This article explains the current research (August 2021) on natural compounds that may help to combat SARS-CoV-2. This round-up of research is simply for educational purposes — please read the studies, talk with your doctor, and make informed decisions. While there are other natural compounds with theoretical efficacy, I’ve chosen only to include the plant compounds that have human clinical trials here.
Related article: Research roundup: Preventing and Mitigating Covid-19
Flavonoids as antiviral agents:
Since ancient times, humans have used medicinal plants for illnesses. Flavonoids are a class of phytochemicals that are found in foods, spices, and medicinal herbs. Researchers have published hundreds of studies on using flavonoids as antivirals for SARS-CoV-2. Most of the research articles explain the mechanism of action for the flavonoid. Importantly though, there are now several placebo-controlled clinical trials showing efficacy for certain natural compounds.[ref]
This article focuses on both the mechanism of why the natural compound has antiviral properties as well as human clinical trials.
Quercetin for prevention and/or treatment
Quercetin is an active phytochemical found in a number of different traditionally used medicinal plants. It has many anti-inflammatory properties.
Theoretical research :
Quite a few in silco (computer) molecular docking studies have identified quercetin as an inhibitor of SARS-CoV-2 through binding to the spike protein.[ref][ref][ref][ref]
Cell studies also show that quercetin inhibits SARS-CoV-2.[ref]
Prevention clinical trial:
A 20-week clinical trial (200 participants) looked at the efficacy of a combo of zinc, quercetin, vitamin D, vitamin C, and l-lysine for preventing SARS-CoV-2. 15% of the control group ended up testing positive for COVID-19, while none in the quercetin/zinc/D/C arm tested positive.[ref]
A trial in healthcare workers looked at the effect of quercetin (500 mg/2x per day) plus vitamin C on Covid risk. This was a fairly small trial. One healthcare worker in the quercetin arm of the trial tested positive for SARS-CoV-2, while 9 workers in the control group (same size groups) tested positive. Importantly here, all but one of the 9 workers who tested positive were symptomatic and likely contagious.
From the clinical trial results:[ref]
“During the follow-up period between March 20 and June 20, 1 out of 71 healthcare workers using QCT and 9 out of 42 healthcare workers in the control group had COVID-19”
Treatment clinical trial:
A (small) randomized clinical trial of patients with mild COVID-19 showed that patients taking quercetin cleared the virus more quickly. One week of quercetin also diminished symptoms in all trial participants, compared to only 20% of patients having symptoms reduced in the standard of care arm of the trial.[ref]
An Italian clinical trial (~150 people) found that 1000 mg of quercetin reduced the risk of hospitalization in COVID-19 patients. Only 9% of the patients in the quercetin arm of the study needed hospitalization, compared to 29% of the patients not taking quercetin. Of note, none of the patients taking quercetin ended up in the ICU, while 8 patients in the stand care arm of the trial needed ICU care.[ref] Keep in mind that this is a fairly small study. While the results are impressive, a larger trial is needed to confirm.
Related article: Quercetin – scientific studies and genetic connections
(Note: interaction with COMT genetic variants)
Nigella Sativa plus Honey
Nigella sativa, also known as Black Seed or Black Cumin, is a medicinal plant native to the Middle East. Interestingly, hundreds of studies on Nigella sativa show that it may help with pathogenic infections (viral, bacterial, fungal), diabetes, cardiovascular disease, and inflammatory disorders.[ref][ref]
In many countries, Nigella sativa seed and honey have traditionally been used together for medicinal purposes. Honey has many antibacterial and immune system modulatory properties.
Treatment Clinical trial:
A recent placebo-controlled randomized clinical trial involving 313 COVID-19 patients in Pakistan looked at the effectiveness of Nigella sativa plus honey compared to a placebo. The patients who received Nigella sativa plus honey were sick for a shorter time than those in the placebo arm of the trial. The Nigella sativa plus honey group patients had a 50% shorter duration of illness, both for moderate and severe illnesses.[ref]
Here is a chart showing the difference in the number of days for symptoms to go away (orange is the group taking Nigella sativa plus honey):
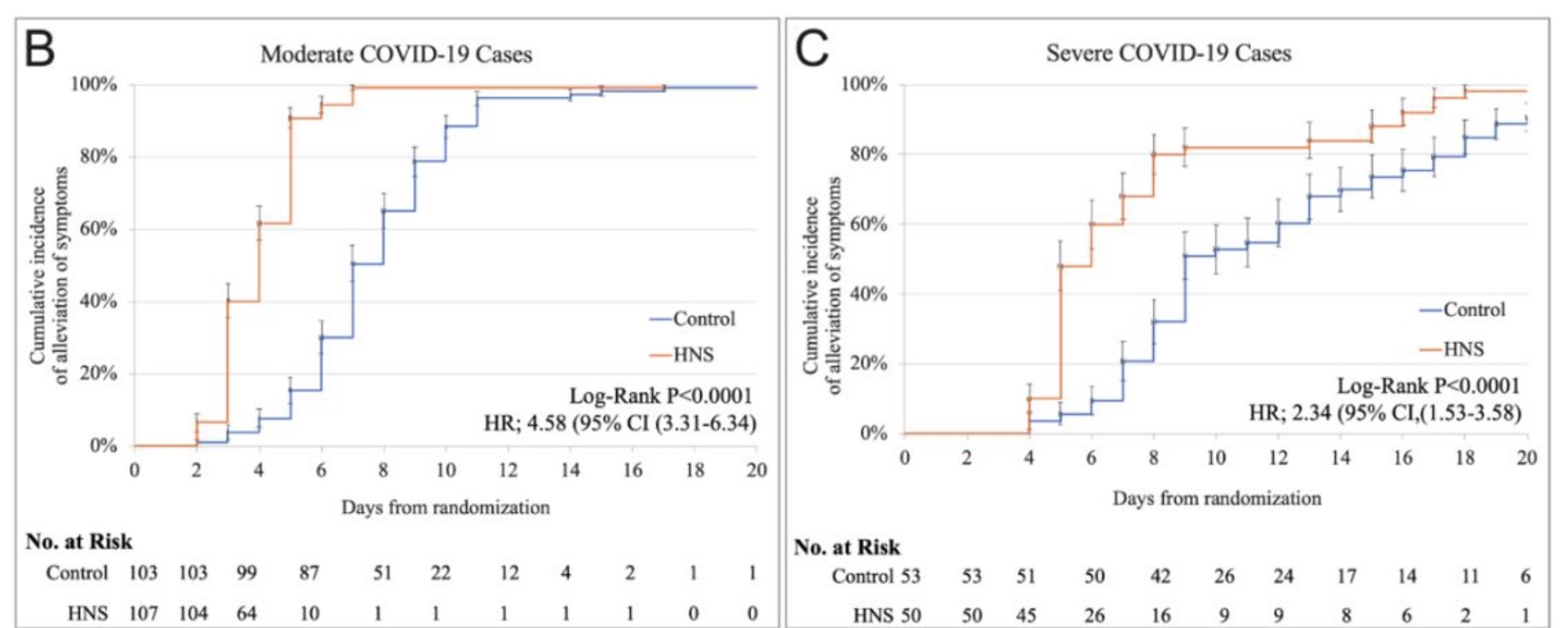
Theoretical research:
Previously, an in silco (computer models) study had shown several of the active phytoconstituents of Nigella sativa to interact with SARS-CoV-2. Specifically, six of the phytoconstituents were predicted to bind to the N-terminal binding domain.[ref]
Other in silco studies have shown that several phytoconstituents of Nigella sativa may be effective both in blocking replication and in decreasing the ability of the virus to enter cells.[ref]
Within Nigella sativa there are several different active natural compounds that seem to have an effect as an anti-viral. Dithymoquinone, a component of Nigella sativa, seems to be the most effective in terms of SARS-CoV-2. [ref]
Is Nigella sativa safe? Talk with your doctor, of course, if you have any questions or are on medications. Otherwise, Nigella sativa seeds have been eaten for thousands of years and are considered non-toxic. A review of many clinical trials that used Nigella sativa showed no serious side effects.[ref]
Curcumin as an antiviral
A component of turmeric, curcumin is a flavonoid with many antiviral and anti-inflammatory properties. Turmeric is traditionally used as a spice in Indian foods and in Ayurvedic and traditional Chinese medicine.
Theoretical research:
A multi-omics approach identified curcumin as one of the top 30 ‘drugs’ to fight SARS-CoV-2.[ref] Other in silco studies point to curcumin as being antiviral and a protease inhibitor (preventing replication).[ref]
Curcumin has been shown to prevent infection from other enveloped viruses, including RSV and the flu.[ref] Studies in the original SARS virus showed it may be effective, and studies on acute respiratory distress syndrome and sepsis show that curcumin may modulate an excessive immune response.[ref]
Additionally, research shows that curcumin directly binds to the S protein.[ref] You may be wondering if taking curcumin at the same time as getting the mRNA vaccine would decrease the efficacy… I can’t find the answer to this question.
Treatment clinical trial:
A small clinical trial (41 participants) found that curcumin decreased the length of stay in the hospital. It also improved oxygen levels by day 2.[ref]
A placebo-controlled clinical trial (n=140) in India used 525 mg of curcumin with piperine twice a day. The results showed that the COVID-19 patients who received the curcumin supplement had earlier symptom recovery and less deterioration than the control group. Patients in the curcumin arm of the trial with mild COVID were 6-fold less likely to need oxygen therapy.[ref]
A triple-blind randomized placebo-controlled clinical trial with mild to moderate covid patients tested curcuminoids in nanomicelles. The group receiving the curcuminoids had symptoms resolve faster including the return of taste and smell.[ref]
Finally, a small clinical trial using nano-curcumin showed that it decreased both IL-6 and IL-1B (inflammatory cytokines) levels in patients ill with COVID-19.[ref]
Related article: Supplement interaction with COMT genetic variant
Conclusion:
While a number of plant compounds theoretically may help with COVID-19, the natural supplements above were tested in clinical trials.
Keep in mind, the trials were small and definitely need to be reproduced, but the safety profile on these supplements is long.
Talk with your doctor or pharmacist about interactions with any prescription medications you are taking.
With the current Delta variant and the reduction in vaccine efficacy, we all need to do what we can to stay healthy. For me, these natural compounds are one tool in my toolbox for helping my immune system fight COVID-19. Supplements aren’t the only tool, though. Metabolic health, good sleep, and other measures are also important.
Read more: Research roundup: Preventing and mitigating COVID-19
Full Reference List
Ahmadi, Reza, et al. “Oral Nano-Curcumin Formulation Efficacy in the Management of Mild to Moderate Outpatient COVID-19: A Randomized Triple-Blind Placebo-Controlled Clinical Trial.” Food Science & Nutrition, vol. 9, no. 8, 2021, pp. 4068–75. Wiley Online Library, https://doi.org/10.1002/fsn3.2226.
Arslan, Bengu, et al. Synergistic Effect of Quercetin and Vitamin C Against COVID-19: Is a Possible Guard for Front Liner. 2020. Europe PMC, https://europepmc.org/article/PPR/PPR239932.
Ashraf, Sohaib, et al. Honey and Nigella Sativa against COVID-19 in Pakistan (HNS-COVID-PK): A Multi-Center Placebo-Controlled Randomized Clinical Trial. 30 Nov. 2020, p. 2020.10.30.20217364. medRxiv, https://www.medrxiv.org/content/10.1101/2020.10.30.20217364v4.
Barh, Debmalya, et al. “Multi-Omics-Based Identification of SARS-CoV-2 Infection Biology and Candidate Drugs against COVID-19.” Computers in Biology and Medicine, vol. 126, Nov. 2020, p. 104051. PubMed Central, https://doi.org/10.1016/j.compbiomed.2020.104051.
Di Pierro, Francesco, et al. “Potential Clinical Benefits of Quercetin in the Early Stage of COVID-19: Results of a Second, Pilot, Randomized, Controlled and Open-Label Clinical Trial.” International Journal of General Medicine, vol. 14, 2021, pp. 2807–16. PubMed, https://doi.org/10.2147/IJGM.S318949.
—. “Potential Clinical Benefits of Quercetin in the Early Stage of COVID-19: Results of a Second, Pilot, Randomized, Controlled and Open-Label Clinical Trial.” International Journal of General Medicine, vol. 14, 2021, pp. 2807–16. PubMed, https://doi.org/10.2147/IJGM.S318949.
Ibrahim, Mahmoud A. A., et al. “In Silico Drug Discovery of Major Metabolites from Spices as SARS-CoV-2 Main Protease Inhibitors.” Computers in Biology and Medicine, vol. 126, Nov. 2020, p. 104046. PubMed Central, https://doi.org/10.1016/j.compbiomed.2020.104046.
Jena, Atala B., et al. “Catechin and Curcumin Interact with S Protein of SARS-CoV2 and ACE2 of Human Cell Membrane: Insights from Computational Studies.” Scientific Reports, vol. 11, no. 1, Jan. 2021, p. 2043. www.nature.com, https://doi.org/10.1038/s41598-021-81462-7.
Kandeil, Ahmed, et al. “Bioactive Polyphenolic Compounds Showing Strong Antiviral Activities against Severe Acute Respiratory Syndrome Coronavirus 2.” Pathogens (Basel, Switzerland), vol. 10, no. 6, June 2021, p. 758. PubMed, https://doi.org/10.3390/pathogens10060758.
Khan, Jishan, et al. “Identification of Potential Phytochemicals from Citrus Limon against Main Protease of SARS-CoV-2: Molecular Docking, Molecular Dynamic Simulations and Quantum Computations.” Journal of Biomolecular Structure & Dynamics, July 2021, pp. 1–12. PubMed, https://doi.org/10.1080/07391102.2021.1947893.
Khazeei Tabari, Mohammad Amin, et al. “Flavonoids as Promising Antiviral Agents against SARS-CoV-2 Infection: A Mechanistic Review.” Molecules, vol. 26, no. 13, June 2021, p. 3900. PubMed Central, https://doi.org/10.3390/molecules26133900.
—. “Flavonoids as Promising Antiviral Agents against SARS-CoV-2 Infection: A Mechanistic Review.” Molecules, vol. 26, no. 13, June 2021, p. 3900. PubMed Central, https://doi.org/10.3390/molecules26133900.
Koshak, Dr Abdulrahman E., and Prof Emad A. Koshak. “Nigella Sativa L as a Potential Phytotherapy for Coronavirus Disease 2019: A Mini Review of in Silico Studies.” Current Therapeutic Research, vol. 93, Jan. 2020, p. 100602. ScienceDirect, https://doi.org/10.1016/j.curtheres.2020.100602.
Maintz, Laura, and Natalija Novak. “Histamine and Histamine Intolerance.” The American Journal of Clinical Nutrition, vol. 85, no. 5, May 2007, pp. 1185–96. Silverchair, https://doi.org/10.1093/ajcn/85.5.1185.
Malekmohammad, Khojasteh, and Mahmoud Rafieian-Kopaei. “Mechanistic Aspects of Medicinal Plants and Secondary Metabolites against Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2).” Current Pharmaceutical Design, July 2021. PubMed, https://doi.org/10.2174/1381612827666210705160130.
Margolin, Leon, et al. “20-Week Study of Clinical Outcomes of Over-the-Counter COVID-19 Prophylaxis and Treatment.” Journal of Evidence-Based Integrative Medicine, vol. 26, July 2021, p. 2515690X211026193. PubMed Central, https://doi.org/10.1177/2515690X211026193.
Moon, Debbie. “COMT and Supplement Interactions.” Genetic Lifehacks, 27 Apr. 2021, https://www.geneticlifehacks.com/comt-and-supplement-interactions/.
Pawar, Kirti S., et al. “Oral Curcumin With Piperine as Adjuvant Therapy for the Treatment of COVID-19: A Randomized Clinical Trial.” Frontiers in Pharmacology, vol. 12, 2021, p. 1056. Frontiers, https://doi.org/10.3389/fphar.2021.669362.
Praditya, Dimas, et al. “Anti-Infective Properties of the Golden Spice Curcumin.” Frontiers in Microbiology, vol. 10, May 2019, p. 912. PubMed Central, https://doi.org/10.3389/fmicb.2019.00912.
Saber-Moghaddam, Niloofar, et al. “Oral Nano-Curcumin Formulation Efficacy in Management of Mild to Moderate Hospitalized Coronavirus Disease-19 Patients: An Open Label Nonrandomized Clinical Trial.” Phytotherapy Research, vol. 35, no. 5, 2021, pp. 2616–23. Wiley Online Library, https://doi.org/10.1002/ptr.7004.
Salehi, Bahare, et al. “Nigella Plants – Traditional Uses, Bioactive Phytoconstituents, Preclinical and Clinical Studies.” Frontiers in Pharmacology, vol. 12, Apr. 2021, p. 625386. PubMed Central, https://doi.org/10.3389/fphar.2021.625386.
Siddiqui, Sahabjada, et al. “Virtual Screening of Phytoconstituents from Miracle Herb Nigella Sativa Targeting Nucleocapsid Protein and Papain-like Protease of SARS-CoV-2 for COVID-19 Treatment.” Journal of Biomolecular Structure & Dynamics, pp. 1–21. PubMed Central, https://doi.org/10.1080/07391102.2020.1852117.
Tavakkoli, Alireza, et al. “Review on Clinical Trials of Black Seed (Nigella Sativa ) and Its Active Constituent, Thymoquinone.” Journal of Pharmacopuncture, vol. 20, no. 3, Sept. 2017, pp. 179–93. PubMed, https://doi.org/10.3831/KPI.2017.20.021.
Thimmulappa, Rajesh K., et al. “Antiviral and Immunomodulatory Activity of Curcumin: A Case for Prophylactic Therapy for COVID-19.” Heliyon, vol. 7, no. 2, Feb. 2021, p. e06350. PubMed Central, https://doi.org/10.1016/j.heliyon.2021.e06350.
Umar, Abd Kakhar. “Flavonoid Compounds of Buah Merah (Pandanus Conoideus Lamk) as a Potent SARS-CoV-2 Main Protease Inhibitor: In Silico Approach.” Future Journal of Pharmaceutical Sciences, vol. 7, no. 1, 2021, p. 158. PubMed, https://doi.org/10.1186/s43094-021-00309-0.
Valizadeh, Hamed, et al. “Nano-Curcumin Therapy, a Promising Method in Modulating Inflammatory Cytokines in COVID-19 Patients.” International Immunopharmacology, vol. 89, no. Pt B, Dec. 2020, p. 107088. PubMed, https://doi.org/10.1016/j.intimp.2020.107088.
Yimer, Ebrahim M., et al. “Nigella Sativa L. (Black Cumin): A Promising Natural Remedy for Wide Range of Illnesses.” Evidence-Based Complementary and Alternative Medicine : ECAM, vol. 2019, May 2019, p. 1528635. PubMed Central, https://doi.org/10.1155/2019/1528635.

