Key takeaways:
~ Your genetic variants influence which microbes are likely to live in your gut.
~ Your gut microbiome impacts your weight.
~ Together, understanding your genetics, your diet, and your gut microbiome can help with weight management.
Your Genetic Variants Influence Your Gut Microbiome:
The human gut microbiome is made up of bacteria, viruses, fungi, archaea, and obelisks that inhabit the ecosystem of our intestinal tract. It plays an integral role in our overall health.
Research increasingly shows that host genetics – your gene variants – play an important role in shaping which microbes thrive in our gut. Your genetic variants interact with environmental influences like diet, lifestyle, and medication to shape your gut microbiome.
Heritability and Twin Studies:
Studies of twins and families have revealed that certain components of the gut microbiome are heritable. This means that genetic variants partly explain why some people naturally have higher levels of specific microbes than others.[ref]
Genetic variants can change the mucosal barrier that hosts microbes in the intestines and
Let’s take a look at how and why some of your genes influence your microbiome, and how these two factors come together to impact metabolic health.
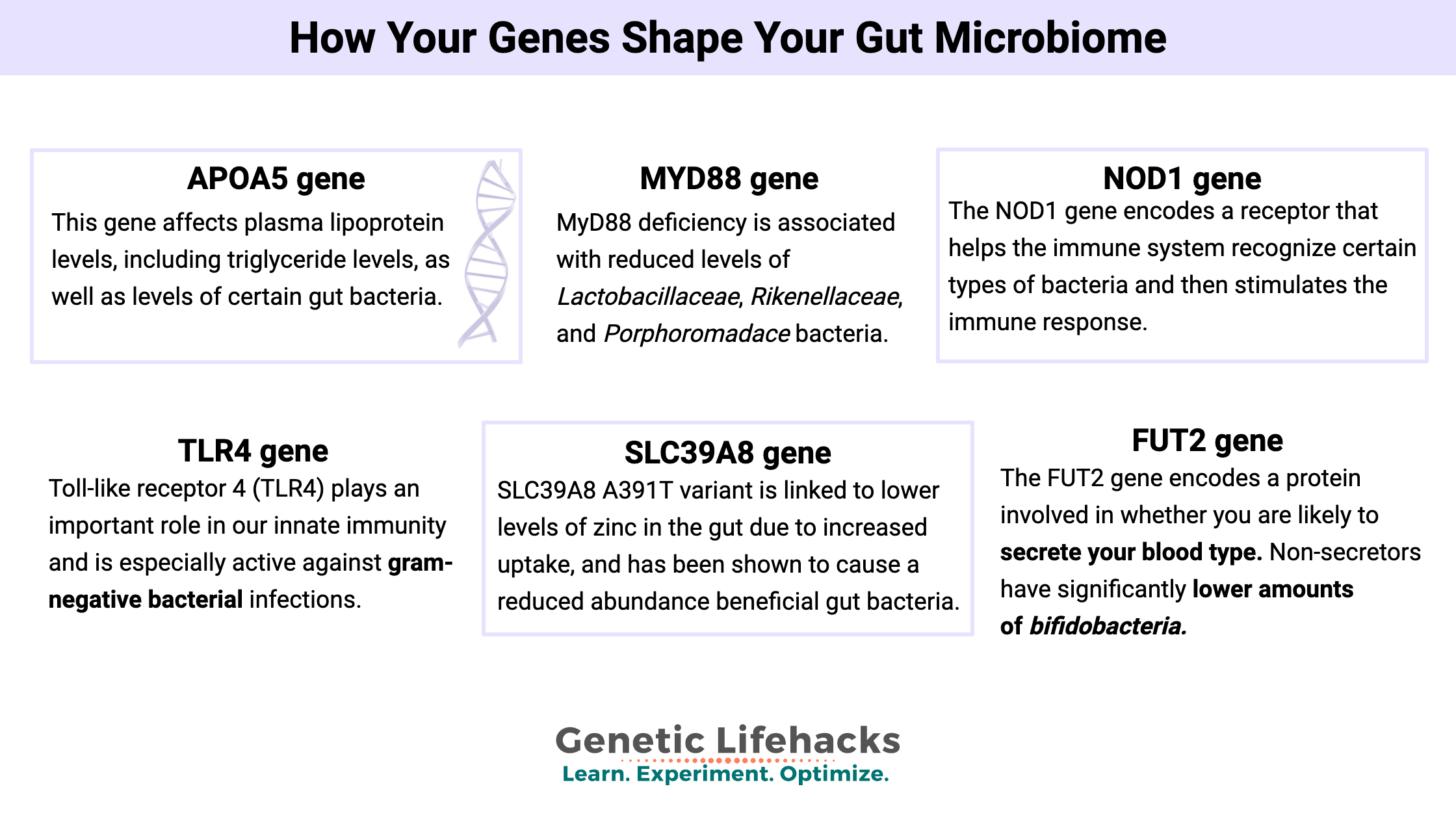
APOA5 gene:
This gene affects plasma lipoprotein levels, including triglyceride levels, as well as levels of certain gut bacteria. Several SNPs in this gene have associations with triglyceride levels and obesity risk.
An April 2016 twin study looked at metabolic syndrome, the gut microbiome, and genetics. They found that rs651821, a SNP in the APOA5 gene, influenced the risk of metabolic syndrome. The study results found that metabolic syndrome was more common in those with the C allele, and for each C allele, there was an average increase in triglycerides of 24.65 mg/dL. In looking at the gut microbiome, the study found that those with the minor allele (C) had fewer Bifidobacterium regardless of their MetS status. Bifidobacterium species have links (in this study and in others) to lower BMI and better overall health.[ref]
MYD88 gene:
The MYD88 gene is a signal receptor involved in the innate immune system. It’s a part of the TLR (Toll-like receptor) and interleukin-1 receptor (IL-1R) signaling pathways, which regulate the immune response to foreign invaders like bacteria – including gut bacteria.
A mouse study using mice bred to be deficient in (MyD88 knockout) found that the mice had significantly reduced levels of Lactobacillaceae (Firmicutes), Rikenellaceae (Bacteroidetes), and Porphoromadaceae (Bacteroidetes) bacteria compared to mice with normal MyD88.[ref]
Another study found that MyD88 is important in clearing Listeria monocytogenes infection. Listeria infections usually come from eating contaminated foods, giving some people a nasty case of food poisoning. Normally the intestinal mucosa can protect against systemic infection, but MyD88 deficiency increases susceptibility to systemic infection by the pathogen.[ref] Other mouse studies have found that MyD88 knockout mice are more susceptible to tuberculosis.[ref] This has been confirmed in humans with MyD88 polymorphisms as well.
A recent mouse study found that MyD88 knockout mice had an increased number of bacteria in contact with the epithelial wall of the intestine. Normally, the intestinal mucosal layers protect the cell wall of the intestines from direct contact with bacteria.[ref]
NOD1 gene:
The NOD1 gene encodes nucleotide-binding oligomerization domain containing 1, which is a receptor that helps the immune system recognize certain types of bacteria and then stimulates the immune response.
Mice deficient in Nod1 have increased susceptibility to H. pylori.[ref] Other studies have shown that NOD1 variants lead to an increased risk of inflammatory bowel diseases in some populations. One study concluded, “Taken together, these data may suggest that NOD1 plays a variable role in different populations that could depend upon environmental and dietary factors.”[ref]
TLR4 gene:
Toll-like receptor 4 (TLR4) plays an important role in our innate immunity and is especially active against gram-negative bacterial infections. TLR4 variants have been investigated in conjunction with cancer risk, vaccine response, and transplant rejection, among other things.[ref][ref]
SLC39A8 gene:
SLC39A8, also called ZIP8, is a metal ion transporter in the intestines as well as on cell membranes. Zinc and manganese are the primary metals transported by SLC39A8, and they are also key to a balanced immune response. SLC39A8 is upregulated in response to a pathogen infection. The gut microbiome is also affected by how much zinc is available in the gut vs. taken up into the body. The gut microbiome actually uses about 20% of your dietary zinc, so the amount left for the gut microbiome can affect which types of bacteria are more likely to be abundant.
High levels of zinc supplementation increase in relative abundance of Bacteroidetes and decrease Firmicutes sp. Along those same lines, the SLC39A8 A391T variant, which is linked to lower levels of zinc and manganese in the gut due to increased uptake, has been shown to cause a reduced abundance of Anaerostipes, Coprococcus, Roseburia, Lachnospira, SMB53, Ruminococcaceae, Eubacterium, Dorea, and Bacteroides.[ref]
FUT2 gene:
The FUT2 gene encodes a protein involved in whether you are likely to secrete your blood type in your saliva and gut mucosa. Blood type is determined by glycans, which can also be used by gut microbes as food. Thus, non-secretors are likely to have a different microbial composition due to the lack of secreted blood-type glycans in the mucosal layer.
A 2011 study showed that non-secretors had significantly lower amounts of bifidobacteria in their gut microbiome. This makes sense because bifidobacteria are fed, in part, by the oligosaccharides in the intestinal mucosa. The same study showed that non-secretors also had a lower diversity of bacteria.[ref]
Being a non-secretor of your blood type also influences viral infections, and non-secretors are unlikely to get sick from norovirus or rotavirus (stomach flu) infection.
The Gut Microbiome Strongly Influences Your Weight
Several studies have come out recently showing that those who are overweight have a different gut microbiome composition than those who are lean. There have also been interesting mouse studies showing that transplanting feces from obese mice into lean mice causes lean mice to become obese.
A 2010 study found that supplementation with the probiotic Lactobacillus gasseri SBT2055 decreased abdominal fat and body weight.[ref] The case for our microbes helping to shape our weight is fairly strong.
Now let’s take a look at how your genetic variants impact the function of these genes.
Gut and Weight Genotype Report:
Access this content:
An active subscription is required to access this content.
Lifehacks:
Promoting bifidobacteria:
Eating prebiotic fiber, such as inulin or FOS, may increase your bifidobacteria.[ref]
There are probiotics available containing multiple strains of bifidobacteria.
More to read:
A recent study did an extensive analysis of the microbes associated with visceral fat, BMI, and other obesity markers.[ref] The study also found several genes correlated with specific bacteria that are associated with obesity. The SNPs (none of which are included in 23andMe results) were in the FHIT, TDRGI, and ELAVL4 genes. The full study is open to read and worth checking out.
Recap of your genes:
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
Gut Genes: How Your Genetic Variants Impact Your Gut Microbiome
References: