Key takeaways:
- Candida is normally harmless, but shifts to a pathogen when the immune balance is disrupted.
- Genetic variants in key immune pathways can strongly influence who is likely to get recurrent or severe Candida infections.
- Candida overgrowth can interact with or exacerbate other conditions, including PANDAS/PANS, fatty liver disease, Alzheimer’s, COPD, IBD, and histamine intolerance.
- Understanding your Candida‑related genetic variants can help tailor prevention strategies and choose the right therapies.
Candida: From Harmless to Opportunistic Invader
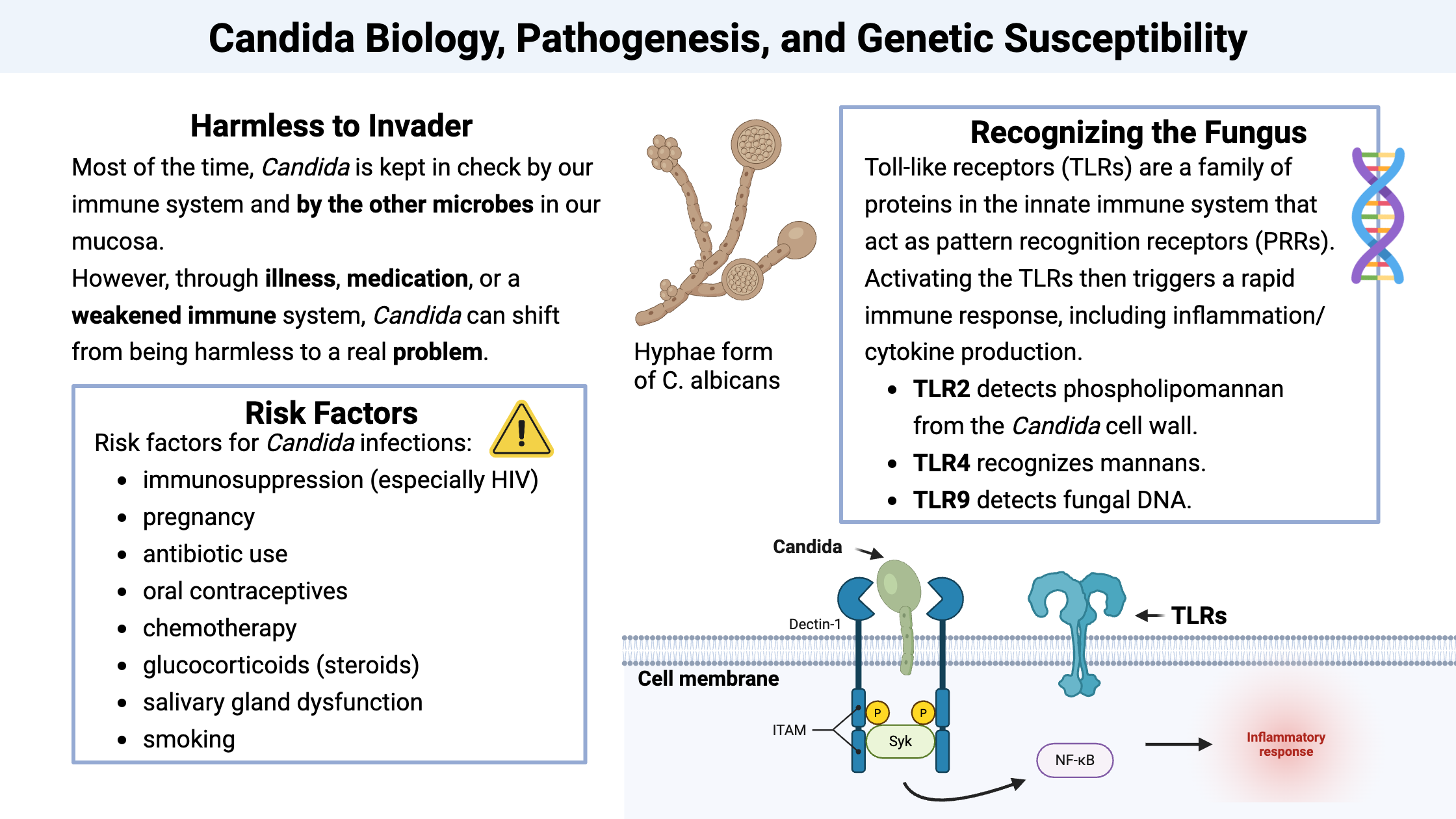
Candida is a fungus that lives in many people’s mucosal surfaces (the mouth, gut, and vagina). Candida species are found in about 30–70% of healthy people at any given time. Most of the time, Candida is kept in check by our immune system and by the other microbes in our mucosa. When everything is in equilibrium, Candida is considered a commensal microbe that can reside on our skin or in our body without causing harm.[ref]
However, when the balance tips, through illness, medication, or a weakened immune system, Candida can shift from being harmless to a real problem.
What do I mean by problem? Candida can cause oral thrush and yeast infections, which are usually mild (even if they don’t feel that way at the time). It can also cause skin or nail changes. At the serious end of the spectrum, Candida can invade the organs or the bloodstream, which has a mortality rate of over 30%.
Let’s dig into the details on Candida infections, and then we will go into how and why your genes can increase susceptibility to chronic or more serious infection. We will finish up in the Lifehacks section with studies on natural ways to treat Candida – coupled with the genetic information to determine what is more likely to work for you.
Candida species and risk factors:
Candida albicans is the most common culprit for a Candida infection — for an overgrowth beyond what the body can handle. Other species like C. glabrata, C. auris, and C. parapsilosis have become more common recently due to widespread antifungal use and are often found in healthcare settings. For example, C. auris was first identified in Japan in 2009, but has now caused outbreaks in over 45 countries and is often resistant to common antifungal drugs.[ref]
Risk factors for Candida infections include:[ref][ref]
- immunosuppression (especially HIV)
- pregnancy
- antibiotic use
- oral contraceptives
- chemotherapy
- glucocorticoids (steroids)
- salivary gland dysfunction, dentures (oral candida)
- smoking
Severe invasive infections tend to occur in people with severely compromised immune systems, including those recovering from surgery, chemotherapy that causes neutropenia, or spending extended time in intensive care.[ref]
How Candida causes damage: Candidalysin toxin and filaments
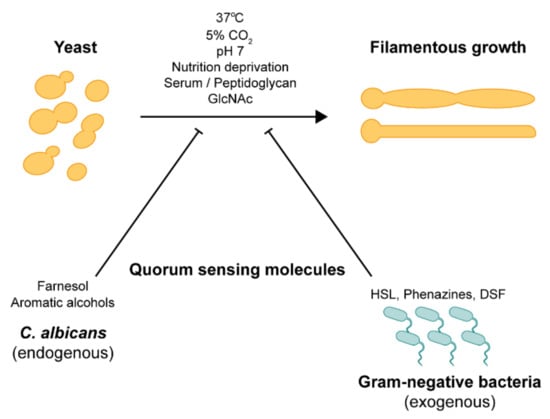
Candida species, and especially C. albicans, can transition between forms, which helps it survive and then cause invasive infections.[ref]
- Blastospores: Single, oval-shaped cells that reproduce asexually by budding, like what you see with yeast when making bread. This form is generally associated with normal, non-invasive (commensal) growth.
- Pseudohyphae: Chains of elongated yeast cells that have failed to detach from the parent cell after budding.
- True Hyphae: These are long, continuous filaments. Hyphae are highly virulent and allow the fungus to invade epithelial cells and damage tissue.
Candida can also produce a toxin called candidalysin. It’s a small peptide that is secreted when the fungus invades host cells in the hyphal form. Essentially, candidalysin punches holes in the cell membranes, causing calcium to flood in, mitochondria to malfunction, and cells to die.
The transition of the yeast form to the hyphae form can happen with a couple of environmental stimuli, including higher temperature, nutrient depletion, high CO2 or low oxygen (hypoxia), and certain bacterial molecules from gram-negative bacteria.[ref]
When candidalysin breaks open cells, it releases zinc and iron, which Candida uses to grow.
Candidalysin isn’t just a problem because it causes cell death; it also triggers a cascade of signaling that leads to the release of inflammatory cytokines. It releases IL-1B, IL-6, and IL-1a — as well as activating the NLRP3 inflammasome.[ref]
The NLRP3 inflammasome then increases inflammatory cytokines to kill off the pathogen. While you want a strong initial inflammatory response, if it doesn’t clear the fungus quickly, elevated NLRP3 activation can be a problem for people with genetic variants in NLRP3 that increase activity. [ref]
In addition to producing the candidalysin toxin, Candida can also secrete extracellular vesicles (EVs) that contain proteins, nucleic acids, and virulence factors that allow them to signal to other fungal cells. These extracellular vesicles can also activate our immune response. [ref]
How your immune system fights Candida:
It’s important to note here that there are multiple ways that the immune system can fight Candida (and most pathogens). There are a lot of genetic variants in the immune system pathways – some increasing response to certain pathogens and some decreasing response to others. This variation means that we all fight pathogens in multiple ways, and slightly differently — with our genetic pathways shaped by the pathogens that our ancestors were exposed to and survived.
| Component | Key functions |
|---|---|
| Pattern recognition | Innate immunity senses fungal cell wall components, initiating the first line of defense. |
| TLRs | TLR2 recognizes phospholipomannan; TLR4 recognizes mannans; TLR9 detects fungal DNA, all driving rapid cytokine/inflammatory responses. |
| CLRs (dectin‑1, etc.) | Dectin‑1 binds β‑glucans and signals via CARD9 to promote Th17 responses; mannose‑binding lectin tags Candida for complement‑mediated clearance. |
| NLRP3 inflammasome | Senses candidalysin‑induced damage, activating IL‑1β/IL‑18 and amplifying inflammation. |
| Macrophages | Engulf and kill Candida using TLRs/lectin receptors; microplastic ingestion impairs their ability to clear fungi and apoptotic cells. |
| Interferon/STING | Candida EVs can trigger type I interferons and engage the STING pathway, classically associated with antiviral defense. |
| Adaptive/Th17 | IL‑17A, IL‑17F, and IL‑22 at mucosal surfaces induce antimicrobial peptides and recruit neutrophils; Th1 (IFN‑γ, IL‑12) supports systemic control. |
| T‑cell status | T‑cell exhaustion increases risk of invasive Candida; effective coordination of innate and adaptive arms is crucial for clearance. |
Let’s dig into each of these in detail.
Innate immune response:
The immune system defense starts with pattern recognition of the components that make up the fungal cell wall. The different fungal wall components activate specific receptors in the innate immune system. (See your immune response genes in the genotype report below.)
Toll-like receptors: Toll-like receptors (TLRs) are a family of proteins in the innate immune system that act as pattern recognition receptors (PRRs). They are expressed on cells like macrophages and dendritic cells, and they identify pathogen-associated molecular patterns (PAMPs), which are carbohydrates or proteins found only on pathogens and not in the body. Activating the TLRs then triggers a rapid immune response, including inflammation/cytokine production.[ref]
- TLR2 detects phospholipomannan from the Candida cell wall.
- TLR4 recognizes mannans.
- TLR9 detects fungal DNA.
C-type lectin receptors (CLRs): This family, including dectin-1, the mannose receptor, dectin-2, and DC-SIGN, has turned out to be the dominant pathway for fungal recognition in humans.
- Dectin-1 is the main receptor for β-glucans and signals through the adaptor CARD9 to drive Th17 immune response. This is especially important for defending mucosal surfaces against Candida.
- Mannose-binding lectin (MBL) helps tag Candida for destruction by the complement system.[ref]
Related article: Mannose-binding lectin deficiency mutations
NLRP3 inflammasomes: The NLRP3 inflammasome acts inside the cell as a second line of detection. It senses the membrane damage caused by candidalysin and activates IL-1β and IL-18 to amplify the immune response.[ref]
Related article: NLRP3 inflammasome genetic variants
Macrophages: Within the immune system, macrophages a type of white blood cell that can engulf and destroy both your own cells that need to be recycled and pathogens, including fungi. Macrophages utilize the toll-like receptors and lectin receptors from above to recognize Candida, engulf it, and destroy it.
Microplastics increase fungal pathogens: A 2026 study on microplastics shows that macrophages also engulf the microscopic plastic particles that are now ubiquitous in our bodies. This then negatively affects the macrophage’s ability to effectively clear out fungal pathogens (and also decreases the clearance of our body’s cells that naturally turn over). [ref]
Related article: Microplastics
Interferon and STING: Type I interferons are rapidly released by immune system cells in response to viral pathogens, and interferon induces genes that inhibit viral reproduction. A relatively new finding is that C. albicans releases extracellular vesicles that stimulate type I interferons (even though they are fungal EVs instead of viral). The STING pathway is also involved here in the activation of interferons.[ref]
Adaptive immune response:
The adaptive immune response also comes into play with Candida. IL-17 and IL-22 in mucosal surfaces (e.g. the gut mucosa or the tongue) can stimulate antimicrobial peptide production. This keeps Candida in check, normally. For systemic infections, Th1 responses driven by IFN-γ and IL-12 take the lead. Whether you clear the infection or it persists depends in large part on how effectively these innate and adaptive arms work together.
T cells: Another type of immune cell, T cells can also play a role in keeping Candida in check. A 2022 study showed that in patients with T cell exhaustion, invasive Candida infections are more common.
Related article: T cell exhaustion
IL-17 and adaptive immune response: Following innate recognition, the adaptive immune response kicks in. For mucosal defense against Candida, Th17 response, which involves the production of IL-17A, IL-17F, and IL-22, is important. These cytokines stimulate epithelial cells to produce antimicrobial peptides and recruit neutrophils to sites of infection. [ref]
Related article: IL-17 genes
Connections with other neurological or immune system conditions:
While Candida infections can occur as standalone issues, such as in babies with thrush (oral candida infection), a lot of the time, Candida exacerbates other conditions.
Here are some of the overlapping conditions where Candida may play a role:
PANDAS or PANS – Pediatric Acute-onset Neuropsychiatric Syndrome (PANDAS) is a neurological condition in children that is caused by activation of the immune system, attacking neuronal cells. This results in neurological symptoms, including anxiety, depression, oppositional behavior, and sensory abnormalities in children. It often follows a bacterial infection.
Autism behavior changes – Candida infections and subsequent inflammation are associated with ‘flares’ of behavior changes in children with autism.[ref]
Fatty liver disease – NAFLD or MASLD, as it is now called, involves inflammation in the liver and IL-17A. A 2025 study found that liver fibrosis in fatty liver disease can involve fungal species, including C. albicans.[ref]
Related article: Fatty liver disease (MASLD)
Alzheimer’s – Candida is found in some amyloid-beta plaques in Alzheimer’s disease.[ref] If the fungi reach the brain via a systemic infection and a disrupted blood-brain barrier, Candida can cause glial granulomas that produce amyloid-beta plaque.[ref]
Related article: APOE and Alzheimer’s risk
Food intolerance and histamine reactions – Candidaovergrowth in the gastrointestinal tract increases food intolerances and mast cell release of histamine.[ref]
Related articles: Mast cell activation syndrome & Histamine Intolerance
Anxiety and mood changes – Animal studies show that the reason Candida overgrowth can cause anxiety or mood disorders is that it alters the gut-brain axis and alters cortisol levels. Candida changes the endocannabinoid system by decreasing AEA levels.[ref]
Related articles: Anxiety genes & HPA axis (cortisol)
Muscle and joint pain – While uncommon, some fungal infections, including Candida overgrowth, can cause musculoskeletal inflammation and pain.[ref]
COPD – Acute exacerbations in COPD can be triggered by a number of factors, including bacterial or fungal infections. A 2025 study in COPD patients showed that 97% had fungi in their lung samples, with Candida being the dominant species during exacerbations.[ref]
Inflammatory bowel diseases – Some patients with IBD have an aggravated response to the extracellular vesicles released by C. albicans.[ref]
Related article: IBD genes
Why genes matter: Not everyone responds the same way
Even among people with similar risk factors, such as immunosuppression from corticosteroids, some people are naturally more susceptible to Candida infections than others. Genetic variations, both rare mutations and common SNPs, play a significant role in susceptibility. For example, rare mutations that cause primary immunodeficiency can increase the risk of many pathogenic fungal infections.
When looking at fungal infections – or pathogens in general – a strong initial inflammatory response can be beneficial in preventing the infection. However, if the pathogen is able to take hold and multiply, then that excessive inflammatory response can cause problems associated with chronic inflammation.
Genotype report: Candida
Access this content:
An active subscription is required to access this content.
Lifehacks for Candida
Diet and foods to reduce Candida:
Candida diet: There are a lot of popular articles on what to eat to avoid Candida overgrowth. Most of the point to avoid sugar and gluten.
While there aren’t a lot of studies showing that diet matters for Candida, there are studies showing that the gut microbiome balance is important. Focusing on whole foods, vegetables, and fruits, and avoiding processed foods with added sugar is a good general goal for a balanced gut microbiome. Some sites recommend avoiding dairy due to the lactose (milk sugar).[ref] Note that everyone is different in what makes their gut microbiome happy and balanced, so the advice here is pretty general. Some people may do best with a high fiber, high vegetable diet, while others have a balanced gut with lower fiber.
Coconut oil: Studies show that coconut oil may have an inhibitory effect on Candida growth and biofilm formation.[ref][ref] Incorporating coconut oil into your diet may help a big with preventing Candida overgrowth.
Garlic: Garlic contains several antimicrobial components, and studies show that it decreases the growth of C. albicans.[ref]
Gut microbiome and Candida:
When the gut microbiome is balanced, Candida is much less likely to become problematic with overgrowth or biofilm formation.
Probiotics may help:
- A meta-analysis of clinical trials showed that people who were taking probiotics were at an almost 50% decreased risk of oral Candida overgrowth.[ref]
- Beneficial probiotics include Lactobacillus and Bifidobacteria [ref]
- L. rhamnosus GG has been shown in studies to interfere with Candida growth and hyphae formation.[ref]
Natural supplements for Candida infections
Quite a few natural, plant-based supplements have been studied for their effect on Candida. Please be sure to consult your doctor if you have questions about whether a supplement is right for you – especially if you are already on prescription medications that may interact.
Berberine:
Berberine has significant antifungal activity against Candida species, including drug-resistant strains in vitro. It works by disrupting Candida cell walls and inhibiting biofilm formation by interfering with nutrient absorption.[ref]
Access this content:
An active subscription is required to access this content.
Related articles and topics:
Epstein-Barr Virus: Genetic Risks, Reactivation, and Chronic Illnesses
References: