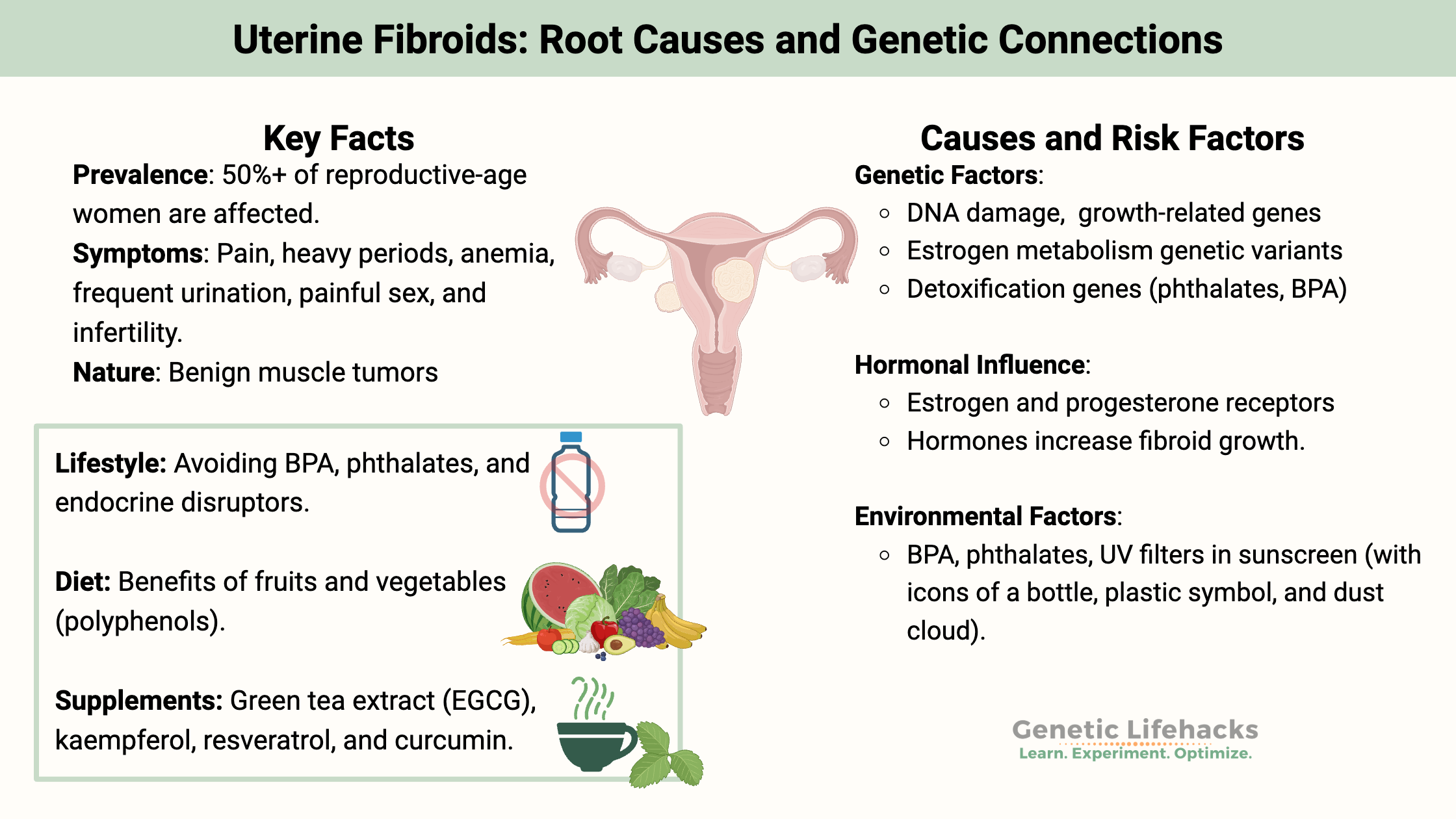
Key takeaways:
~ Uterine fibroids are benign tumors that grow in the muscle cells of the uterus.
~ While estimates vary, at least half of American women who are of reproductive age have fibroids.[ref]
~ Genetic variants related to estrogen metabolism, vascular growth, and detoxification all increase the odds of having fibroids.
~ Environmental exposure to endocrine disruptors increases the risk and growth of fibroids.
This article digs into the causes of fibroids, explains how your genetic variants can add to the susceptibility, and offers solutions backed by research.
Members will see their genotype report below and the solutions in the Lifehacks section. Consider joining today.Uterine fibroids:
Symptoms of fibroids include:
- Painful or heavy periods
- Anemia (due to heavy periods)
- Frequent need to urinate
- Painful sex
- Low back pain
Fibroids can also play a role in infertility for some women.
Not all women have symptoms with fibroids – it depends on the location and size.
What is a fibroid? Uterine leiomyoma, another name for fibroids, means smooth muscle tumor. It is a benign growth in the uterus.
Causes of fibroids
 You would think that something that affects more than half of women would be well understood and have good solutions available. Oddly enough, there are still questions surrounding the causes of fibroids, and a lot of the solutions available are either ineffective or come with significant side effects.
You would think that something that affects more than half of women would be well understood and have good solutions available. Oddly enough, there are still questions surrounding the causes of fibroids, and a lot of the solutions available are either ineffective or come with significant side effects.
Genes and Lifestyle:
Genetics plays a role in the susceptibility to fibroids, but, as you will see, lifestyle factors are also important.[ref]
Stem cells and growth
Fibroids are medically classified as benign tumors, meaning they are nearly always non-cancerous growths.
Genetic studies show that a fibroid will arise from one cell that has somewhat unchecked growth. Stem cells are present in the muscles of the uterus, which need to be able to grow and change both monthly and during pregnancy.[ref][ref]
The stem cells in the uterine muscles are capable of replicating into more muscle tissue – that is their whole purpose. When there is DNA damage in a growth-related gene, the stem cells go on a growth spree and become fibroids. Additionally, the conditions for growth and proliferation of the cells need to be right (see below). The body’s normal way of dealing with damaged DNA also needs to be overridden.[ref]
Estrogen, progesterone, and growth
Estrogen and progesterone are involved in the formation and growth of fibroids, which occur mainly during the reproductive years when estrogen and progesterone levels are high. Fibroids usually decrease in size once a woman reaches menopause.
The cells in a fibroid have increased estrogen and progesterone receptors. Animal studies show that the addition of estrogen alone, or estrogen plus progesterone, can increase the size of fibroids.[ref]
In the uterus, estrogen upregulates IFG-1 (insulin-like growth factor 1), EGFR, TGF-beta1. These are all growth-related molecules.[ref]
Interleukin-1 (IL-1) is an inflammatory cytokine that can inhibit REST, which is a tumor suppressor. Interleukin-1 also happens to be upregulated in fibroid tissue, thus decreasing the tumor suppressor.[ref]
Exposure to endocrine disruptors
Adding fuel to the fibroid fire, exposure to endocrine-disrupting chemicals such as BPA and phthalates also increases the risk and growth of fibroids.
BPA is a component of plastics and a known endocrine disruptor, similar in structure to estrogen. A cell study shows that exposure to low levels of BPA increases the proliferation of fibroid cells.[ref] A study of 495 women who were diagnosed with fibroids found that they had higher concentrations of BPA in them than women without fibroids.[ref]
2-hydroxy-4-methoxybenzophenone, a UV filter found in sunscreens, has links to an increase in fibroids.[ref]
Phthalates, found in plastics, vinyl, and artificial fragrances, act as endocrine-disrupting chemicals and have links to an increased risk of fibroids. This was especially true in women who carried the GSTM1 null genotype.[ref]
In fact, a 2022 study found that higher exposure to phthalates increased the growth of fibroids. The researcher found that phthalate metabolites increased tryptophan and kynurenine levels as well as activated the aryl hydrocarbon receptor in fibroid cells.[ref]
In-utero and infant exposures to DES
Early-onset fibroids are linked to exposure to diethylstilbestrol (DES) in utero. DES, an estrogen medication, was used from 1940 to 1971 for pregnant women. The daughters of women who took DES were at a higher risk for fibroids at a younger age.[ref]
Microplastics:
Microplastics are microscopic particles of plastic that are ubiquitous in our modern food supply, water, and even air. Microplastics are both a source of inflammation (foreign particle) as well as giving off endocrine-disrupting chemicals.
A 2024 study showed that microplastic particles are found in uterine fibroids. The study showed that all 16 samples of fibroids contained microplastics and that there was a higher quantity of microplastics in the myometrium tissue of women with fibroids compared to a control group without fibroids.[ref]
A 2024 preprint study also shows that microplastics are found in the follicular fluid in the ovaries. The study involved 18 women undergoing reproductive help, and found that 75% had significant amounts of microplastic particles in their ovaries.[ref]
Viral infection:
Infection with cytomegalovirus in the uterine tissue leads to an associated 6-fold increase in the risk of fibroids. On the other hand, Chlamydia trachomatis decreased the risk of fibroids.[ref][ref]
Fibroids Genotype Report:
Access this content:
An active subscription is required to access this content.
Lifehacks: Natural solutions for fibroids
Diet changes to prevent fibroids
Women eating >2 servings of fruits per day had a slightly decreased risk of uterine fibroids compared to women eating less than 2 servings per week. A higher intake of animal sources of vitamin A seems linked to a slightly decreased risk of uterine fibroids.[ref]
Another study found that frequent milk and soybean use was linked with an increased risk of fibroids.[ref] Unfortunately, the study lumped both of those together as a category, so I’m not sure which one was important. The implication here is that additional phytoestrogens or bovine hormones could increase fibroids. Since most dairy now has no added hormones, my guess is that the phytoestrogens in soy were the main culprit.
Food additives, sweeteners, and preserved foods have links to an increased risk of fibroids. (Not really helpful since the study doesn’t really narrow down or define those terms.)[ref]
Oral contraceptive use has links to an increased risk of fibroids.[ref] Alternatively, some types of oral contraceptives can also be used to slow menstrual bleeding and decrease the symptoms of fibroids. Talk with your gynecologist on this one.
Avoid phthalates and BPA:
Phthalates are a chemical found in plastics, vinyl, and artificial fragrances (personal care products, air fresheners, and laundry products). They act as estrogen mimics in the body. The level of phthalate metabolites is associated with the size of fibroids – more phthalates in the body = bigger fibroids.[ref][ref]
Similarly, higher BPA levels have links to increased odds of fibroids. BPA can be found in some plastics, household dust, and thermal printed receipts.
What can you realistically do to avoid phthalates and BPA? Check your personal care products for phthalates, and look for laundry products without artificial fragrances included. Regular dusting and vacuuming have been shown to decrease BPA. Handling thermal printed receipts, especially with wet hands, increases BPA fairly significantly in the body.
Related articles: Detoxification genetic pathways of phthalates and BPA
Reducing fibroids with natural supplements:
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
Thyroid Hormones: Genes, Hypothyroidism, and T4/T3 Conversion
References: