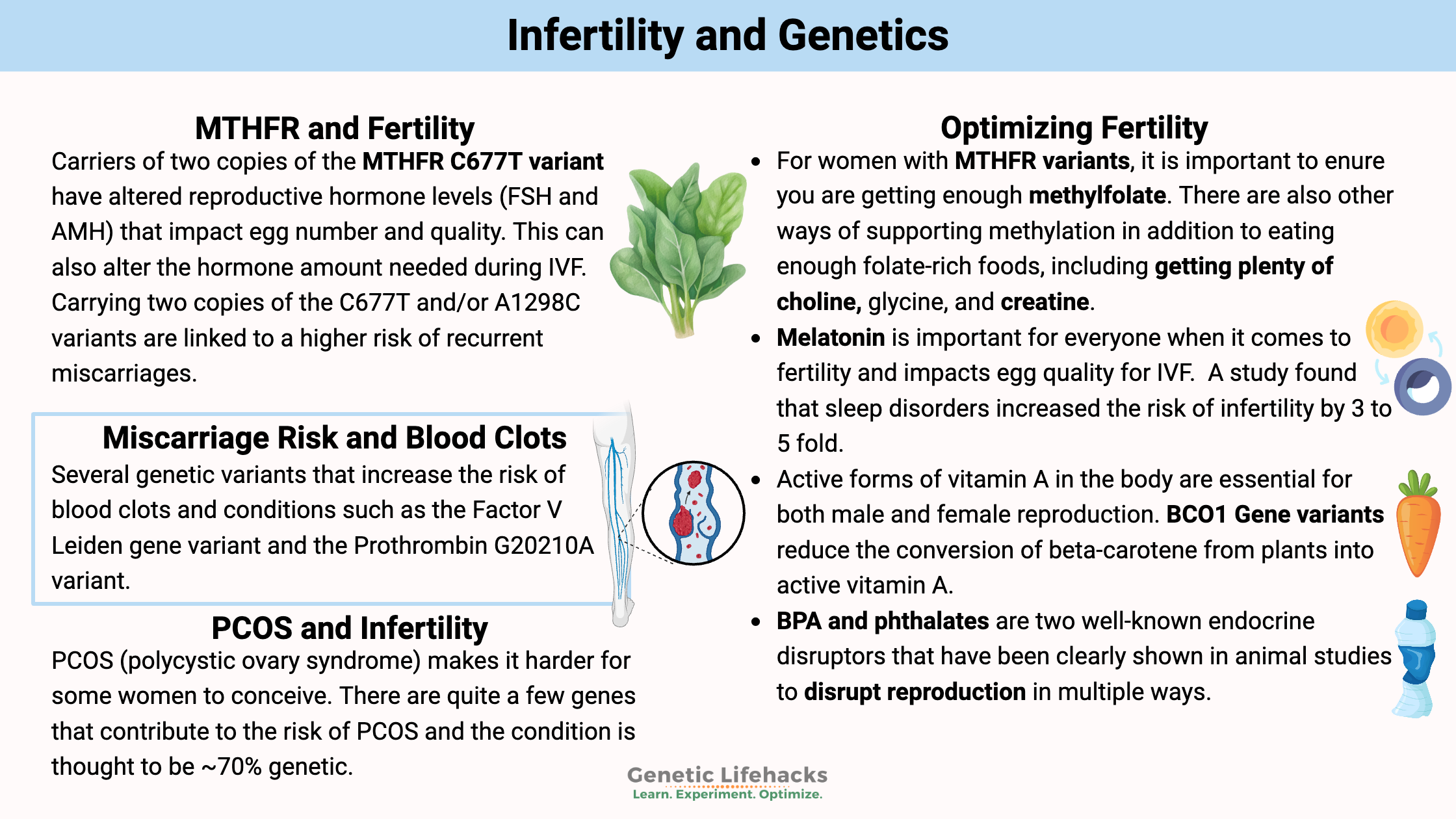
Key takeaways:
~ About 12% of women in the U.S. experience fertility challenges, with that rate increasing to 25% in women over 30.
~ Genetic variants can influence many factors associated with infertility, such as hormone levels, ovarian health, and the likelihood of structural or autoimmune issues.
~ Understanding your relevant genetic variants can provide insight into possible solutions for your root cause of infertility.
Infertility: Hereditary Causes
According to the CDC, estimates show 12% of US women have impaired fertility. For women over age 30, that number rises to 25%![ref][ref]
Some of the causes of infertility for women can be categorized as follows:
- Hormonal issues: thyroid problems, PCOS, HPA axis dysregulation, reproductive hormone dysregulation
- Structural issues: fibroids, blocked fallopian tubes, other structural abnormalities
- Ovarian insufficiency or premature menopause
- Recurrent miscarriages and more
This article highlights some of the common genetic variants that may play a role in infertility. It is just a starting point… My hope is that it gives you enough information to get started on figuring out the root cause of your problems when trying to conceive.
A quick crash course in reproduction:
A lot of people think of pregnancy in basic terms: sperm meets egg, and 9 months later you have a baby. Birds do it, bees do it. Heck, even trees do it in their own way.
But when you get down to the specific details, it gets a lot more complicated…
Ovarian follicles:
At puberty, women have around 300,000 to 400,000 follicles in their ovaries. These primordial follicles are tiny fluid-filled sacs that contain the oocyte (immature egg cell). Over the course of a woman’s reproductive years, only 400 – 500 eggs will reach maturity.
Hormones stimulate the development of some of the follicles each month. Usually only one will reach maturity each month, at which point ovulation occurs. (The other follicles are broken down and reabsorbed.)
That egg cell that gets released at ovulation is what can get fertilized by the sperm, resulting in a pregnancy if all goes well. The egg needs to be healthy with no DNA damage.
Reproductive hormone levels:
For all of this to happen, the reproductive hormones need to be at the right levels and at the right time. Follicle-stimulating hormone (FSH) rises at the right time to stimulate the immature follicles to begin to mature. Without FSH, the immature follicles will go through apoptosis (cell death).
When the follicle reaches a certain size, it will start secreting estrogen. This causes a surge in gonadotrophin-releasing hormone (GnRH).
The surge in GnRH causes a surge in luteinizing hormone (LH) secretion, which triggers ovulation.
Decreased fertility as we age:
As women age, they have fewer follicles in their ovaries, eventually reaching the end of the reproductive span at menopause. A hormone called AMH (anti-mullerian hormone) is often used as a marker of ‘ovarian reserve’, an estimate of available eggs.
But the ovaries aren’t like a gumball machine, spitting out good gumballs (eggs) up until the last one comes out. Oxidative damage, insults to the cells, etc. cause egg quality to decline as we age.
Conditions that can decrease fertility rates include PCOS (polycystic ovarian syndrome), hypothyroidism, diabetes, and autoimmune diseases.
The risk of recurrent miscarriages also increases with age. Blood clots can increase the risk of recurrent miscarriages, and several genetic variants cause increased clotting.
We will dive into how your genes specifically interact with possible causes of infertility in the Genotype Report section…
Fertility Genotype Report:
Access this content:
An active subscription is required to access this content.
Lifehacks: Natural solutions when TTC
I’ve thrown a lot of information into this article, but it is just a portion of what goes into the mix for fertility.
If you are dealing with infertility, genetic variants are probably playing a role — whether through PCOS, altering your vitamin or hormone levels, or increasing risk of problems from toxins. But this is just a part of the picture… stress, sleep, and diet are all important also.
Getting help:
Obviously, the first place to go for help when trying to conceive is your OB/GYN for testing. Your OB can do ultrasounds and other tests to rule out a physical cause. Hormone testing is also available to make sure everything is normal there.
If you are looking for a professional who is familiar with Genetic Lifehacks reports, check out the list of PRO members.
Optimizing fertility:
Your overall health impacts your ability to reproduce. This makes sense for all animals and especially for humans.
There are quite a few genetic variants affecting the very basic aspects of health that also affect fertility. You need healthy egg cells in order to get pregnant. This list is a bit of a hodge-podge of some of those variants that affect the quality of the oocyte.[ref]
Melatonin genes that impact fertility:
Whether you carry the melatonin receptor variant (above) or not, melatonin is important for everyone when it comes to fertility. This is especially true for older women who are TTC – melatonin levels drop as we get older.
The ‘sleep hormone’ does a lot in the body. Within cells, it acts as a natural antioxidant, reducing reactive oxygen species. Melatonin receptors are found in high levels in the ovaries and in the follicle cells. Melatonin turns out to be fairly important in egg quality for IVF.[ref][ref][ref][ref] Melatonin also may play a role in keeping the mother’s body from rejecting the fetus.[ref]
How can you increase melatonin? Two free things you can do that will greatly impact melatonin levels: Block 100% of blue light at night and get out in the sunshine during the day.
Related article: Key to health: melatonin
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
BPA: How Your Genes Influence BPA Detoxification
BPA, a chemical found in some plastics, has been linked to a variety of effects on people including obesity, insulin resistance, and epigenetic effects on the fetus. It is everywhere in our food supply. In fact, a CDC report showed that 92% of people have BPA in their urine.
Detoxifying Phthalates: Genes and Diet
Plastics are everywhere – and a source of the chemicals that we are exposed to on a daily basis. One component of plastics is a class of compounds referred to as phthalates, which can act as an endocrine disruptor and mimic estrogen.
PCOS: Genetics and Root Causes
Polycystic ovarian syndrome (PCOS) is an endocrine disorder causing an increase in androgen hormone production in women. It also increases the risk of infertility. PCOS affects 5 -10% of premenopausal women, and genetics plays a large role in whether you have PCOS.
BCO1 Gene: Converting Beta-Carotene to Vitamin A
Genetics plays a huge role in how well you convert beta-carotene into vitamin A! Discover how well you convert beta-carotene into retinol.
References:
Al-Achkar, Walid, et al. “Association of Methylenetetrahydrofolate Reductase C677T and A1298C Gene Polymorphisms With Recurrent Pregnancy Loss in Syrian Women.” Reproductive Sciences (Thousand Oaks, Calif.), vol. 24, no. 9, Sept. 2017, pp. 1275–79. PubMed, https://doi.org/10.1177/1933719116682874.
Capalbo, A., et al. “The 312N Variant of the Luteinizing Hormone/Choriogonadotropin Receptor Gene (LHCGR) Confers up to 2·7-Fold Increased Risk of Polycystic Ovary Syndrome in a Sardinian Population.” Clinical Endocrinology, vol. 77, no. 1, July 2012, pp. 113–19. PubMed, https://doi.org/10.1111/j.1365-2265.2012.04372.x.
Carlomagno, Gianfranco, et al. “From Implantation to Birth: Insight into Molecular Melatonin Functions.” International Journal of Molecular Sciences, vol. 19, no. 9, Sept. 2018, p. 2802. PubMed Central, https://doi.org/10.3390/ijms19092802.
Choi, Youngsok, et al. “Genetic Variation of Methylenetetrahydrofolate Reductase (MTHFR) and Thymidylate Synthase (TS) Genes Is Associated with Idiopathic Recurrent Implantation Failure.” PloS One, vol. 11, no. 8, 2016, p. e0160884. PubMed, https://doi.org/10.1371/journal.pone.0160884.
England, Fertility Centers of New. “Can Infertility Be Treated? | About Infertility.” Fertility Centers of New England, https://www.fertilitycenter.com/our-services/about-infertility/. Accessed 17 May 2022.
FastStats. 20 Dec. 2021, https://www.cdc.gov/nchs/fastats/infertility.htm.
Gao, Hui, and Fang-biao Tao. “Prothrombin G20210A Mutation Is Associated with Recurrent Pregnancy Loss: A Systematic Review and Meta-Analysis Update.” Thrombosis Research, vol. 135, no. 2, Feb. 2015, pp. 339–46. PubMed, https://doi.org/10.1016/j.thromres.2014.12.001.
Goodarzi, Mark O., et al. “Replication of Association of DENND1A and THADA Variants with Polycystic Ovary Syndrome in European Cohorts.” Journal of Medical Genetics, vol. 49, no. 2, Feb. 2012, pp. 90–95. PubMed Central, https://doi.org/10.1136/jmedgenet-2011-100427.
Gu, Bon-Hee, et al. “Genetic Variations of Follicle Stimulating Hormone Receptor Are Associated with Polycystic Ovary Syndrome.” International Journal of Molecular Medicine, vol. 26, no. 1, July 2010, pp. 107–12. www.spandidos-publications.com, https://doi.org/10.3892/ijmm_00000441.
Hubacek, Jaroslav A., et al. “Association of MTHFR Genetic Variants C677T and A1298C on Predisposition to Spontaneous Abortion in Slavonic Population.” Clinica Chimica Acta; International Journal of Clinical Chemistry, vol. 440, Feb. 2015, pp. 104–07. PubMed, https://doi.org/10.1016/j.cca.2014.11.018.
Joham, Anju E., et al. “Prevalence of Infertility and Use of Fertility Treatment in Women with Polycystic Ovary Syndrome: Data from a Large Community-Based Cohort Study.” Journal of Women’s Health (2002), vol. 24, no. 4, Apr. 2015, pp. 299–307. PubMed, https://doi.org/10.1089/jwh.2014.5000.
Jusić, Amela, et al. “The Association of Factor V G1961A (Factor V Leiden), Prothrombin G20210A, MTHFR C677T and PAI-1 4G/5G Polymorphisms with Recurrent Pregnancy Loss in Bosnian Women.” Medicinski Glasnik: Official Publication of the Medical Association of Zenica-Doboj Canton, Bosnia and Herzegovina, vol. 15, no. 2, Aug. 2018, pp. 158–63. PubMed, https://doi.org/10.17392/948-18.
Keefe, David, et al. “Oocyte Competency Is the Key to Embryo Potential.” Fertility and Sterility, vol. 103, no. 2, Feb. 2015, pp. 317–22. PubMed, https://doi.org/10.1016/j.fertnstert.2014.12.115.
Lerchbaum, E., et al. “Susceptibility Loci for Polycystic Ovary Syndrome on Chromosome 2p16.3, 2p21, and 9q33.3 in a Cohort of Caucasian Women.” Hormone and Metabolic Research = Hormon- Und Stoffwechselforschung = Hormones Et Metabolisme, vol. 43, no. 11, Oct. 2011, pp. 743–47. PubMed, https://doi.org/10.1055/s-0031-1286279.
Li, Chao, et al. “Association of Rs10830963 and Rs10830962 SNPs in the Melatonin Receptor (MTNR1B) Gene among Han Chinese Women with Polycystic Ovary Syndrome.” Molecular Human Reproduction, vol. 17, no. 3, Mar. 2011, pp. 193–98. PubMed, https://doi.org/10.1093/molehr/gaq087.
Lissalde-Lavigne, G., et al. “Factor V Leiden and Prothrombin G20210A Polymorphisms as Risk Factors for Miscarriage during a First Intended Pregnancy: The Matched Case-Control ‘NOHA First’ Study.” Journal of Thrombosis and Haemostasis: JTH, vol. 3, no. 10, Oct. 2005, pp. 2178–84. PubMed, https://doi.org/10.1111/j.1538-7836.2005.01581.x.
Nishihara, Takuji, et al. “Oral Melatonin Supplementation Improves Oocyte and Embryo Quality in Women Undergoing in Vitro Fertilization-Embryo Transfer.” Gynecological Endocrinology: The Official Journal of the International Society of Gynecological Endocrinology, vol. 30, no. 5, May 2014, pp. 359–62. PubMed, https://doi.org/10.3109/09513590.2013.879856.
Pacchiarotti, Alessandro, et al. “Effect of Myo-Inositol and Melatonin versus Myo-Inositol, in a Randomized Controlled Trial, for Improving in Vitro Fertilization of Patients with Polycystic Ovarian Syndrome.” Gynecological Endocrinology: The Official Journal of the International Society of Gynecological Endocrinology, vol. 32, no. 1, 2016, pp. 69–73. PubMed, https://doi.org/10.3109/09513590.2015.1101444.
Pau, Cindy T., et al. “Phenotype and Tissue Expression as a Function of Genetic Risk in Polycystic Ovary Syndrome.” PLoS ONE, vol. 12, no. 1, Jan. 2017, p. e0168870. PubMed Central, https://doi.org/10.1371/journal.pone.0168870.
Pietropolli, A., et al. “Plasminogen Activator Inhibitor-1, Factor V, Factor II and Methylenetetrahydrofolate Reductase Polymorphisms in Women with Recurrent Miscarriage.” Journal of Obstetrics and Gynaecology: The Journal of the Institute of Obstetrics and Gynaecology, vol. 34, no. 3, Apr. 2014, pp. 229–34. PubMed, https://doi.org/10.3109/01443615.2013.836476.
Reznikoff-Etiévan, M. F., et al. “Factor V Leiden and G20210A Prothrombin Mutations Are Risk Factors for Very Early Recurrent Miscarriage.” BJOG: An International Journal of Obstetrics and Gynaecology, vol. 108, no. 12, Dec. 2001, pp. 1251–54. PubMed, https://doi.org/10.1111/j.1471-0528.2001.00298.x.
Ridker, P. M., et al. “Factor V Leiden Mutation as a Risk Factor for Recurrent Pregnancy Loss.” Annals of Internal Medicine, vol. 128, no. 12 Pt 1, June 1998, pp. 1000–03. PubMed, https://doi.org/10.7326/0003-4819-128-12_part_1-199806150-00007.
Shahrokhi, Seyedeh Z., et al. “The Relationship Between the MTHFR C677T Genotypes to Serum Anti-Müllerian Hormone Concentrations and In Vitro Fertilization/Intracytoplasmic Sperm Injection Outcome.” Clinical Laboratory, vol. 63, no. 5, May 2017, pp. 927–34. PubMed, https://doi.org/10.7754/Clin.Lab.2016.161104.
Shi, X., et al. “Maternal Genetic Polymorphisms and Unexplained Recurrent Miscarriage: A Systematic Review and Meta-Analysis.” Clinical Genetics, vol. 91, no. 2, Feb. 2017, pp. 265–84. PubMed, https://doi.org/10.1111/cge.12910.
Spinedi, Eduardo, and Daniel P. Cardinali. “The Polycystic Ovary Syndrome and the Metabolic Syndrome: A Possible Chronobiotic-Cytoprotective Adjuvant Therapy.” International Journal of Endocrinology, vol. 2018, July 2018, p. e1349868. www.hindawi.com, https://doi.org/10.1155/2018/1349868.
Tagliaferri, Valeria, et al. “Melatonin Treatment May Be Able to Restore Menstrual Cyclicity in Women With PCOS: A Pilot Study.” Reproductive Sciences (Thousand Oaks, Calif.), vol. 25, no. 2, Feb. 2018, pp. 269–75. PubMed, https://doi.org/10.1177/1933719117711262.
Tannus, Samer, et al. “Treatment Strategies for the Infertile Polycystic Ovary Syndrome Patient.” Women’s Health (London, England), vol. 11, no. 6, Nov. 2015, pp. 901–12. PubMed, https://doi.org/10.2217/whe.15.40.
Thaler, C. J. “Folate Metabolism and Human Reproduction.” Geburtshilfe Und Frauenheilkunde, vol. 74, no. 9, Sept. 2014, pp. 845–51. PubMed Central, https://doi.org/10.1055/s-0034-1383058.
Zeng, Shuangshuang, et al. “MTHFR C677T Polymorphism Is Associated with Follicle-Stimulating Hormone Levels and Controlled Ovarian Hyperstimulation Response: A Retrospective Study from the Clinical Database.” Fertility and Sterility, vol. 111, no. 5, May 2019, pp. 982-990.e2. ScienceDirect, https://doi.org/10.1016/j.fertnstert.2019.01.016.
Zou, Ju, et al. “Association of Luteinizing Hormone/Choriogonadotropin Receptor Gene Polymorphisms with Polycystic Ovary Syndrome Risk: A Meta-Analysis.” Gynecological Endocrinology: The Official Journal of the International Society of Gynecological Endocrinology, vol. 35, no. 1, Jan. 2019, pp. 81–85. PubMed, https://doi.org/10.1080/09513590.2018.1498834.