What is non-living and doesn’t have DNA –but yet, it can infect, replicate, and eventually kill you? The answer… a simple type of protein called a prion.
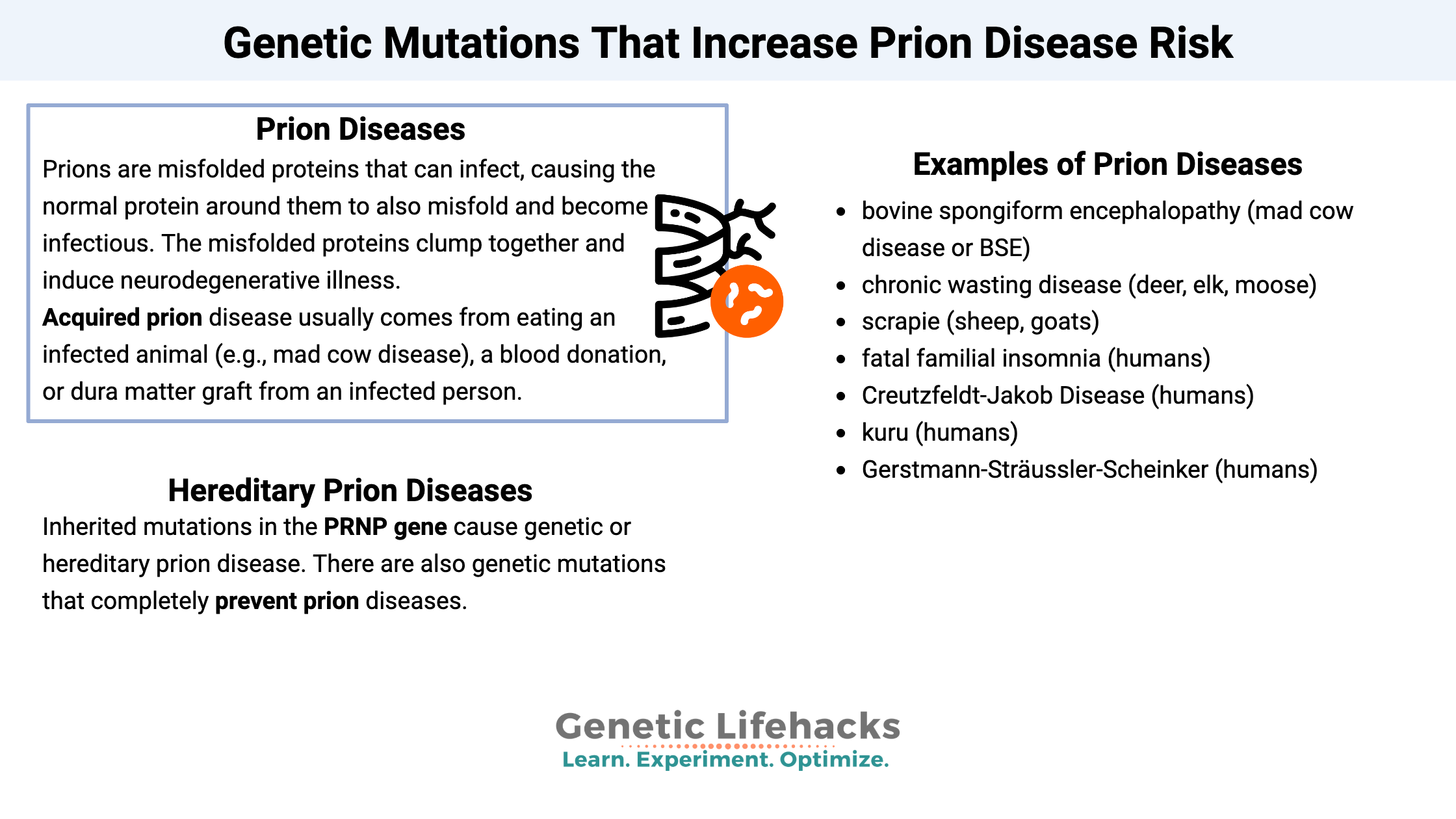
Prions are a difficult concept to grasp. They’re misfolded protein molecules, but not in the same way other proteins misfold. Prions can infect, causing the normal protein around them to also misfold and become infectious. The misfolded proteins clump together and induce neurodegenerative illness.
Prions, unlike bacteria, viruses, and other pathogens, do not contain DNA or RNA, but they can spread in an infectious manner. Instead of replicating, the misfolded proteins travel around the body, causing normal proteins to misfold.
In this article, I will explain some of the background science on prions and examine how genetic mutations are involved. Finally, I’ll explore some current theories linking Alzheimer’s and Parkinson’s as prion-related diseases.
History and background: Prion diseases
The word prion comes from “proteinaceous infectious particle”.
Although prions were recognized and isolated for the first time in the early 1980s, animal variants of the disease have been known since antiquity. Prions aren’t limited to animals, though. Plants, fungi, and yeast have all been discovered to have prions in recent years.[ref][ref]
Examples of prion diseases include:
- bovine spongiform encephalopathy (mad cow disease or BSE)
- chronic wasting disease (deer, elk, moose)
- scrapie (sheep, goats)
- fatal familial insomnia (humans)
- Creutzfeldt-Jakob Disease (humans)
- kuru (humans)
- Gerstmann-Sträussler-Scheinker (humans)
Prions cause fatal, neurodegenerative diseases.
That is a sentence that strikes fear in the hearts of nearly everyone. Human prion disease symptoms tend to strike during mid-life – anywhere from age 28 to 70.
They can arise from eating an animal infected with the disease, and prion diseases can also arise spontaneously or due to genetic mutations.
Creutzfeldt-Jakob Disease (CJD) is a fatal neurodegenerative disease caused by cell death due to prions in the brain. It causes progressive neurodegenerative problems such as tremors, problems with movement, changes in personality, and dementia.[ref]
CJD can have a variety of causes:
- Sporadic CJD arises without a known cause (74% – 90% of cases)
- CJD can be caused by inheriting a genetic mutation (<15% of cases)
- Acquired CJD can be transferred by exposure (<6%)[ref][ref]
Acquired prion disease usually comes from eating an infected animal (e.g., mad cow disease), a blood donation, or dura matter graft from an infected person. (CDC info on CJD)
Brain matter infected with prions cannot be rendered safe by normal means.
Cooking and disinfectants do not destroy prions. Some measures render medical equipment safe after possible prion exposure, which has almost eliminated the transfer of prion disease through surgery.
All of this from a misfolded protein…
Why is protein folding important?
Misfolded protein – somehow, this term brings to mind my failed attempts at origami.
Let me back up a bit and give you some context…
Your genes code for proteins, which make up your body’s structure and functions. Proteins form structural elements in cells, making up the receptors on cell surfaces. The enzymes that cause the biochemical reactions to happen inside cells are proteins.
Proteins are assembled from 20 different amino acids (in humans and most animals).
But they aren’t just long straight chains of amino acids; the order of the amino acids also controls how the protein folds up onto itself. The importance of protein folding is crucial to its function. Therefore, the protein doesn’t function correctly if it isn’t completed correctly. For example, a protein may need a hydrophobic region sticking out to anchor it to the cell membrane. Without being folded correctly, it may not be able to anchor to the right spot.
Quick analogy: Think of Lego blocks as your amino acids. You could take those blocks and make them into a truck — or — a dinosaur. The blocks are the same, but they are assembled differently. Any 4-year-old can tell you that the truck is not the same as a dinosaur.
With proteins, different conformations change or eliminate their function.
Proteins are degraded and recycled back into their amino acid components all the time. How long a protein exists in a cell depends on the protein, but the half-life can be from minutes to years. Most proteins hang around for a few days and then degrade.
Misfolded proteins often happen in a cell. They usually are unstable and break down quickly.
It isn’t true of prions… Prion proteins are structurally stable and resist denaturing (being broken down). They stick around even though they aren’t functioning correctly. This stability, plus their ability to cause normal proteins to also misfold, is the crux of the problem.
Here is a good article on protein folding if you want to know more.
What do prions do in the brain and elsewhere?
The protein that makes up the prions is coded for by the PRNP gene. The normal PRNP protein exists throughout all tissues in the body but is found at higher concentrations in the brain. The physiological functions of the PRNP protein in its normal form include myelin maintenance, circadian rhythm regulation, cellular differentiation, immune regulation, copper utilization, iron uptake and transport, and possibly a role in memory formation.[ref] Oddly, with all those different functions, the PRNP protein is one that we (and animals) can live without.
Research also points to the PRNP protein playing a role in stopping viral replication. It is thought to be part of the brain’s immune response to viruses.[ref] A 2021 animal study found that a neurotropic strain of influenza A could induce the conversion of normal PRNP proteins into prions.[ref]
When the PRNP protein misfolds, it becomes a prion. This misfolding could be due to an inherited mutation, which arose when DNA damage happened to a cell or through the ingestion of tissue from an infected animal.
These misfolded but stable prions can recruit normally folded PRNP to become misfolded. Since they don’t break down and clear out, they can build up in the brain and nervous tissue. This lack of clearance eventually causes cell death and tissue damage in the brain.
When looking at samples of brain tissue infected with a prion disease, it has small holes in it and a spongy appearance.
It takes many months to several decades for enough of the misfolded proteins to build up. It is an exponential growth curve that is slow to start but then builds quickly at the end.
Evolutionary biologists explain that prions are a way that genetically identical cells can have diverse phenotypes. Essentially, proteins can pass along information without using traditional means of inheritance such as RNA or DNA. For example, prions in yeasts allow identical cells to adapt to environmental changes.[ref]
Hereditary prion diseases:
Inherited mutations in the PRNP gene cause genetic or hereditary prion disease. Between 1 to 2 people per million are diagnosed each year with this rare disease.[ref]
Not only are there mutations that cause familial CJD, but there are also genetic mutations that completely prevent prion diseases.[ref]
Traditionally, hereditary prion diseases were thought to be passed from parent to child, with the offspring having a 50/50 chance of getting the disease.
But wait… the science here has gotten a little more interesting on this topic since the advent of inexpensive genetic testing.
A 2016 study examined the number of individuals who carried a PRNP mutation in two different data sets. One data set had 60,000 people, and the other was 500,000+ people from 23andMe. The study investigated the number of PRNP pathogenic mutations carried in the populations. One big part of this was to see how penetrance for a disease carries out in a population. Inherited mutations in the PRNP gene were always thought to cause prion diseases.
The study results showed that prion mutations were about 30 times more common in the genetic data than expected.[ref] So why do some people carry the mutation that supposedly causes CJD, yet don’t seem to have the disease? That is one question researchers are still trying to answer.
Another question researchers raise is whether other neurodegenerative diseases are a type of prion disease.
Alzheimer’s and Parkinson’s Diseases. A link to prions?
For the past few years, researchers have been bringing together the idea that Alzheimer’s and Parkinson’s are prion-like diseases.
A 2015 article in the journal Science highlighted and explained the overlap between prion disease and common neurogenerative diseases.[ref]
Research now points to Alzheimer’s and Parkinson’s disease being similar to prion disease in a lot of ways. Both diseases are caused by abnormal proteins causing damage to the brain.
Similar to Creutzfeldt-Jakob Disease (CJD), most Alzheimer’s and Parkinson’s disease cases are sporadic – not caused by rare mutations in the APP or SNCA genes. Both diseases are caused by a build-up of insoluble proteins in the brain over decades. Both are neurodegenerative diseases that progress without a cure.
However, one big difference is that Alzheimer’s and Parkinson’s diseases are not considered transmissible or contagious, although some animal studies question this. Infectious sources of CJD (e.g., eating cow brains infected with mad cow disease) show that it can pass from one source to another. However, not everyone who eats infected meat (cow, deer, etc.) gets the disease. Infectiousness is considered a hallmark of prion diseases.
The evidence cited for tau protein tangles in AD being similar to prions includes that they “spread along neural pathways from one brain region to another”. Additionally, the α-synuclein protein in Parkinson’s forms Lewy bodies that “spreads along neural pathways in the brain, beginning in the dorsal motor nucleus of the glossopharyngeal and vagal nerves, the olfactory bulb, and the anterior olfactory nucleus”. And mouse studies show that injecting α-synuclein from Lewy-body dementia patients into the mice brains causes the formation of mouse α-synuclein inclusions. [ref]
There are genetic connections between Alzheimer’s and Creutzfeldt-Jakob Disease. A genetic variant in the PRNP (prion protein) gene has decreased the risk of CJD and Alzheimers. Not all studies back up the connection, though.[ref][ref] Additionally, a variant in the protein that cleaves the amyloid precursor protein increases the risk of CJD.[ref]
Some researchers speculate that prions or prion-like proteins could be at the root of all neurodegenerative diseases. In fact, recent research showed Alzheimer’s like amyloid-beta plaque and tau tangles in 6 out of 8 brain autopsy results of people who had Creutzfeldt-Jacob disease. The results were remarkable because the brains were from people who died in middle age – long before Alzheimer’s pathology should ever appear in the brain.[ref]
Recently, research has linked the α-synuclein protein that causes Parkinson’s with the pathology of Alzheimer’s disease. Over half of the brain autopsies of people with AD also show high concentrations of α-synuclein aggregates. And 90% of people with the early-onset hereditary forms of AD have aggregated α-synuclein deposits in the amygdala. But while these co-exist and perhaps enhance the pathology, it has also been found in about 25% of normal brain autopsy reports that individuals without dementia had Lewy body or Alzheimer’s pathology in their brain.[ref]
Intriguingly, animal studies show that deleting the PRNP gene prevents the hereditary, early-onset form of Alzheimer’s.[ref]
Prion Diseases Genotype Report:
Access this content:
An active subscription is required to access this content.
Lifehacks:
There are no magic cures or supplements to prevent or cure prion diseases, but you can take precautions by not eating prion-containing meat. There are laws now governing the handling and feeding of cows, as well as lots of testing in place to prevent the spread of mad cow disease. To be super cautious, avoid eating beef brains.
For hunters, this means being aware of whether chronic wasting disease is prevalent in your area if you are eating deer, elk, or moose meat.
In some areas of the US and Canada, testing your meat before consuming it should be a priority. https://cpw.state.co.us/learn/Pages/ResearchCWD.aspx
You can learn more and support prion research through the Prion Alliance, a 501(3)(c) non-profit run by researchers at the Broad Institute.
Manganese and Prions:
The prion protein binds to copper and other metals, such as manganese. High levels of manganese are found in the brains of people with CJD. Manganese is essential at low levels, but neurotoxic at higher levels – causing Parkinson-like symptoms and destruction of the dopaminergic brain regions. Animal studies show that manganese chelation therapy can extend the life of animals with prion disease.[ref][ref]
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
Alzheimer’s and APOE genotype
One very important gene and extremely well researched for Alzheimer’s disease is the APOE gene. This gene carries cholesterol and other fats in your bloodstream, and a common variant of the gene is strongly linked to a higher risk of Alzheimer’s.
Genetic variants that increase the risk of Parkinson’s disease
Parkinson’s disease is a neurological disorder caused by the degradation of dopamine-producing neurons in a part of the brain called the substantia nigra. It affects up to 10 million people worldwide.
Essential Tremor: Causes and Possible Solutions
Essential tremor (ET) is a neurological disease causing involuntary shaking in places of the body while performing tasks such as writing or eating. Learn more about this disease and discover the genetic causes of ET.
Boosting NAD+ to Reverse Aging? Overview of NR and NMN
Explore the research about how nicotinamide riboside (NR) and NMN are being used to reverse aging. Learn about how your genes naturally affect your NAD+ levels and how this interacts with the aging process.
References:
Calero, Olga, et al. “A Common BACE1 Polymorphism Is a Risk Factor for Sporadic Creutzfeldt-Jakob Disease.” PLoS ONE, vol. 7, no. 8, Aug. 2012, p. e43926. PubMed Central, https://doi.org/10.1371/journal.pone.0043926.
Chapman, J., et al. “Fatal Insomnia in a Case of Familial Creutzfeldt-Jakob Disease with the Codon 200(Lys) Mutation.” Neurology, vol. 46, no. 3, Mar. 1996, pp. 758–61. PubMed, https://doi.org/10.1212/wnl.46.3.758.
“Colorado Parks and Wildlife.” Colorado Parks and Wildlife, https://cpw.state.co.us. Accessed 15 June 2022.
Das, Alvin S., and Wen-Quan Zou. “Prions: Beyond a Single Protein.” Clinical Microbiology Reviews, vol. 29, no. 3, July 2016, pp. 633–58. PubMed Central, https://doi.org/10.1128/CMR.00046-15.
Del Bo, Roberto, et al. “Is M129V of PRNP Gene Associated with Alzheimer’s Disease? A Case-Control Study and a Meta-Analysis.” Neurobiology of Aging, vol. 27, no. 5, May 2006, p. 770.e1-770.e5. PubMed, https://doi.org/10.1016/j.neurobiolaging.2005.05.025.
Hara, Hideyuki, and Suehiro Sakaguchi. “Virus Infection, Genetic Mutations, and Prion Infection in Prion Protein Conversion.” International Journal of Molecular Sciences, vol. 22, no. 22, Nov. 2021, p. 12439. PubMed, https://doi.org/10.3390/ijms222212439.
Kim, Yong-Chan, and Byung-Hoon Jeong. “The First Meta-Analysis of the M129V Single-Nucleotide Polymorphism (SNP) of the Prion Protein Gene (PRNP) with Sporadic Creutzfeldt-Jakob Disease.” Cells, vol. 10, no. 11, Nov. 2021, p. 3132. PubMed, https://doi.org/10.3390/cells10113132.
Lathe, Richard, and Jean-Luc Darlix. “Prion Protein PRNP: A New Player in Innate Immunity? The Aβ Connection.” Journal of Alzheimer’s Disease Reports, vol. 1, no. 1, pp. 263–75. PubMed Central, https://doi.org/10.3233/ADR-170037. Accessed 15 June 2022.
—. “Prion Protein PRNP: A New Player in Innate Immunity? The Aβ Connection.” Journal of Alzheimer’s Disease Reports, vol. 1, no. 1, pp. 263–75. PubMed Central, https://doi.org/10.3233/ADR-170037. Accessed 15 June 2022.
Loh, Doris, and Russel J. Reiter. “Melatonin: Regulation of Prion Protein Phase Separation in Cancer Multidrug Resistance.” Molecules, vol. 27, no. 3, Jan. 2022, p. 705. PubMed Central, https://doi.org/10.3390/molecules27030705.
Mead, Simon, et al. “Genome-Wide Association Study in Multiple Human Prion Diseases Suggests Genetic Risk Factors Additional to PRNP.” Human Molecular Genetics, vol. 21, no. 8, Apr. 2012, pp. 1897–906. PubMed Central, https://doi.org/10.1093/hmg/ddr607.
Minikel, Eric Vallabh, et al. “Quantifying Penetrance in a Dominant Disease Gene Using Large Control Cohorts.” Science Translational Medicine, vol. 8, no. 322, Jan. 2016, p. 322ra9. PubMed Central, https://doi.org/10.1126/scitranslmed.aad5169.
NM_000311.4(PRNP):C.314C>T (p.Pro105Leu) AND Gerstmann-Straussler-Scheinker Syndrome – ClinVar – NCBI. https://www.ncbi.nlm.nih.gov/clinvar/RCV000014343.25/. Accessed 15 June 2022.
NM_000311.5(PRNP):C.598G>A (p.Glu200Lys) AND Genetic Prion Disease – ClinVar – NCBI. https://www.ncbi.nlm.nih.gov/clinvar/RCV000020253.1/. Accessed 15 June 2022.
Nozaki, Ichiro, et al. “Prospective 10-Year Surveillance of Human Prion Diseases in Japan.” Brain: A Journal of Neurology, vol. 133, no. 10, Oct. 2010, pp. 3043–57. PubMed, https://doi.org/10.1093/brain/awq216.
Prion Alliance. http://www.prionalliance.org/. Accessed 15 June 2022.
Prion Disease: MedlinePlus Genetics. https://medlineplus.gov/genetics/condition/prion-disease/. Accessed 15 June 2022.
Risk for Travelers | Variant Creutzfeldt-Jakob Disease, Classic (CJD) | Prion Disease | CDC. 13 Oct. 2021, https://www.cdc.gov/prions/vcjd/risk-travelers.html.
Salazar, Santiago V., et al. “Conditional Deletion of Prnp Rescues Behavioral and Synaptic Deficits after Disease Onset in Transgenic Alzheimer’s Disease.” The Journal of Neuroscience, vol. 37, no. 38, Sept. 2017, pp. 9207–21. PubMed Central, https://doi.org/10.1523/JNEUROSCI.0722-17.2017.
Twohig, Daniel, and Henrietta M. Nielsen. “α-Synuclein in the Pathophysiology of Alzheimer’s Disease.” Molecular Neurodegeneration, vol. 14, June 2019, p. 23. PubMed Central, https://doi.org/10.1186/s13024-019-0320-x.
Vollmert, C., et al. “Significant Association of a M129V Independent Polymorphism in the 5′ UTR of the PRNP Gene with Sporadic Creutzfeldt‐Jakob Disease in a Large German Case‐control Study.” Journal of Medical Genetics, vol. 43, no. 10, Oct. 2006, p. e53. PubMed Central, https://doi.org/10.1136/jmg.2006.040931.
Wise, Jacqui. “Naturally Occurring Genetic Variation That Prevents Prion Disease Is Identified.” BMJ, vol. 350, June 2015, p. h3186. www.bmj.com, https://doi.org/10.1136/bmj.h3186.
https://cen.acs.org/articles/95/i31/Protein-folding-Much-intricate-thought.html. Accessed 15 June 2022.