Key takeaways:
~ HbA1c is a test value used to diagnose and manage diabetes.
~ Blood glucose levels affect HbA1c over the course of a few months.
~ HbA1c isn’t always an accurate measure of blood glucose levels, though.
~ Some people have genetic variants in red blood cell related genes that cause higher or lower HbA1c, impacting whether it is a true marker for glucose levels.
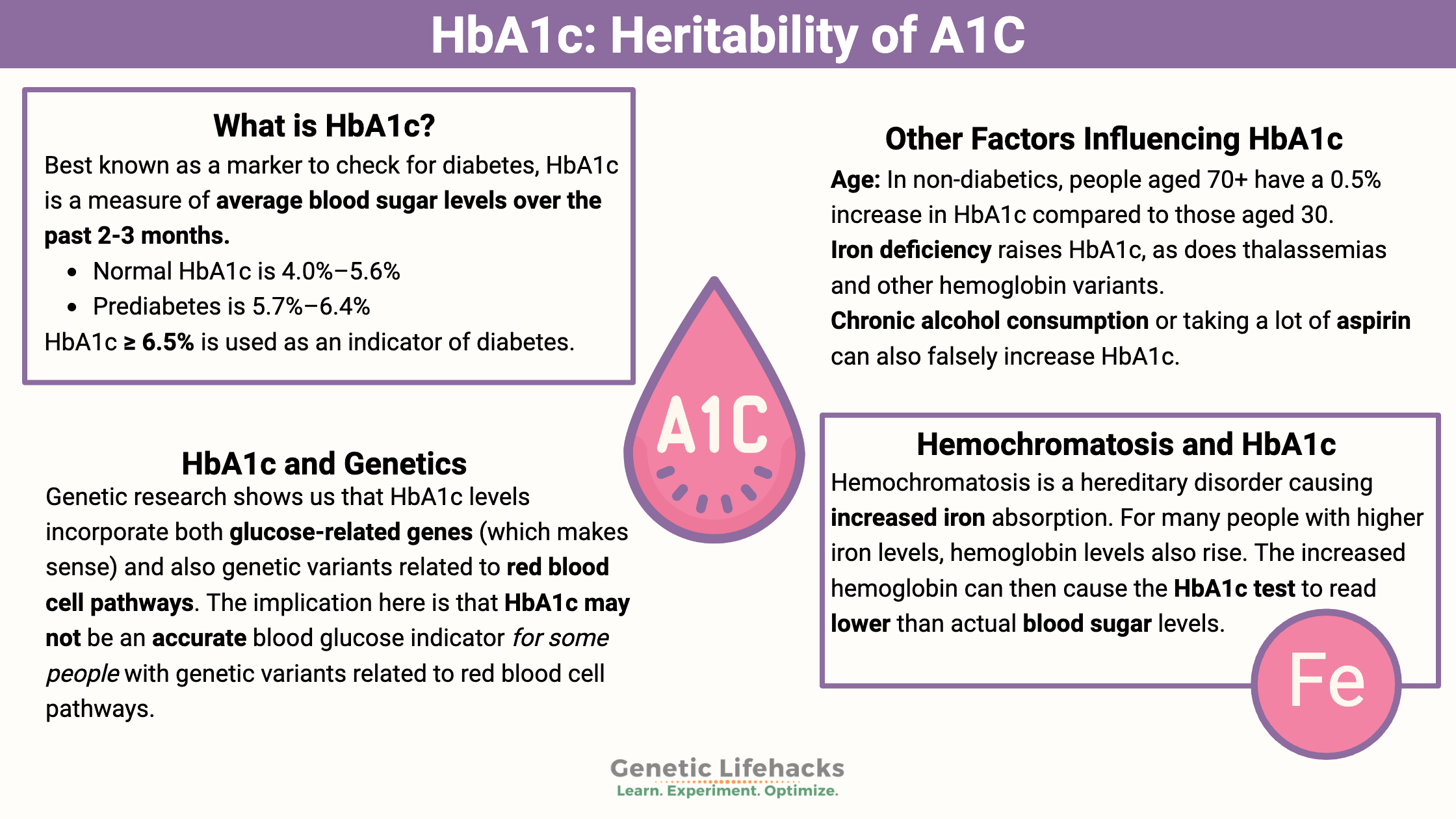
What is HbA1c?
Best known as a marker to check for diabetes, HbA1c is a measure of average blood sugar levels over the past 2-3 months.
HbA1c stands for hemoglobin A1c, also known as glycated hemoglobin.
High levels of HbA1c indicate that blood glucose has been elevated for a while. This is in contrast to just checking your blood sugar which gives you a snapshot of what is happening at the moment. For someone with diabetes, higher HbA1c levels can show that a medication is not working optimally. High levels are associated with an increased risk of diabetes-related complications such as heart disease, kidney failure, and retinopathy.[ref]
What is glycated hemoglobin?
Glycation is an irreversible change to a protein by the addition of a glucose molecule. When blood sugar levels are elevated for a period of time, the hemoglobin molecules are more likely to become glycated.[ref]
Hemoglobin is the part of the red blood cell carrying oxygen throughout the body.
Red blood cells turn over about every 110 days (~3 months), and the percentage of red blood cells that are glycated is proportional to the blood glucose level. For example, high blood glucose causes a high percentage of glycated hemoglobin.
Thus, HbA1c (percentage of glycated hemoglobin) gives a good estimate of whether blood sugar has been elevated over the past few months.[ref]
It’s a good estimate for most people… I’ll explain the genetic exceptions in a minute.
A1c test results:
For people who don’t have diabetes:
- Normal HbA1c is 4.0%–5.6%
- Prediabetes is 5.7%–6.4%
HbA1c ≥ 6.5% is used as an indicator of diabetes.[ref][ref] This number can vary slightly, depending on which reference you look at.
If you have diabetes and are on medication, the doctor will usually give you a target range for HbA1c.
What influences HbA1c levels?
Your average blood glucose levels impact your HbA1c levels, of course.
However, there are other influences on HbA1c, making the test not as accurate for determining average blood glucose:
- For example, if red blood cells turn over more quickly than average, the HbA1c level will be lower, and thus not a good representation of average blood sugar.
- If hemoglobin levels are low, such as in someone with anemia, this can also throw off the accuracy of HbA1c levels.
- Finally, certain genetic variants impact red blood cells, either increasing or decreasing HbA1c.
Let’s dive into all of these scenarios in more detail:
Heritability of HbA1c
There is a fairly strong genetic component to getting diabetes, so it makes sense that HbA1c would also have some kind of genetic connection.
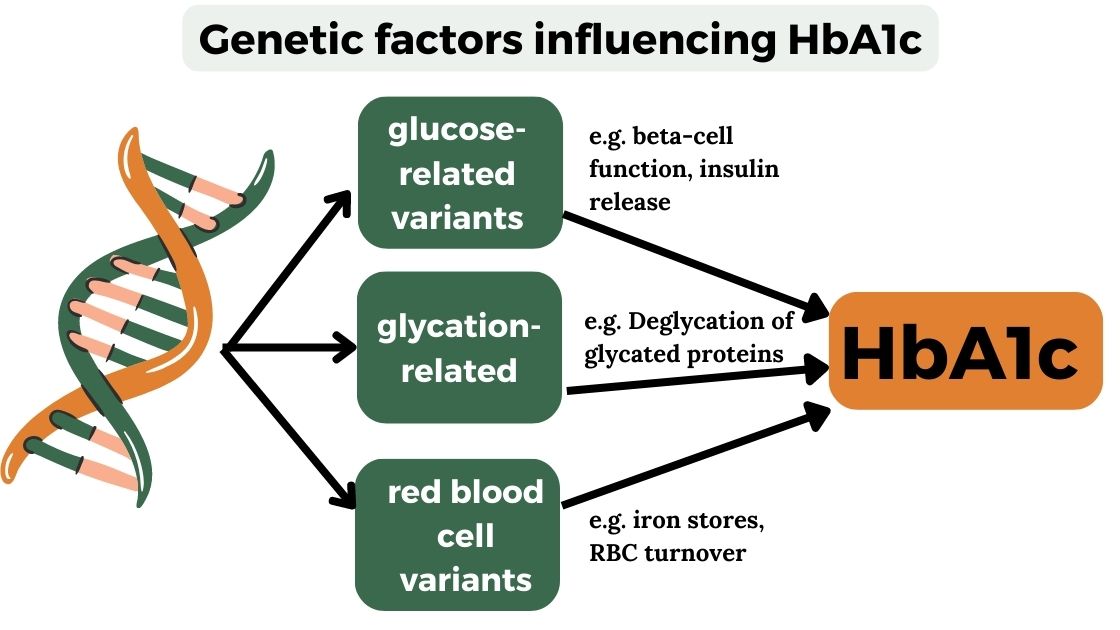
Genetic variants also influence HbA1c, with heritability estimated at over 50%.[ref]
But… it isn’t as simple as ‘the genes for diabetes cause HbA1c to be high because glucose is high’.
Genetic research shows us that HbA1c levels incorporate both glucose-related genes (which makes sense) and also genetic variants related to red blood cell pathways.[ref]
The implication here is that HbA1c may not be an accurate blood glucose indicator for some people with genetic variants related to red blood cell pathways.
I’m going to use the terms ‘artificial’ and ‘falsely low’ to indicate the factors that influence A1c outside of blood glucose levels. These aren’t actually artificial reasons, but rather the reasons for HbA1c tests not accurately representing average blood glucose levels.
Other factors influencing HbA1c readings:
Age: In non-diabetics, people aged 70+ have a 0.5% increase in HbA1c compared to those aged 30.[ref]
Iron deficiency raises HbA1c, as does thalassemias and other hemoglobin variants. Severe vitamin B12 deficiency causing anemia can also increase HbA1c. This is irrespective of blood glucose levels.[ref]
Hemolytic anemia causes blood cells to turn over more quickly than normal, and this can cause a lower HbA1c reading that doesn’t reflect blood glucose levels.
Smoking increases HbA1c levels by 0.1% on average, but it doesn’t change fasting blood glucose levels.[ref] This is something a doctor will likely consider when looking at your HbA1c levels.
Chronic alcohol consumption or taking a lot of aspirin (salicylates) can also falsely increase HbA1c.[ref]
Recent blood donations will also decrease HbA1c due to lowering hemoglobin levels.[ref]
What happens if HbA1c isn’t a good indicator of your blood glucose levels?
Knowledge is power here.
For people with significant genetic variants that cause HbA1c to read lower than it should for their glucose levels, this can cause doctors to miss out on diagnosing them with diabetes. Additionally, if HbA1c is artificially or falsely low in someone with diabetes, their medications may need to be adjusted.
HbA1c for African Americans
When reading through research studies on HbA1c, I saw several older studies that stated HbA1c levels could be incorrect (falsely low) for African Americans. There are even headlines in mainstream articles stating that HbA1c is less accurate in African Americans.[ref] This is statistically due to a couple of the red blood cell mutations (listed below) being more common in people of African ancestry.
It’s important to understand that most people with African ancestry do not have the mutations that cause HbA1c to be strongly impacted. Statistically, when you look at the population group as a whole, the HbA1c levels are more likely to be off. But this shouldn’t be applied to individuals.
Thus, you need to look at genetic variants (or other tests) to know whether anyone, regardless of race, is likely to have HbA1c levels that differ from the norm.
Hemochromatosis and HbA1c:
Hemochromatosis is a hereditary disorder causing increased iron absorption. For many people with higher iron levels, hemoglobin levels also rise. The increased hemoglobin can then cause the HbA1c test to read lower than actual blood sugar levels.[ref] Of course, there could be other variable at play in someone with hemochromatosis, depending on the amount of damage to the liver or pancreas from stored iron.
HbA1c Genotype Report:
Access this content:
An active subscription is required to access this content.
Lifehacks for reducing HbA1c:
Talk with your doctor if you have diabetes about how your specific genetic variants may need to be considered when looking at specific HbA1c targets for diabetes. Your doctor can order an HbA1c test, or you can do the test yourself with a home meter or by ordering lab tests yourself.
If your HbA1c is higher than you would like it to be, check to see how your genetic variants impact your risk for diabetes and blood glucose.
There are multiple pathways involved in diabetes and high blood glucose levels. Understanding the pathways affecting your blood glucose can help you to pinpoint which diet and supplements may be more effective. For example, people with MTNR1B variants may find that changing meal timing is essential, while people with SLC30A8 variants may want to ensure they get plenty of zinc in their diet.
Access this content:
An active subscription is required to access this content.
Related Article and Topics:
References:
Byrne, Hugh, et al. “Effects of Self-Directed Exercise Programmes on Individuals with Type 2 Diabetes Mellitus: A Systematic Review Evaluating Their Effect on HbA1c and Other Metabolic Outcomes, Physical Characteristics, Cardiorespiratory Fitness and Functional Outcomes.” Sports Medicine (Auckland, N.Z.), vol. 47, no. 4, Apr. 2017, pp. 717–33. PubMed, https://doi.org/10.1007/s40279-016-0593-y.
Chen, Peng, et al. “Multiple Nonglycemic Genomic Loci Are Newly Associated With Blood Level of Glycated Hemoglobin in East Asians.” Diabetes, vol. 63, no. 7, July 2014, pp. 2551–62. PubMed Central, https://doi.org/10.2337/db13-1815.
Coban, Erkan, et al. “Effect of Iron Deficiency Anemia on the Levels of Hemoglobin A1c in Nondiabetic Patients.” Acta Haematologica, vol. 112, no. 3, 2004, pp. 126–28. www.karger.com, https://doi.org/10.1159/000079722.
Grimsby, Jonna L., et al. “Race-Ethnic Differences in the Association of Genetic Loci with HbA1c Levels and Mortality in U.S. Adults: The Third National Health and Nutrition Examination Survey (NHANES III).” BMC Medical Genetics, vol. 13, Apr. 2012, p. 30. PubMed Central, https://doi.org/10.1186/1471-2350-13-30.
Haimoto, Hajime, et al. “Long-Term Effects of a Diet Loosely Restricting Carbohydrates on HbA1c Levels, BMI and Tapering of Sulfonylureas in Type 2 Diabetes: A 2-Year Follow-up Study.” Diabetes Research and Clinical Practice, vol. 79, no. 2, Feb. 2008, pp. 350–56. PubMed, https://doi.org/10.1016/j.diabres.2007.09.009.
Harris, Richard. “The A1C Blood Sugar Test May Be Less Accurate In African-Americans.” NPR, 7 Feb. 2017. NPR, https://www.npr.org/sections/health-shots/2017/02/07/513891129/the-a1c-blood-sugar-test-may-be-less-accurate-in-african-americans.
Juarez, Deborah Taira, et al. “Significance of HbA1c and Its Measurement in the Diagnosis of Diabetes Mellitus: US Experience.” Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy, vol. 7, Oct. 2014, pp. 487–94. PubMed Central, https://doi.org/10.2147/DMSO.S39092.
Leong, Aaron, and Eleanor Wheeler. “Genetics of HbA1c: A Case Study in Clinical Translation.” Current Opinion in Genetics & Development, vol. 50, June 2018, pp. 79–85. ScienceDirect, https://doi.org/10.1016/j.gde.2018.02.008.
Lira Neto, José Claudio Garcia, et al. “Efficacy of Cinnamon as an Adjuvant in Reducing the Glycemic Biomarkers of Type 2 Diabetes Mellitus: A Three-Month, Randomized, Triple-Blind, Placebo-Controlled Clinical Trial.” Journal of the American Nutrition Association, vol. 41, no. 3, 2022, pp. 266–74. PubMed, https://doi.org/10.1080/07315724.2021.1878967.
Lo, Ken Sin, et al. “Genetic Association Analysis Highlights New Loci That Modulate Hematological Trait Variation in Caucasians and African Americans.” Human Genetics, vol. 129, no. 3, Mar. 2011, pp. 307–17. PubMed, https://doi.org/10.1007/s00439-010-0925-1.
Moon, Jee-Young, et al. “A Genome-Wide Association Study Identifies Blood Disorder–Related Variants Influencing Hemoglobin A1c With Implications for Glycemic Status in U.S. Hispanics/Latinos.” Diabetes Care, vol. 42, no. 9, Sept. 2019, pp. 1784–91. PubMed Central, https://doi.org/10.2337/dc19-0168.
NEGRATO, Carlos Antonio, et al. “Periodontal Disease and Diabetes Mellitus.” Journal of Applied Oral Science, vol. 21, no. 1, 2013, pp. 1–12. PubMed Central, https://doi.org/10.1590/1678-7757201302106.
Parry‐Strong, Amber, et al. “Very Low Carbohydrate (Ketogenic) Diets in Type 2 Diabetes: A Systematic Review and Meta‐analysis of Randomized Controlled Trials.” Diabetes, Obesity & Metabolism, vol. 24, no. 12, Dec. 2022, pp. 2431–42. PubMed Central, https://doi.org/10.1111/dom.14837.
Radin, Michael S. “Pitfalls in Hemoglobin A1c Measurement: When Results May Be Misleading.” Journal of General Internal Medicine, vol. 29, no. 2, Feb. 2014, pp. 388–94. PubMed Central, https://doi.org/10.1007/s11606-013-2595-x.
Roth, J., et al. “HbA1c and Age in Non-Diabetic Subjects: An Ignored Association?” Experimental and Clinical Endocrinology & Diabetes: Official Journal, German Society of Endocrinology [and] German Diabetes Association, vol. 124, no. 10, 2016, pp. 637–42. PubMed, https://doi.org/10.1055/s-0042-105440.
Ryle, A. J., et al. “A Study of the Effect of Diet on Glycosylated Haemoglobin and Albumin Levels and Glucose Tolerance in Normal Subjects.” Diabetic Medicine: A Journal of the British Diabetic Association, vol. 7, no. 10, Dec. 1990, pp. 865–70. PubMed, https://doi.org/10.1111/j.1464-5491.1990.tb01320.x.
Sherwani, Shariq I., et al. “Significance of HbA1c Test in Diagnosis and Prognosis of Diabetic Patients.” Biomarker Insights, vol. 11, July 2016, pp. 95–104. PubMed Central, https://doi.org/10.4137/BMI.S38440.
Soranzo, Nicole, et al. “Common Variants at 10 Genomic Loci Influence Hemoglobin A1C Levels via Glycemic and Nonglycemic Pathways.” Diabetes, vol. 59, no. 12, Dec. 2010, pp. 3229–39. PubMed Central, https://doi.org/10.2337/db10-0502.
Sørensen, Erik, et al. “Genetic Factors Influencing Ferritin Levels in 14,126 Blood Donors: Results from the Danish Blood Donor Study.” Transfusion, vol. 56, no. 3, Mar. 2016, pp. 622–27. PubMed, https://doi.org/10.1111/trf.13397.
Soulimane, Soraya, et al. “HbA1c, Fasting and 2-Hour Plasma Glucose in Current-, Ex-, and Non-Smokers: A Meta-Analysis.” Diabetologia, vol. 57, no. 1, Jan. 2014, pp. 30–39. PubMed Central, https://doi.org/10.1007/s00125-013-3058-y.
Su, Lingyu, et al. “Toothbrushing, Blood Glucose and HbA1c: Findings from a Random Survey in Chinese Population.” Scientific Reports, vol. 6, July 2016, p. 28824. PubMed Central, https://doi.org/10.1038/srep28824.
Sugimoto, Takeshi, et al. “Alterations in HbA1c Resulting from the Donation of Autologous Blood for Elective Surgery in Patients with Diabetes Mellitus.” Blood Transfusion, vol. 12, no. Suppl 1, Jan. 2014, pp. s209–13. PubMed Central, https://doi.org/10.2450/2013.0271-12.
Tian, Jiao, et al. “The Effect of Curcumin on Lipid Profile and Glycemic Status of Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis.” Evidence-Based Complementary and Alternative Medicine: ECAM, vol. 2022, 2022, p. 8278744. PubMed, https://doi.org/10.1155/2022/8278744.
Tsukui, S., et al. “Moderate-Intensity Regular Exercise Decreases Serum Tumor Necrosis Factor-Alpha and HbA1c Levels in Healthy Women.” International Journal of Obesity and Related Metabolic Disorders: Journal of the International Association for the Study of Obesity, vol. 24, no. 9, Sept. 2000, pp. 1207–11. PubMed, https://doi.org/10.1038/sj.ijo.0801373.
Turbiville, Donald, et al. “Iron Overload in an HFE Heterozygous Carrier: A Case Report and Literature Review.” Laboratory Medicine, vol. 50, no. 2, Apr. 2019, pp. 212–17. PubMed, https://doi.org/10.1093/labmed/lmy065.
Xi, Huihui, et al. “Flaxseed Supplementation Significantly Reduces Hemoglobin A1c in Patients with Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis.” Nutrition Research (New York, N.Y.), vol. 110, Dec. 2022, pp. 23–32. PubMed, https://doi.org/10.1016/j.nutres.2022.12.008.
Xie, Wenting, et al. “Glucose-Lowering Effect of Berberine on Type 2 Diabetes: A Systematic Review and Meta-Analysis.” Frontiers in Pharmacology, vol. 13, 2022, p. 1015045. PubMed, https://doi.org/10.3389/fphar.2022.1015045.
Yan, Rengna, et al. “A Novel Type 2 Diabetes Risk Allele Increases the Promoter Activity of the Muscle-Specific Small Ankyrin 1 Gene.” Scientific Reports, vol. 6, Apr. 2016, p. 25105. PubMed, https://doi.org/10.1038/srep25105.
Zaki, Hany A., et al. “A Comparative Study Evaluating the Effectiveness Between Ketogenic and Low-Carbohydrate Diets on Glycemic and Weight Control in Patients With Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis.” Cureus, vol. 14, no. 5, May 2022, p. e25528. PubMed, https://doi.org/10.7759/cureus.25528.