will see their genotype report below and the solutions in the Lifehacks section.
MC4R, or melanocortin 4 receptor, is a receptor in the brain that is part of how appetite is regulated and energy is balanced. When activated by alpha-melanocyte-stimulating hormone (𝛼-MSH), MC4R promotes the satiety signal (feeling full) and increases your energy expenditure. Mutations in the MC4R that cause a deficiency in the receptor are linked to obesity due to hyperphagia.
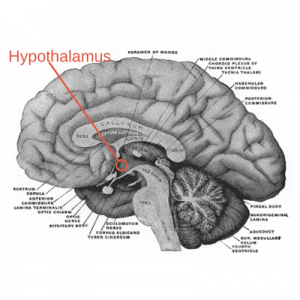
MC4R is part of the leptin pathway. Activating MC4R causes a decrease in food intake along with an increase in energy expenditure, thus decreasing body weight.[ref]
The arcuate nucleus is a region within the hypothalamus in our brain. This cluster of nerve cells has several important functions, including controlling appetite.
The, the normal way things work is that you eat, you’re full for a while, and then your body signals that you should eat again.
There are hundreds of research studies on the genetic variants of MC4R, and most of the variants studied show a decrease in MC4R, resulting in increased BMI. If there aren’t enough of the receptors generated by the cell, then the ‘stop eating’ signal can be impaired. Thyroid hormone levels also play a role in the production of MC4R.[ref]
Hormones, such as leptin, signal the brain to produce α-MSH (alpha-melanocyte-stimulating hormone), which is a peptide hormone involved in appetite, skin pigmentation, and inflammation.
In the central nervous system, α-MSH is cleaved from proopiomelanocortin (POMC). It is then released into the hypothalamus and binds to MC4R receptor-expressing neurons, decreasing food intake and increasing energy expenditure.[ref] α-MSH is derived from the POMC hormone, which is a pituitary gland hormone that is the precursor to ACTH and other MSHs.
However, another molecule called the agouti-signaling peptide can also bind to the MC4R receptor, blocking it from binding with α-MSH. The agouti-signaling peptide can be thought of as an ‘off switch’, with α-MSH being the ‘on switch’.
MC4R deficiency is not only tied to increased appetite and higher BMI throughout life, but also to greater muscle mass. In my mind, the term ‘big-boned’ leaps to mind, which is backed up by studies showing the relation to bone mass, fat mass, and the MC4R variants.[ref][ref]
While obesity is always included as a risk factor for heart disease, it turns out that people who are obese due to genetic mutations in MC4R have lower cholesterol and triglyceride levels and a lower risk of cardiovascular disease. Researchers found that after a high-fat meal, the rise in lipid levels is lower in people with MC4R mutations than normal.[ref]
The α-MSH hormone does more than just bind to the MC4R receptor and moderate feeding, it also stimulates the production of pigment (melanin) in the hair and skin. Named for the agouti coloring of animal coats, the agouti-related peptide also plays a role in pigmentation.
In studies of MC4R genetic variants in childhood obesity, activity offsets the propensity toward higher BMI. In other words, more active children with the variant were less likely to be obese than sedentary children with the variant.[ref] While I can see some of you mentally saying ‘duh’, the point is that while some kids can be sedentary and thin, not everyone is made that way. So this is one more reason for encouraging (not nagging or bullying!) kids to be more active.
Anderson, Olivia S., et al. “Epigenetic Responses Following Maternal Dietary Exposure to Physiologically Relevant Levels of Bisphenol A.” Environmental and Molecular Mutagenesis, vol. 53, no. 5, June 2012, pp. 334–42. PubMed, https://doi.org/10.1002/em.21692.
Apalasamy, Yamunah Devi, et al. “Association of Melanocortin-4 Receptor Gene Polymorphisms with Obesity-Related Parameters in Malaysian Malays.” Annals of Human Biology, vol. 40, no. 1, Jan. 2013, pp. 102–06. PubMed, https://doi.org/10.3109/03014460.2012.720709.
Arble, Deanna M., et al. “The Melanocortin-4 Receptor Integrates Circadian Light Cues and Metabolism.” Endocrinology, vol. 156, no. 5, May 2015, pp. 1685–91. PubMed Central, https://doi.org/10.1210/en.2014-1937.
Bayhaghi, Giti, et al. “Descriptive Analysis of MC4R Gene Variants Associated with Obesity Listed on ClinVar.” Science Progress, vol. 107, no. 4, Nov. 2024, p. 00368504241297197. PubMed Central, https://doi.org/10.1177/00368504241297197.
Been, Latonya F., et al. “Replication of Association between a Common Variant near Melanocortin-4 Receptor Gene and Obesity-Related Traits in Asian Sikhs.” Obesity (Silver Spring, Md.), vol. 18, no. 2, Feb. 2010, pp. 425–29. PubMed, https://doi.org/10.1038/oby.2009.254.
Cauchi, Stéphane, et al. “Combined Effects of MC4R and FTO Common Genetic Variants on Obesity in European General Populations.” Journal of Molecular Medicine (Berlin, Germany), vol. 87, no. 5, May 2009, pp. 537–46. PubMed, https://doi.org/10.1007/s00109-009-0451-6.
Chambers, John C., et al. “Common Genetic Variation near MC4R Is Associated with Waist Circumference and Insulin Resistance.” Nature Genetics, vol. 40, no. 6, June 2008, pp. 716–18. PubMed, https://doi.org/10.1038/ng.156.
Heid, Iris M., et al. “Association of the MC4R V103I Polymorphism with the Metabolic Syndrome: The KORA Study.” Obesity (Silver Spring, Md.), vol. 16, no. 2, Feb. 2008, pp. 369–76. PubMed, https://doi.org/10.1038/oby.2007.21.
Huang, Tao, et al. “Dietary Protein Modifies the Effect of the MC4R Genotype on 2-Year Changes in Appetite and Food Craving: The POUNDS Lost Trial.” The Journal of Nutrition, vol. 147, no. 3, Mar. 2017, pp. 439–44. PubMed, https://doi.org/10.3945/jn.116.242958.
Knüppel, Sven, et al. “Evaluation of 41 Candidate Gene Variants for Obesity in the EPIC-Potsdam Cohort by Multi-Locus Stepwise Regression.” PloS One, vol. 8, no. 7, 2013, p. e68941. PubMed, https://doi.org/10.1371/journal.pone.0068941.
Kong, Xiaomu, et al. “Obesity-Related Genomic Loci Are Associated with Type 2 Diabetes in a Han Chinese Population.” PLoS ONE, vol. 9, no. 8, Aug. 2014, p. e104486. PubMed Central, https://doi.org/10.1371/journal.pone.0104486.
Kvaløy, K., et al. “Sex-Specific Effects of Weight-Affecting Gene Variants in a Life Course Perspective–The HUNT Study, Norway.” International Journal of Obesity (2005), vol. 37, no. 9, Sept. 2013, pp. 1221–29. PubMed, https://doi.org/10.1038/ijo.2012.220.
Lim, Jane Jia Xin, et al. “Diverse Obesity Trajectories in a Family Including Identical Twins with a Pathogenic <italic>MC4R</Italic> Variant.” Obesity Facts, vol. 18, no. 6, 2025, pp. 646–55. PubMed, https://doi.org/10.1159/000546795.
Liu, Gaifen, et al. “Common Variants near MC4R Are Associated with General and Visceral Adiposity in European- and African-American Youth.” The Journal of Pediatrics, vol. 156, no. 4, Apr. 2010, pp. 598-605.e1. PubMed Central, https://doi.org/10.1016/j.jpeds.2009.10.037.
Loos, Ruth J. F., et al. “Common Variants near MC4R Are Associated with Fat Mass, Weight and Risk of Obesity.” Nature Genetics, vol. 40, no. 6, June 2008, pp. 768–75. PubMed Central, https://doi.org/10.1038/ng.140.
Nguyen, Long The, et al. “Maternal High-Fat Diet Induces Metabolic Stress Response Disorders in Offspring Hypothalamus.” Journal of Molecular Endocrinology, vol. 59, no. 1, July 2017, pp. 81–92. PubMed, https://doi.org/10.1530/JME-17-0056.
Obesity, Epigenetics, and Gene Regulation | Learn Science at Scitable. http://www.nature.com/scitable/topicpage/obesity-epigenetics-and-gene-regulation-927. Accessed 4 Mar. 2026.
Petry, Clive J., et al. “Association between a Common Variant near MC4R and Change in Body Mass Index Develops by Two Weeks of Age.” Hormone Research in Paediatrics, vol. 73, no. 4, 2010, pp. 275–80. PubMed, https://doi.org/10.1159/000284392.
Pokushalov, Evgeny, et al. “The Impact of Glucomannan, Inulin, and Psyllium Supplementation (SolowaysTM) on Weight Loss in Adults with FTO, LEP, LEPR, and MC4R Polymorphisms: A Randomized, Double-Blind, Placebo-Controlled Trial.” Nutrients, vol. 16, no. 4, Feb. 2024, p. 557. PubMed Central, https://doi.org/10.3390/nu16040557.
Qi, Lu, et al. “The Common Obesity Variant near MC4R Gene Is Associated with Higher Intakes of Total Energy and Dietary Fat, Weight Change and Diabetes Risk in Women.” Human Molecular Genetics, vol. 17, no. 22, Nov. 2008, pp. 3502–08. PubMed Central, https://doi.org/10.1093/hmg/ddn242.
Rajcsanyi, Luisa Sophie, et al. “Unexpected Identification of Obesity-Associated Mutations in LEP and MC4R Genes in Patients with Anorexia Nervosa.” Scientific Reports, vol. 14, Mar. 2024, p. 7067. PubMed Central, https://doi.org/10.1038/s41598-024-57517-w.
Rojo, Daniela, et al. “Mouse Models for V103I and I251L Gain of Function Variants of the Human MC4R Display Decreased Adiposity but Are Not Protected against a Hypercaloric Diet.” Molecular Metabolism, vol. 42, Sept. 2020, p. 101077. PubMed Central, https://doi.org/10.1016/j.molmet.2020.101077.
Speliotes, Elizabeth K., et al. “Association Analyses of 249,796 Individuals Reveal Eighteen New Loci with Body Mass Index.” Nature Genetics, vol. 42, no. 11, Nov. 2010, pp. 937–48. PubMed Central, https://doi.org/10.1038/ng.686.
Stutzmann, Fanny, et al. “Non-Synonymous Polymorphisms in Melanocortin-4 Receptor Protect against Obesity: The Two Facets of a Janus Obesity Gene.” Human Molecular Genetics, vol. 16, no. 15, Aug. 2007, pp. 1837–44. PubMed, https://doi.org/10.1093/hmg/ddm132.
Timpson, Nicholas J., et al. “How Does Body Fat Influence Bone Mass in Childhood? A Mendelian Randomization Approach.” Journal of Bone and Mineral Research: The Official Journal of the American Society for Bone and Mineral Research, vol. 24, no. 3, Mar. 2009, pp. 522–33. PubMed, https://doi.org/10.1359/jbmr.081109.
VCV000014336.61 – ClinVar – NCBI. https://www.ncbi.nlm.nih.gov/clinvar/variation/14336/. Accessed 4 Mar. 2026.
Wu, Qiwen, et al. “Alpha-Melanocyte-Stimulating Hormone-Mediated Appetite Regulation in the Central Nervous System.” Neuroendocrinology, vol. 113, no. 9, 2023, pp. 885–904. PubMed, https://doi.org/10.1159/000530804.
Xi, Bo, et al. “Influence of Physical Inactivity on Associations between Single Nucleotide Polymorphisms and Genetic Predisposition to Childhood Obesity.” American Journal of Epidemiology, vol. 173, no. 11, June 2011, pp. 1256–62. PubMed, https://doi.org/10.1093/aje/kwr008.
Zhang, Youjin, et al. “The Rs17782313 Polymorphism near MC4R Gene Confers a High Risk of Obesity and Hyperglycemia, While PGC1α Rs8192678 Polymorphism Is Weakly Correlated with Glucometabolic Disorder: A Systematic Review and Meta-Analysis.” Frontiers in Endocrinology, vol. 14, 2023, p. 1210455. PubMed, https://doi.org/10.3389/fendo.2023.1210455.
Zobel, Dorit P., et al. “Variants near MC4R Are Associated with Obesity and Influence Obesity-Related Quantitative Traits in a Population of Middle-Aged People: Studies of 14,940 Danes.” Diabetes, vol. 58, no. 3, Mar. 2009, pp. 757–64. PubMed, https://doi.org/10.2337/db08-0620.
Zorn, Stefanie, et al. “Obesity Due to MC4R Deficiency Is Associated with Reduced Cholesterol, Triglycerides and Cardiovascular Disease Risk.” Nature Medicine, vol. 31, no. 12, Dec. 2025, pp. 4180–88. www.nature.com, https://doi.org/10.1038/s41591-025-03976-1.
 After you have eaten, your body releases the satiety hormone, leptin.
After you have eaten, your body releases the satiety hormone, leptin.