Key takeaways:
~ Age-related macular degeneration (AMD) is the most common cause of blindness in the elderly.
~ Genetics and lifestyle factors are both important in AMD.
~ Importantly, genetic variants also impact which supplements for AMD are likely to be helpful and which could be doing more harm than good.
What causes macular degeneration?
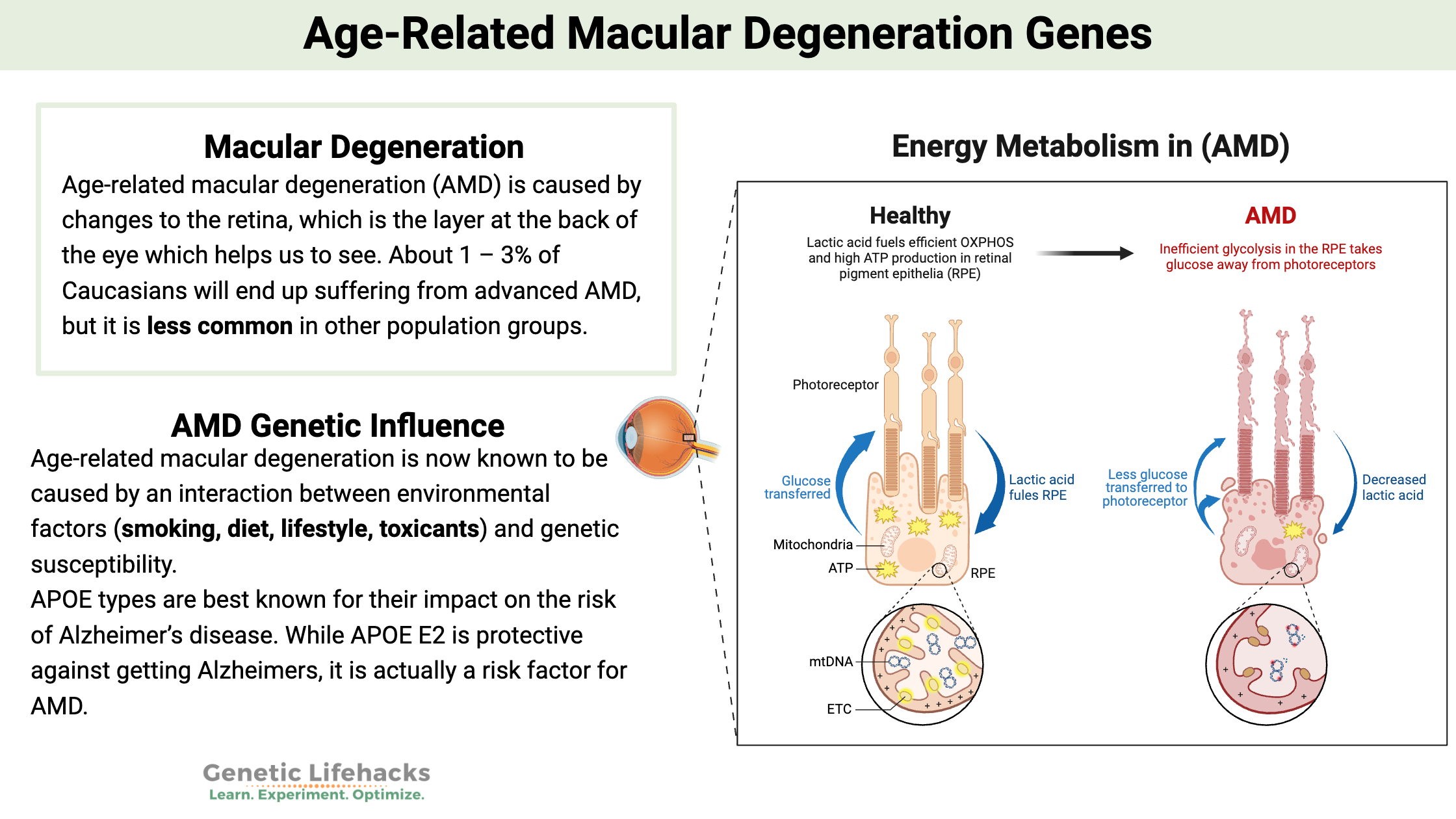
Age-related macular degeneration (AMD) is caused by changes to the retina, which is the layer at the back of the eye that helps us to see. Specifically, the changes happen in the center of the retina, known as the macula.
AMD is the leading cause of vision loss in older people. About 1 – 3% of Caucasians will end up suffering from advanced AMD, but it is less common in other population groups.
Macular degeneration causes a loss of central vision. It can start as a blurry spot in the middle of the field of vision, progressing to a dark spot. Straight lines may look distorted, and colors may be darker or less vivid.
Wet and Dry Age-related macular degeneration:
There are two types of AMD – wet and dry:
Dry age-related macular degeneration is caused by metabolic end products collecting under the top layer of the retina.
- These buildups are called drusen.
- It leads to scarring and thinning of the retina.
- Dry AMD causes a gradual loss of central vision.
- Dry AMD is also called geographic atrophy
Wet age-related macular degeneration is caused by abnormal, leaky blood vessels growing into the retina after the formation of the drusen.
- The abnormal blood vessels cause swelling or bleeding in the retina.
- Wet AMD is also called neovascular AMD and occurs in about 10-20% of cases.
- Wet AMD usually is linked with a more rapid loss of vision, but in certain cases, it can be gradual.
- There are several new treatments that can slow the progression of wet AMD, including injectable medications that decrease swelling and blood vessel formation.
~ AMD is a leading cause of vision loss in older adults, affecting the macula, the central part of the retina.
~ There are two main types: dry (atrophic) and wet (neovascular) AMD.
~ Symptoms include blurry or distorted central vision and difficulty reading or recognizing faces.
Let’s dig into how and why the immune system attacks the eye and causes AMD.
How the complement system genetic variants increase AMD risk:
Age-related macular degeneration is now known to be caused by an interaction between environmental factors (smoking, diet, lifestyle, and toxicants) and genetic susceptibility.
Genetic studies from the early 2000s pointed researchers in the direction of the complement system, which is part of our immune system.
The complement system enhances (or complements :-) the ability of antibodies and phagocytic cells to attack microbes — and also to clear out damaged cells.
The outermost layer of the retina is the retinal pigment epithelium. This layer is important for moving ions and nutrients (e.g., vitamin A, glucose) in and out of the layer that contains the photoreceptors needed for vision.
AMD originates from cell death in the photoreceptor and retinal pigment epithelium cells. This causes holes or dark spots in vision.[ref]
What causes cell death for the photoreceptors and/or epithelial cells?
The body has various ways of killing cells that it thinks are pathogenic. The complement system acts as one of the known ways in this process.
Activation of the complement system can occur in a variety of different ways, including the lectin pathway (e.g., mannose-binding lectin), classical pathway, the alternative pathway, or through the intrinsic pathway (C3, C5).
Complement factor H (CHF) is an inhibitor or regulator of the complement cascade. Its role is to regulate the complement system so that it attacks pathogens instead of damaging your own cells. (Your own cells have complement factor H, but bacterial cell surfaces don’t.)
Activating the complement pathway results in the formation of something called a membrane attack complex, which causes the rupture of a cell membrane. Great when it is attacking a bacterial pathogen, but not great when it happens to the retinal pigment epithelium cells.
So everything needs to be in balance here. While there are a variety of ways to activate the complement pathway, complement factor H keeps the complement system from being overly active.[ref]
Completely unique in the body, the retinal pigment epithelium has the ability to create and express a variety of different complement system proteins normally only created in the liver. Basically, the eye has something called ‘immune privilege’, meaning that the body’s normal immune response (via the bloodstream) is not very active there. Instead, the eye has its own separate immune response.
Back to AMD and the complement system:
A buildup of cellular oxidative stress due to smoking or simply aging causes an increased amount of oxidative stress and cellular debris. This causes inflammatory signaling to occur, activating the complement system.
Too much complement activation causes damage to the retina. Thus, excessive cellular stress increases inflammation and complement activation causing damage when the complement system becomes overly activated.
Research shows that higher levels of complement activation occur in eyes with age-related macular degeneration.[ref][ref]
This over-activation can be due to not having enough of the ‘stop’ protein, called complement factor H. Additionally, complement factor H interacts with complement factor I, so low complement factor I can also be important in halting the over-activation of the complement system. Or the over-activation can be due to variants that increase the natural activity of the complement system.
When retinal epithelial cells are destroyed, they can’t grow back. At least not very quickly or easily. So the key is to keep the oxidative stress in retinal cells at a lower level, thus preventing the complement cascade from being activated in the eye.
~ Oxidative stress can damage retinal cells, activating the complement system.
~ Without enough complement factor H to put out the ‘stop’ sign, the retinal cells can be destroyed.
APOE and Macular Degeneration:
APOE types are best known for their impact on the risk of Alzheimer’s disease. APOE E2 is protective against getting Alzheimer’s, while APOE E4 significantly increases the risk of Alzheimer’s. The APOE E3 type is neutral for Alzheimer’s risk.
Studies show that while APOE E2 is beneficial for Alzheimer’s prevention, it is actually a risk factor for AMD, increasing the relative risk by 80%. On the other hand, people with APOE E4 alleles are at about a 30% decreased risk for macular degeneration.[ref]
Using a mouse model of the three APOE types, researchers have figured out that ” APOE2 provokes and APOE4 inhibits the cardinal AMD features, inflammation, degeneration, and exaggerated neovascularization.” The APOE E4 allele decreases APOE and CCL2 in the retina, while the E2 allele increases APOE and increases retinal inflammatory cells.[ref]
Age-related Macular Degeneration Genotype Report:
The heritability of AMD is estimated to be as high as 70% for advanced AMD.[ref][ref] Thus, genetics (along with lifestyle) plays an important role in the susceptibility and progression of macular degeneration.
Keep in mind when reading through any study or article about genetics and AMD that the studies are talking about increased (or decreased) relative risk. If the lifetime risk for a Caucasian who is elderly is 2% or 1 in 50, then doubling that risk makes it 1 in 25.
Access this content:
An active subscription is required to access this content.
Lifehacks:
If genetics plays such a big role, is there anything you can do about AMD?
Absolutely! Knowing your genetic risk can help you determine which diet and lifestyle changes you can make at a younger age to prevent AMD. While you can’t change your genes, you can reduce your risk by controlling modifiable factors: don’t smoke, eat a healthy diet rich in leafy greens and fish, exercise, and have regular eye exams
Lifestyle factors:
In addition to age, other factors such as cigarette smoking, UV and blue light exposure, being female, lighter-colored eyes, cardiovascular disease, and obesity can all increase the risk of AMD.[ref] Some of these you can’t change, but making efforts to reduce blue light exposure, stay at a healthy weight, and take care of your heart can all be important.
Smoking:
One genetic study points out that the link between the ARMS2 variant and AMD is particularly risky for smokers or former smokers.[ref]
Cataract surgery: likely not a problem
A number of studies have looked at the link between cataract surgery and an increased risk of macular degeneration. Most of the large studies find no increase in risk from cataract surgery.[ref][ref] There are a couple of early studies, though, that did find a link between cataract surgery and late AMD.[ref] Questions remain here as to whether it is surgery that caused a link to AMD vs. lifestyle factors that increased both cataracts and AMD.
AREDS2 Supplement trial: Preventing Progression of AMD
The AREDS2 trial looked at the effect of supplementing with specific antioxidants, vitamins, and minerals in preventing the progression of AMD. Overall, the trial showed some success, reducing progression by 19%.[ref]
The AREDS2 trial supplement contained:
- 80 mg zinc
- 2 mg copper
- 500 mg vitamin C
- 400 IU vitamin E
- 10 mg lutein
- 2 mg zeaxanthin
The new AREDS3 version includes B vitamins and a lot less zinc. One issue with the AREDS2 formula, with 80 mg of zinc, is that it exceeded the NIH’s recommended upper limit for zinc (40 mg of zinc).
While you will often see an AREDS supplement suggested for preventing macular degeneration, that may not be the right approach for everyone.
Antioxidant supplements may make AMD worse for people with ARMS2 variants:
One study found that for people who carry the ARMS2 risk allele, there was a deleterious response – a worsening of macular degeneration – in people who took antioxidant supplements (vitamins E, C, lutein, and zeaxanthin). For people with the ARMS2 variants, a zinc supplement alone without added antioxidants was beneficial.[ref]
One question here is whether it is one specific antioxidant, such as vitamin E, that causes people with the ARMS2 risk allele to be at a greater risk.
Other studies, though, don’t show that antioxidants are harmful, necessarily, for people with the ARMS2 variants — but they do back up the idea that zinc is most helpful.[ref]
Antioxidant supplements help with CFH variants:
For patients with no ARMS2 variants but with CFH variants, antioxidant supplements (along with adequate zinc) decreased the progression of AMD.[ref][ref]
Recap of the supplement response and genetic variant interactions:
| Gene/Variant | Risk Impact | Supplement Response |
|---|---|---|
| CFH | ↑ AMD risk | Antioxidants helpful |
| ARMS2 | ↑ AMD risk | Zinc helpful, antioxidants may harm |
| APOE E2 | ↑ AMD risk | Not supplement-specific |
| APOE E4 | ↓ AMD risk | Not supplement-specific |
Natural Supplements that protect against macular degeneration (plus interactions with genes):
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
References: