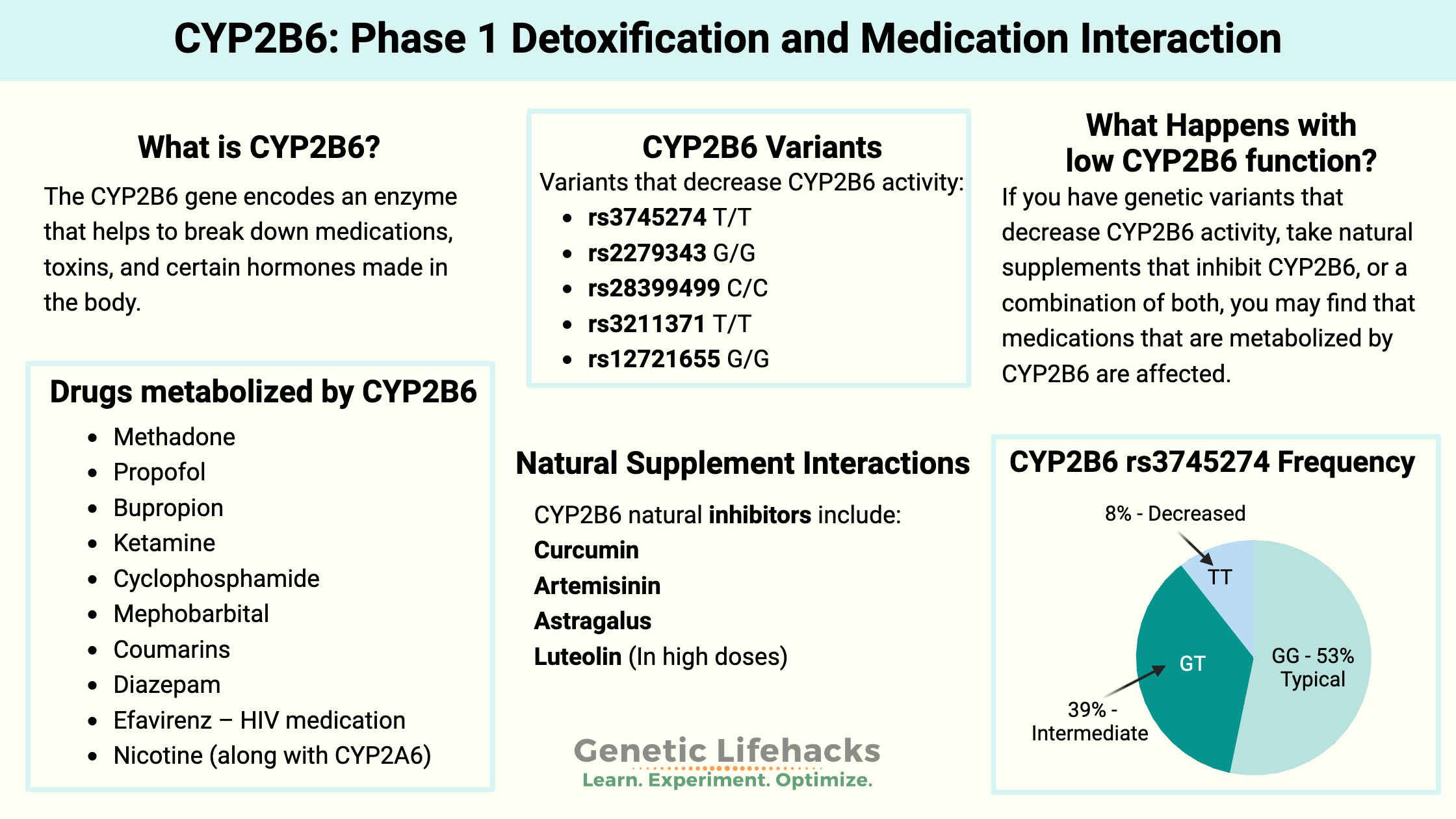
will see their genotype report below and the solutions in the Lifehacks section.
The CYP2B6 gene is part of the CYP450 (cytochrome P45) family of phase I genes. It encodes an enzyme that helps break down (metabolize) certain substances, making them more easily eliminated from the body.
CYP2B6 breaks down medications, toxins, and certain hormones made in the body (endogenous substances).
Genetic variants in the CYP2B6 gene cause large variations in how the enzyme works, depending on the variant. For some people, genetic variants cause significant decreases in enzyme function, while others may have faster enzyme function.[ref]
Just like prescription and OTC medications, natural supplements can also interact with the CYP enzymes.
While we often focus on drug interactions, the CYP enzymes all break down substances that are naturally made in the body – endogenous compounds. Similarly to drugs, many of the endogenous substances broken down by the CYP enzymes can also be metabolized by multiple routes, multiple enzymes. Thus, an impaired CYP2B6 enzyme may not be a huge problem unless combined with other CYP variants.
Phthalates are endocrine disruptors that can mimic estrogen. Some phthalates are also metabolized by CYP2B6.[ref]
Mice with reduced CYP2B6 function ended up becoming more obese and having higher triglycerides and cholesterol (compared to normal mice) when fed a bunch of corn oil.[ref]
The CYP2B6 enzyme also metabolizes organophosphates at lower concentrations, such as from normal exposure to pesticide residue.[ref] Additionally, CYP2B6 is the only enzyme in the body that can break down methoxychlor, which is an insecticide used against flies. It is a banned substance in the EU and the US.[ref]
Ahmad, Taha, et al. “Tell-Tale SNPs: The Role of CYP2B6 in Methadone Fatalities.” Journal of Analytical Toxicology, vol. 41, no. 4, May 2017, pp. 325–33. PubMed, https://doi.org/10.1093/jat/bkw135.
Appiah-Opong, Regina, et al. “Inhibition of Human Recombinant Cytochrome P450s by Curcumin and Curcumin Decomposition Products.” Toxicology, vol. 235, nos. 1–2, June 2007, pp. 83–91. PubMed, https://doi.org/10.1016/j.tox.2007.03.007.
Bloom, Adam Joseph, et al. “Nicotine Oxidation by Genetic Variants of CYP2B6 and in Human Brain Microsomes.” Pharmacology Research & Perspectives, vol. 7, no. 2, Mar. 2019, p. e00468. PubMed Central, https://doi.org/10.1002/prp2.468.
Campesi, Ilaria, et al. “The Sex–Gender Effects in the Road to Tailored Botanicals.” Nutrients, vol. 11, no. 7, July 2019, p. 1637. PubMed Central, https://doi.org/10.3390/nu11071637.
“ClinPGx.” ClinPGx, https://www.clinpgx.org/literature/14895137. Accessed 17 Feb. 2026.
“ClinPGx.” ClinPGx, https://www.clinpgx.org/variant/PA166155409/clinicalAnnotation. Accessed 17 Feb. 2026.
Crane, Alice L., et al. “Effect of CYP2B6*6 and CYP2C19*2 Genotype on Chlorpyrifos Metabolism.” Toxicology, vol. 293, no. 0, Mar. 2012, pp. 115–22. PubMed Central, https://doi.org/10.1016/j.tox.2012.01.006.
Croom, Edward L., et al. “Human Variation in CYP-Specific Chlorpyrifos Metabolism.” Toxicology, vol. 276, no. 3, Oct. 2010, pp. 184–91. PubMed, https://doi.org/10.1016/j.tox.2010.08.005.
Damiri, Basma, and William S. Baldwin. “Cyp2b-Knockdown Mice Poorly Metabolize Corn Oil and Are Age-Dependent Obese.” Lipids, vol. 53, no. 9, Sept. 2018, pp. 871–84. PubMed Central, https://doi.org/10.1002/lipd.12095.
Duarte, Hélder, et al. “Population Approach to Efavirenz Therapy.” Journal of Pharmaceutical Sciences, vol. 106, no. 10, Oct. 2017, pp. 3161–66. PubMed, https://doi.org/10.1016/j.xphs.2017.06.004.
Haas, David W., et al. “Relationships between CYP2B6 Polymorphisms and Pharmacokinetics Following a Single Dose of Nevirapine or Efavirenz in African Americans.” The Journal of Infectious Diseases, vol. 199, no. 6, Mar. 2009, pp. 872–80. PubMed Central, https://doi.org/10.1086/597125.
Hedrich, William D., et al. “Insights into CYP2B6-Mediated Drug–Drug Interactions.” Acta Pharmaceutica Sinica. B, vol. 6, no. 5, Sept. 2016, pp. 413–25. PubMed Central, https://doi.org/10.1016/j.apsb.2016.07.016.
Isaza, Carlos, et al. “Influence of CYP2B6 Genotype on Methadone Dosage in Patients from the Methadone Maintenance Treatment (MMT) Program in Pereira, Colombia.” Life, vol. 13, no. 4, Apr. 2023, p. 1038. PubMed Central, https://doi.org/10.3390/life13041038.
Kim, Harim, et al. “Inhibition of Cytochrome P450 2B6 by Astragalus Extract Mixture HT042.” Toxicological Research, vol. 36, no. 3, July 2020, pp. 195–201. PubMed, https://doi.org/10.1007/s43188-019-00027-z.
Li, Yibai, et al. “CYP2B6*6 Allele and Age Substantially Reduce Steady-State Ketamine Clearance in Chronic Pain Patients: Impact on Adverse Effects.” British Journal of Clinical Pharmacology, vol. 80, no. 2, Aug. 2015, pp. 276–84. PubMed Central, https://doi.org/10.1111/bcp.12614.
Lin, Hsia-lien, et al. “Roles of Residues F206 and V367 in Human CYP2B6: Effects of Mutations on Androgen Hydroxylation, Mechanism-Based Inactivation, and Reversible Inhibition.” Drug Metabolism and Disposition, vol. 44, no. 11, Nov. 2016, pp. 1771–79. PubMed Central, https://doi.org/10.1124/dmd.116.071662.
Nguefeu Nkenfou, Carine, et al. “Adverse Drug Reactions Associated with CYP 2B6 Polymorphisms in HIV/AIDS-Treated Patients in Yaoundé, Cameroon.” The Application of Clinical Genetics, vol. 12, 2019, pp. 261–68. PubMed, https://doi.org/10.2147/TACG.S226318.
Obach, R. Scott, et al. “Sertraline Is Metabolized by Multiple Cytochrome P450 Enzymes, Monoamine Oxidases, and Glucuronyl Transferases in Human: An in Vitro Study.” Drug Metabolism and Disposition: The Biological Fate of Chemicals, vol. 33, no. 2, Feb. 2005, pp. 262–70. PubMed, https://doi.org/10.1124/dmd.104.002428.
Ohe, T., et al. “Novel Metabolic Pathway of Estrone and 17beta-Estradiol Catalyzed by Cytochrome P-450.” Drug Metabolism and Disposition: The Biological Fate of Chemicals, vol. 28, no. 2, Feb. 2000, pp. 110–12.
Piver, Bertrand, et al. “Differential Inhibition of Human Cytochrome P450 Enzymes by Epsilon-Viniferin, the Dimer of Resveratrol: Comparison with Resveratrol and Polyphenols from Alcoholized Beverages.” Life Sciences, vol. 73, no. 9, July 2003, pp. 1199–213. PubMed, https://doi.org/10.1016/s0024-3205(03)00420-x.
Robinson, Katherine M., et al. “Clinical Pharmacogenetics Implementation Consortium Guideline for CYP2B6 Genotype and Methadone Therapy.” Clinical Pharmacology and Therapeutics, vol. 116, no. 4, June 2024, pp. 932–38. PubMed Central, https://doi.org/10.1002/cpt.3338.
Tian, Meiping, et al. “Phthalate Side-Chain Structures and Hydrolysis Metabolism Associated with Steroidogenic Effects in MLTC-1 Leydig Cells.” Toxicology Letters, vol. 308, June 2019, pp. 56–64. ScienceDirect, https://doi.org/10.1016/j.toxlet.2019.03.013.
Tomaz, Paulo Roberto Xavier, et al. “CYP2B6 Rs2279343 Polymorphism Is Associated with Smoking Cessation Success in Bupropion Therapy.” European Journal of Clinical Pharmacology, vol. 71, no. 9, Sept. 2015, pp. 1067–73. PubMed, https://doi.org/10.1007/s00228-015-1896-x.
Walsky, Robert L., et al. “Evaluation of 227 Drugs for in Vitro Inhibition of Cytochrome P450 2B6.” Journal of Clinical Pharmacology, vol. 46, no. 12, Dec. 2006, pp. 1426–38. PubMed, https://doi.org/10.1177/0091270006293753.
Wang, Hongbing, and Leslie M. Tompkins. “CYP2B6: New Insights into a Historically Overlooked Cytochrome P450 Isozyme.” Current Drug Metabolism, vol. 9, no. 7, Sept. 2008, pp. 598–610. PubMed Central, https://doi.org/10.2174/138920008785821710.
Wang, Pan-Fen, Alicia Neiner, et al. “Efavirenz Metabolism: Influence of Polymorphic CYP2B6 Variants and Stereochemistry.” Drug Metabolism and Disposition, vol. 47, no. 10, Oct. 2019, pp. 1195–205. PubMed Central, https://doi.org/10.1124/dmd.119.086348.
Wang, Pan-Fen, Yanming Yang, et al. “Natural Products Inhibition of Cytochrome P450 2B6 Activity and Methadone Metabolism.” Drug Metabolism and Disposition: The Biological Fate of Chemicals, vol. 52, no. 3, Feb. 2024, pp. 252–65. PubMed, https://doi.org/10.1124/dmd.123.001578.
Wyen, Christoph, et al. “Impact of CYP2B6 983T>C Polymorphism on Non-Nucleoside Reverse Transcriptase Inhibitor Plasma Concentrations in HIV-Infected Patients.” The Journal of Antimicrobial Chemotherapy, vol. 61, no. 4, Apr. 2008, pp. 914–18. PubMed, https://doi.org/10.1093/jac/dkn029.
Ying, Jing-Yan, et al. “[Research progress on interactions between luteolin (glucosides) and drug-metabolizing enzyme].” Yao Xue Xue Bao = Acta Pharmaceutica Sinica, vol. 43, no. 4, Apr. 2008, pp. 335–42.
Zhang, Jingwei, et al. “Genetic Polymorphisms of CYP2B6 Is a Risk of Metabolic Associated Fatty Liver Disease in Chinese Population.” Toxicology and Applied Pharmacology, vol. 481, Dec. 2023, p. 116770. PubMed, https://doi.org/10.1016/j.taap.2023.116770.
Zhang, Li-Qun, et al. “Genetic Variants, Haplotype Determination, and Function of Novel Alleles of CYP2B6 in a Han Chinese Population.” Heliyon, vol. 10, no. 7, Mar. 2024, p. e28952. PubMed Central, https://doi.org/10.1016/j.heliyon.2024.e28952.