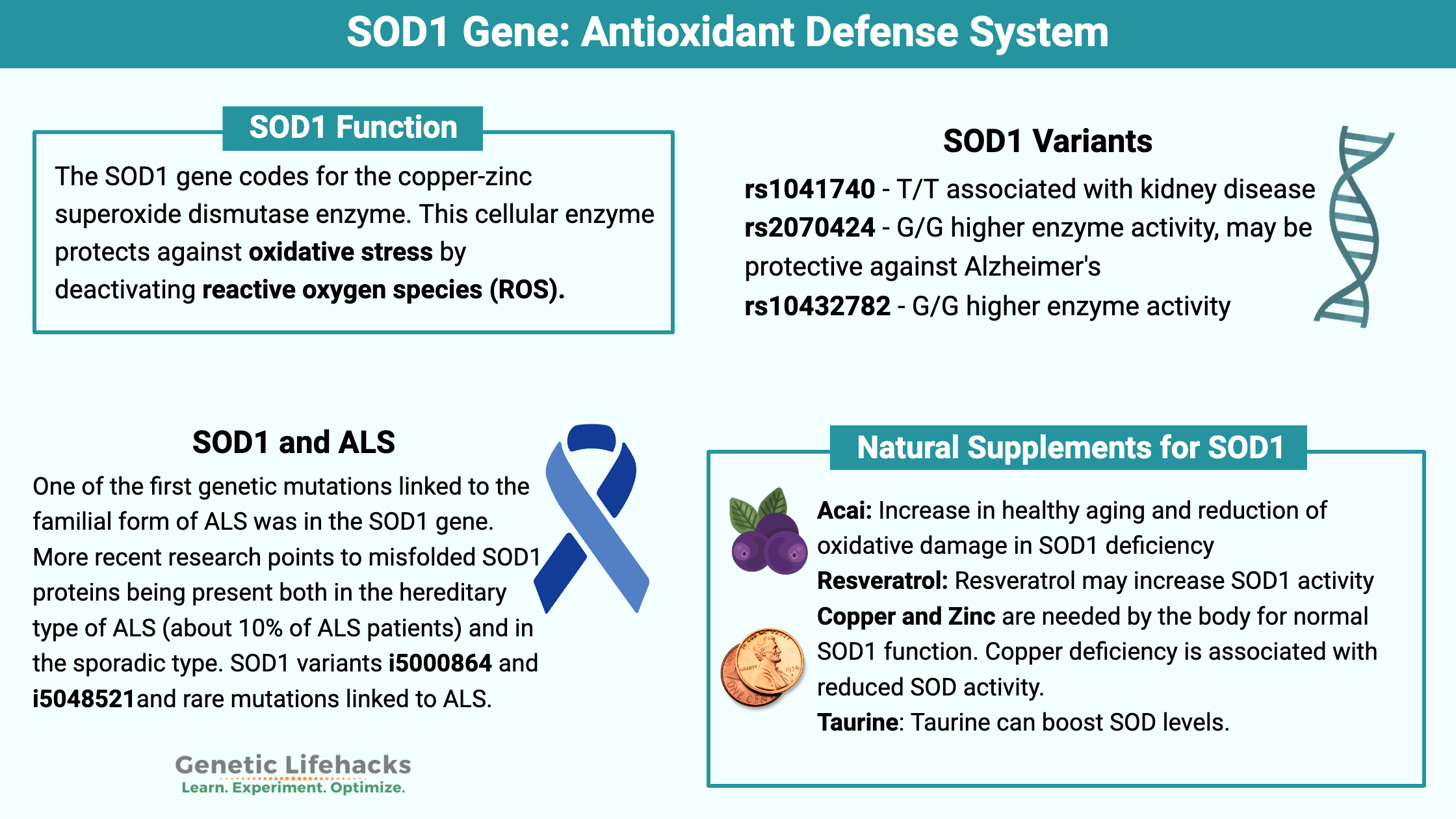
The SOD1 gene codes for the copper-zinc superoxide dismutase enzyme. This cellular enzyme protects against oxidative stress by deactivating reactive oxygen species (ROS).
Three types of superoxide dismutases are part of our built-in antioxidant system:[ref]
- SOD1, found in the cytosol or fluid of cells
- SOD2, a mitochondrial antioxidant
- SOD3, which is extracellular or outside of the cells
SOD1 binds copper and zinc ions together and destroys free radicals. It is an antioxidant that our body produces, which converts superoxide radicals into oxygen and hydrogen peroxide. Other enzymes then break down the hydrogen peroxide.
Balancing out oxidation reactions:
This is all about electrons.. Superoxide is two oxygen molecules bonded together but with one electron missing (O2−). Normal oxygen that we breathe in the air is a stable combo of two oxygen molecules.
The superoxide compound, which lacks an electron, is highly reactive, wanting to bind with another molecule to balance out its charge.
SOD1 – superoxide dismutase 1 – can counteract the superoxide produced in cellular reactions. SOD1’s primary function is to convert superoxide radicals (O2−) into hydrogen peroxide (H2O2) and oxygen (O2). The hydrogen peroxide produced is further converted into water by other enzymes, such as glutathione peroxidase.
While we often think of antioxidants as being good and reactive oxygen species (ROS), such as superoxide, as being ‘bad’, ROS does play a vital role in cell signaling. Our cells need to be in balance with antioxidants and pro-oxidants.[ref]
Additionally, our immune system produces pro-oxidants such as superoxide to kill invading pathogens.
Balance is key, and SOD1 helps provide that balance between ROS and oxidative stress.
Research on SOD1:
Research on the different genetic variants shows that SOD1 impacts heart disease risk, kidney disease, hearing loss, and diabetes. Oxidative stress plays a role in all of these chronic conditions, and decreased SOD1 (or too much ROS) can contribute to the development of these diseases.[ref][ref]
Oxidative stress seems to play a role in Alzheimer’s disease as well, and SOD1 variants have been studied to see if they affect the risk of Alzheimer’s. For rs2070424, the G allele offers protection against Alzheimer’s disease regardless of APOE gene status.[ref][ref]
Related article: Check your APOE genes (only if you want to know more about your Alzheimer’s risk)
Overall, higher SOD activity has links to decreased mortality in women. Oddly, this statistic didn’t hold true for men.[ref]
SOD1 in ALS:
One of the first genetic mutations linked to the familial form of ALS was in the SOD1 gene. More recent research points to misfolded SOD1 proteins being present both in the hereditary type of ALS (about 10% of ALS patients) and in the sporadic type. While there is still a lot that researchers don’t know here, it looks like the SOD1 mutations cause a gain-of-function and altered folding in a way that causes a loss of motor neuron function.[ref]
SOD1 Genotype Report:
SOD1 genetic variants can cause either an increase or a decrease in enzyme activity. Rare mutations in SOD1 have links to several diseases, including an inherited form of ALS (Lou Gehrig’s disease).[ref] More common variants are linked with conditions such as diabetes, cardiovascular disease, kidney disease, and hearing damage.
Access this content:
An active subscription is required to access this content.
Lifehacks for increasing SOD1:
Dietary changes:
A recent clinical trial looked at the effect of a high-antioxidant diet on oxidative stress. The study participants on a Mediterranean diet were also supplemented with apple and bergamot juice. After two weeks, they had an increase in SOD1.[ref]
Increasing zinc was shown in a clinical trial involving multiple myeloma patients to increase SOD1 levels.[ref] The study used a low-dose supplement, but dietary sources of zinc include oysters, beef, pumpkin seeds, crab, dark-meat poultry, and dairy.
4 Natural Supplements to increase SOD1:
Acai:
Freeze-dried acai powder, in an animal model of SOD1 deficiency, shows an increase in healthy aging and reduction of oxidative damage.[ref] Freeze-dried Acai powder can be found online, and acai juice can be found in most grocery stores.
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
Nrf2 Pathway: Increasing the Body’s Ability to Get Rid of Toxins
References:
Gualtieri, Paola, et al. “Antioxidant-Enriched Diet on Oxidative Stress and Inflammation Gene Expression: A Randomized Controlled Trial.” Genes, vol. 14, no. 1, Jan. 2023, p. 206. PubMed, https://doi.org/10.3390/genes14010206.
Jahankhani, Kasra, et al. “Adjuvant Therapy with Zinc Supplementation; Anti-Inflammatory and Anti-Oxidative Role in Multiple Myeloma Patients Receiving Autologous Hematopoietic Stem Cell Transplantation: A Randomized Controlled Clinical Trial.” Biometals: An International Journal on the Role of Metal Ions in Biology, Biochemistry, and Medicine, vol. 37, no. 6, Dec. 2024, pp. 1609–27. PubMed, https://doi.org/10.1007/s10534-024-00630-0.