Key takeaways:
~ Slow-wave sleep is important for sleep quality and cognitive function.
~ Glycine is an amino acid that you can get from food or can create in the body.
~ Glycine acts as an inhibitory neurotransmitter via glycine receptors in certain types of neurons, and this is tied to the amount of slow-wave sleep.
This article digs into the research on how glycine may help to improve sleep, and how genetic variants impact slow-wave sleep.
Better Sleep for a Better Life: Understanding Slow-Wave Sleep & Glycine
Sleep is one of those essential things that everyone should prioritize to maintain good health. Yes, rare mutations reduce your need for sleep, but if you’re not one of the 1 in 10,000 people with the DEC2 mutation, you need to get around 7.5 to 8 hours of sleep per night.
A recent study on sleep and dementia points out (once again) that sleep is essential for good health in aging. The study discovered that getting less sleep in your 50s and 60s, such as 6 hours or fewer per night, raises your risk of dementia in your later years by 30%.[ref][ref]
How can you improve sleep quality?
Of course, the first step is to plan enough time for sleep. Parents of small children understand the importance of getting enough sleep. However, as adults, we tend to overlook the simple math of this. I was surprised when my Oura ring prompted me to start getting ready for bed at 9:15 p.m. However, because I usually get up at about 5:45 a.m., Oura was simply deducting 8 hours and adding a half-hour buffer to brush my teeth and wind down to sleep.
Sleep duration is a straightforward metric… But everyone knows that poor quality sleep negatively impacts how you feel the next day. Slow-wave sleep is a key here.
Your brain activity goes through multiple stages when you sleep. You will have segments of light sleep, REM sleep, and slow-wave sleep.
Why is slow-wave sleep important?
While all the stages of sleep are needed, slow-wave sleep stands out for several reasons:
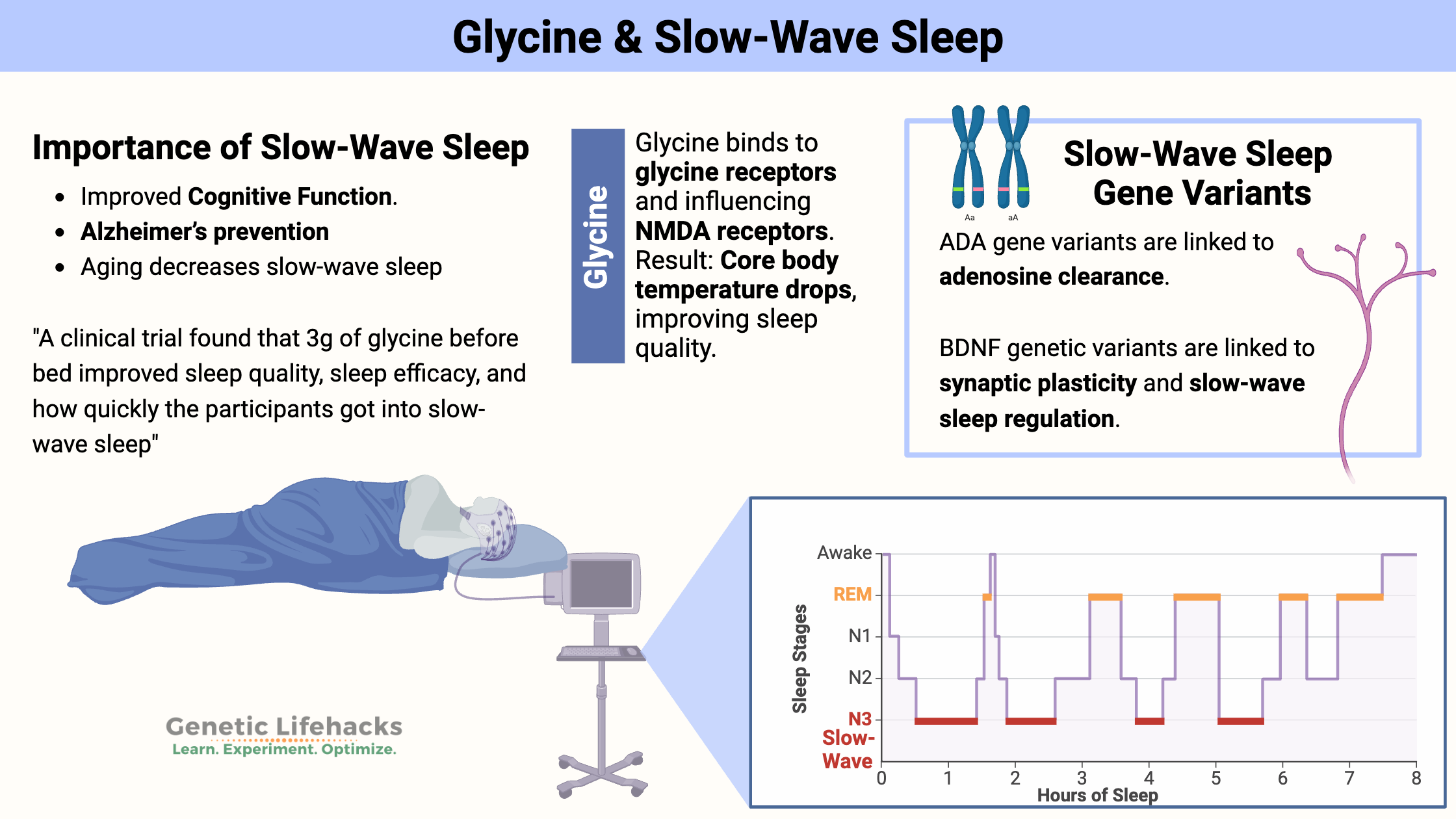
- Better cognitive function
- Preventing Alzheimer’s disease
- Avoiding cognitive decline in aging
Slow-wave sleep is linked to mental acuity. Cognitive tests show that people getting more slow-wave sleep have better response times and accuracy.[ref]
One of the problems with aging is that slow-wave sleep decreases and becomes more fragmented. Researchers think this loss of slow-wave sleep is thought to cause cognitive decline and lead to higher amyloid-beta levels (Alzheimer’s implications).[ref][ref]
The contrary is also true, with research indicating that getting more slow-wave sleep can help avoid cognitive decline. Animal research points to a possible benefit for Alzheimer’s prevention with solid slow-wave sleep.[ref]
How can you increase your slow-wave sleep?
Taking glycine before bed has been shown in studies to improve sleep quality and increase the time spent in slow-wave sleep.
Glycine is an amino acid used throughout the body in many different ways. Your body naturally produces glycine (avg. of 45g per day), and you also get some from your diet (avg. 3-5g per day).
In the central nervous system, glycine acts as an inhibitory neurotransmitter via glycine receptors in certain types of neurons. Additionally, glycine and glutamate act together in the brain in certain types of excitatory neurons.[ref]
So what is going on in the brain when you take glycine at night?
When taking glycine as a supplement, it can easily pass across the blood-brain barrier to be used in the brain.
Glycine is a co-agonist with glutamate as a neurotransmitter in some regions of the brain. NMDA receptors in neurons are activated by glutamate and glycine when certain conditions are met. (The NMDA receptors also activate with certain psychoactive drugs, ketamine, and alcohol.)[ref]
One area of the brain with NMDA receptors is the suprachiasmatic nucleus, the region of the hypothalamus responsible for your circadian rhythm. Animal research shows that activation of these NMDA receptors by glycine decreases body temperature at night and is sleep-promoting.[ref]
Some people naturally get more slow-wave sleep:
Genetics plays a role in how much slow-wave sleep you naturally get. There are a couple of different genes that are important here.
Adenosine levels drop as you sleep:
During the day, adenosine builds up in your brain as your brain uses ATP (adenosine triphosphate) for energy. The ATP molecule gives up a phosphate to generate energy from the chemical bond. ADT (adenosine diphosphate) is formed, which can then give up another phosphate to form AMP (adenosine monophosphate).
One way the body can control adenosine levels is to convert them to inosine using the enzyme adenosine deaminase (ADA). This enzyme is used in the brain to decrease adenosine levels as you sleep.[ref] As adenosine levels drop throughout the night, the pressure to sleep also decreases.[ref]
While researchers have known for a while that adenosine is important in feeling sleepy, recent animal studies have clearly shown that activation of the adenosine receptor, ADORA2A, by the adenosine molecule, induces slow-wave sleep.[ref]
BDNF and Sleep: Interaction with Glycine
BDNF (brain-derived neurotrophic factor) acts on synaptic connections between neurons in the brain, potentiating or increasing them. One of BDNF’s functions is to aid neuroplasticity and memory formation.
Related article: The Interaction Between BDNF and Serotonin
BDNF appears to play a role in slow-wave sleep as well. Low BDNF levels are linked to sleep disturbances and an increased risk of depression.[ref]
Research ties glycine and BDNF together in relation to slow-wave sleep. Animal studies show that BDNF alters glycine uptake by impacting the way glycine transporters work in the neurons.[ref]
Slow Wave Sleep Genotype Report:
Access this content:
An active subscription is required to access this content.
Lifehacks: How to use glycine to increase slow-wave sleep
Research on glycine for sleep quality:
Let’s take a look at studies showing four ways that glycine impacts sleep quality:
1) Improves sleep quality:
A clinical trial found that 3g of glycine before bed improved sleep quality, sleep efficacy, and how quickly the participants got into slow-wave sleep.[ref]
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
Genes that Impact Magnesium Levels
Are your magnesium levels low? Understanding your genes can help you decide whether you may need more magnesium in your diet or via supplements.
Best Supplements to Boost BDNF
Is it smart to boost your BDNF levels? Discover more by digging into the research studies that show when, how, and why it is important to focus on BDNF.
Restless Leg and Periodic Limb Movement Disorder: Genes and Solutions
Many know the frustration of disrupted sleep due to RLS and periodic limb movement disorder (PLMD). Genes play a role in your risk for these disorders, and there are specific research-based treatments to explore.
Insomnia, Sleepless Nights, and Your Genes
Learn about some of the genetic reasons for insomnia — and the solutions that may work for you based on your genetic variants.
References:
Amin, Khalid, et al. “Dietary Glycine Inhibits Angiogenesis during Wound Healing and Tumor Growth.” Cancer Biology & Therapy, vol. 2, no. 2, Apr. 2003, pp. 173–78. PubMed, https://doi.org/10.4161/cbt.2.2.280.
Aroeira, Rita I., et al. “BDNF Modulates Glycine Uptake in Hippocampal Synaptosomes by Decreasing Membrane Insertion of Glycine Transporter 2.” Neurochemistry International, vol. 99, Oct. 2016, pp. 94–102. ScienceDirect, https://doi.org/10.1016/j.neuint.2016.06.007.
Bachmann, Valérie, Federica Klaus, et al. “Functional ADA Polymorphism Increases Sleep Depth and Reduces Vigilant Attention in Humans.” Cerebral Cortex (New York, N.Y.: 1991), vol. 22, no. 4, Apr. 2012, pp. 962–70. PubMed, https://doi.org/10.1093/cercor/bhr173.
Bachmann, Valérie, Carina Klein, et al. “The BDNF Val66Met Polymorphism Modulates Sleep Intensity: EEG Frequency- and State-Specificity.” Sleep, vol. 35, no. 3, Mar. 2012, pp. 335–44. PubMed Central, https://doi.org/10.5665/sleep.1690.
Bannai, Makoto, et al. “The Effects of Glycine on Subjective Daytime Performance in Partially Sleep-Restricted Healthy Volunteers.” Frontiers in Neurology, vol. 3, 2012. Frontiers, https://www.frontiersin.org/article/10.3389/fneur.2012.00061.
Bannai, Makoto, and Nobuhiro Kawai. “New Therapeutic Strategy for Amino Acid Medicine:
Glycine Improves the Quality of Sleep.” Journal of Pharmacological Sciences, vol. 118, no. 2, 2012, pp. 145–48. J-Stage, https://doi.org/10.1254/jphs.11R04FM.
Baril, Andrée-Ann, et al. “Slow-Wave Sleep and MRI Markers of Brain Aging in a Community-Based Sample.” Neurology, vol. 96, no. 10, Mar. 2021, pp. e1462–69. PubMed, https://doi.org/10.1212/WNL.0000000000011377.
Cummings, Kirstie A., and Gabriela K. Popescu. “Glycine-Dependent Activation of NMDA Receptors.” The Journal of General Physiology, vol. 145, no. 6, June 2015, pp. 513–27. PubMed Central, https://doi.org/10.1085/jgp.201411302.
della Monica, Ciro, et al. “Rapid Eye Movement Sleep, Sleep Continuity and Slow Wave Sleep as Predictors of Cognition, Mood, and Subjective Sleep Quality in Healthy Men and Women, Aged 20–84 Years.” Frontiers in Psychiatry, vol. 9, June 2018, p. 255. PubMed Central, https://doi.org/10.3389/fpsyt.2018.00255.
Deuschle, Michael, et al. “Serum Brain-Derived Neurotrophic Factor (BDNF) in Sleep-Disordered Patients: Relation to Sleep Stage N3 and Rapid Eye Movement (REM) Sleep across Diagnostic Entities.” Journal of Sleep Research, vol. 27, no. 1, Feb. 2018, pp. 73–77. DOI.org (Crossref), https://doi.org/10.1111/jsr.12577.
Greene, Robert W., et al. “The Adenosine-Mediated, Neuronal-Glial, Homeostatic Sleep Response.” Current Opinion in Neurobiology, vol. 44, June 2017, pp. 236–42. PubMed Central, https://doi.org/10.1016/j.conb.2017.05.015.
Kawai, Nobuhiro, et al. “The Sleep-Promoting and Hypothermic Effects of Glycine Are Mediated by NMDA Receptors in the Suprachiasmatic Nucleus.” Neuropsychopharmacology, vol. 40, no. 6, May 2015, pp. 1405–16. PubMed Central, https://doi.org/10.1038/npp.2014.326.
Lee, Yee Fun, et al. “Slow Wave Sleep Is a Promising Intervention Target for Alzheimer’s Disease.” Frontiers in Neuroscience, vol. 14, June 2020, p. 705. PubMed Central, https://doi.org/10.3389/fnins.2020.00705.
Mazzotti, Diego Robles, et al. “Adenosine Deaminase Polymorphism Affects Sleep EEG Spectral Power in a Large Epidemiological Sample.” PLOS ONE, vol. 7, no. 8, Aug. 2012, p. e44154. PLoS Journals, https://doi.org/10.1371/journal.pone.0044154.
Oishi, Yo, et al. “Slow-Wave Sleep Is Controlled by a Subset of Nucleus Accumbens Core Neurons in Mice.” Nature Communications, vol. 8, Sept. 2017, p. 734. PubMed Central, https://doi.org/10.1038/s41467-017-00781-4.
Ozan, Erol, et al. “The Effect of Depression, BDNF Gene Val66met Polymorphism and Gender on Serum BDNF Levels.” Brain Research Bulletin, vol. 81, no. 1, Jan. 2010, pp. 61–65. ScienceDirect, https://doi.org/10.1016/j.brainresbull.2009.06.022.
Rétey, J. V., et al. “A Functional Genetic Variation of Adenosine Deaminase Affects the Duration and Intensity of Deep Sleep in Humans.” Proceedings of the National Academy of Sciences of the United States of America, vol. 102, no. 43, Oct. 2005, pp. 15676–81. PubMed Central, https://doi.org/10.1073/pnas.0505414102.
Sugaya, Kimio, et al. “Dietary Glycine Improves Urine Storage Symptoms in Urology Outpatients.” Journal of Complementary & Integrative Medicine, vol. 18, no. 3, Apr. 2021, pp. 617–20. PubMed, https://doi.org/10.1515/jcim-2020-0282.
Varga, Andrew W., et al. “Reduced Slow-Wave Sleep Is Associated with High Cerebrospinal Fluid Aβ42 Levels in Cognitively Normal Elderly.” Sleep, vol. 39, no. 11, Nov. 2016, pp. 2041–48. PubMed Central, https://doi.org/10.5665/sleep.6240.
Wunderlin, Marina, et al. “The Role of Slow Wave Sleep in the Development of Dementia and Its Potential for Preventative Interventions.” Psychiatry Research: Neuroimaging, vol. 306, Dec. 2020, p. 111178. ScienceDirect, https://doi.org/10.1016/j.pscychresns.2020.111178.
Yamadera, Wataru, et al. “Glycine Ingestion Improves Subjective Sleep Quality in Human Volunteers, Correlating with Polysomnographic Changes.” Sleep and Biological Rhythms, vol. 5, no. 2, Apr. 2007, pp. 126–31. Springer Link, https://doi.org/10.1111/j.1479-8425.2007.00262.x.