Key takeaways:
- For most people, type 2 diabetes is due to a combination of lifestyle and genetic risk factors.
- Knowing your root cause can help you prioritize lifestyle changes that may work to prevent, manage, or even reverse type 2 diabetes.
Genetic factors interact with environmental factors: Type 2 diabetes can have different causes, with different pathways that impact glucose regulation. Tailoring your diabetes prevention (or reversal) efforts to fit your genetic susceptibility may be more effective. For example, people with melatonin receptor variants are at a higher risk of diabetes when eating later at night. You can prioritize changes based on your genetic variants.
Adding up the genetic risk:
Researchers have discovered many different genes that increase the relative risk of diabetes. The genetic variants listed below increase (or decrease) the risk of diabetes in most population groups. These variants are well-studied with many replication studies.
For the most part, genetic variants increase the relative risk of diabetes a little bit, often only increasing relative risk by 10-40%. Thus, a single variant may not impact your risk all that much. Instead, the cumulative effects of multiple risk variants seem to be key. Research shows us that, in many cases, the risk is additive. In other words, the more risk alleles you carry, the higher the risk.[ref][ref]
Diet combines with Genetic Risk Score:
Research shows that a ‘Western Diet’ is associated with an increased risk of diabetes only in people with several genetic risk variants. The Western dietary pattern did not have much of an impact on the risk of diabetes in people without the risk variants.[ref]
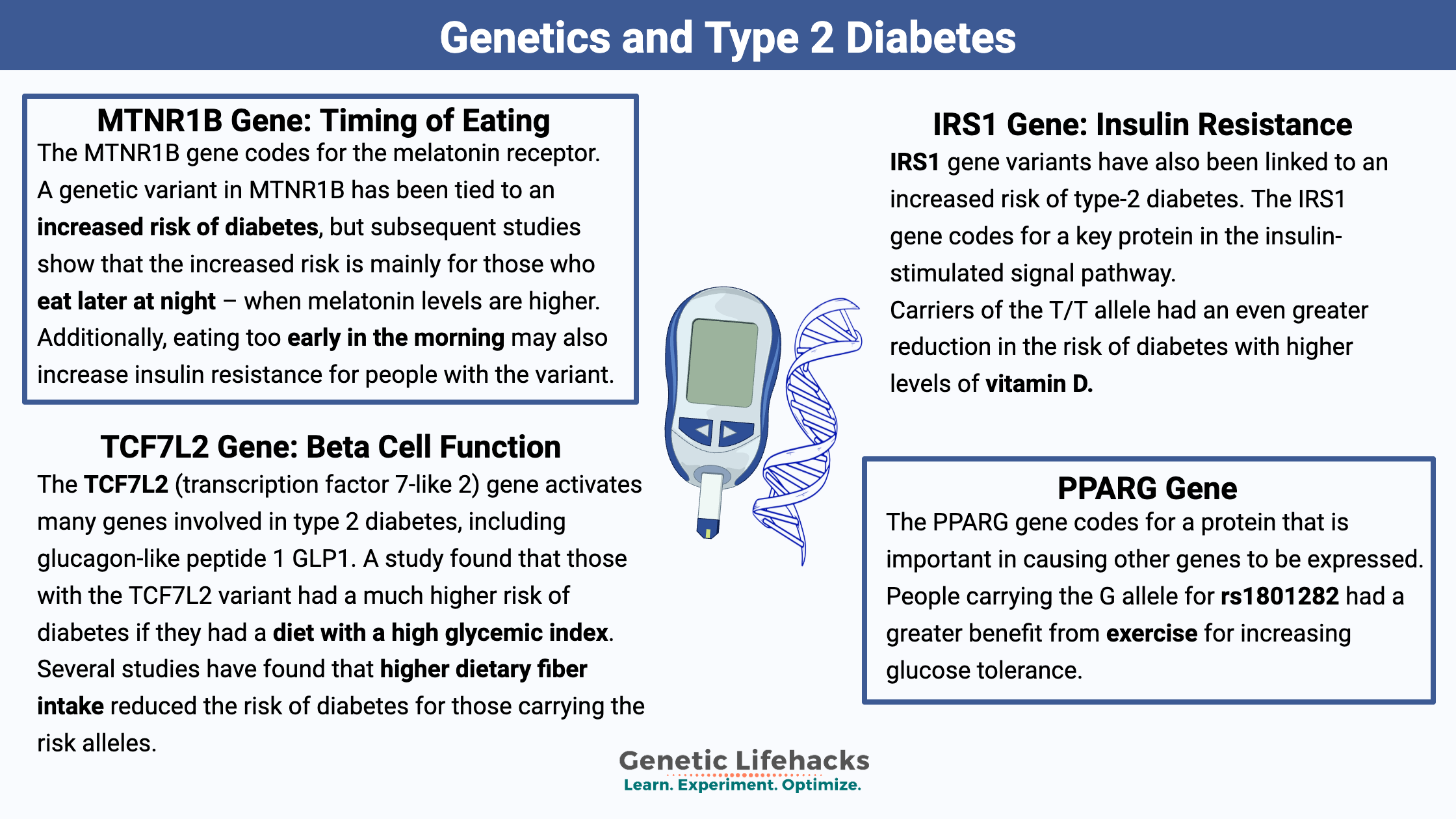
Genetic Variants Linked to Diabetes:
Below are some of the most well-researched genetic variants linked to diabetes. If your genotype matches the risk allele, read through the details below the chart for each variant.
Access this content:
An active subscription is required to access this content.
Related Articles:
Resveratrol: Studies, Genetic Interactions, and Bioavailability
Glutamate: Synthesis, transport, and supplement interactions
Gut Mucosal Barrier & Mucin Genes: Diet and DNA Shape Your Gut Defense
The information provided here is for educational and informational purposes only. Please consult with your doctor for any medical advice.