Key takeaways:
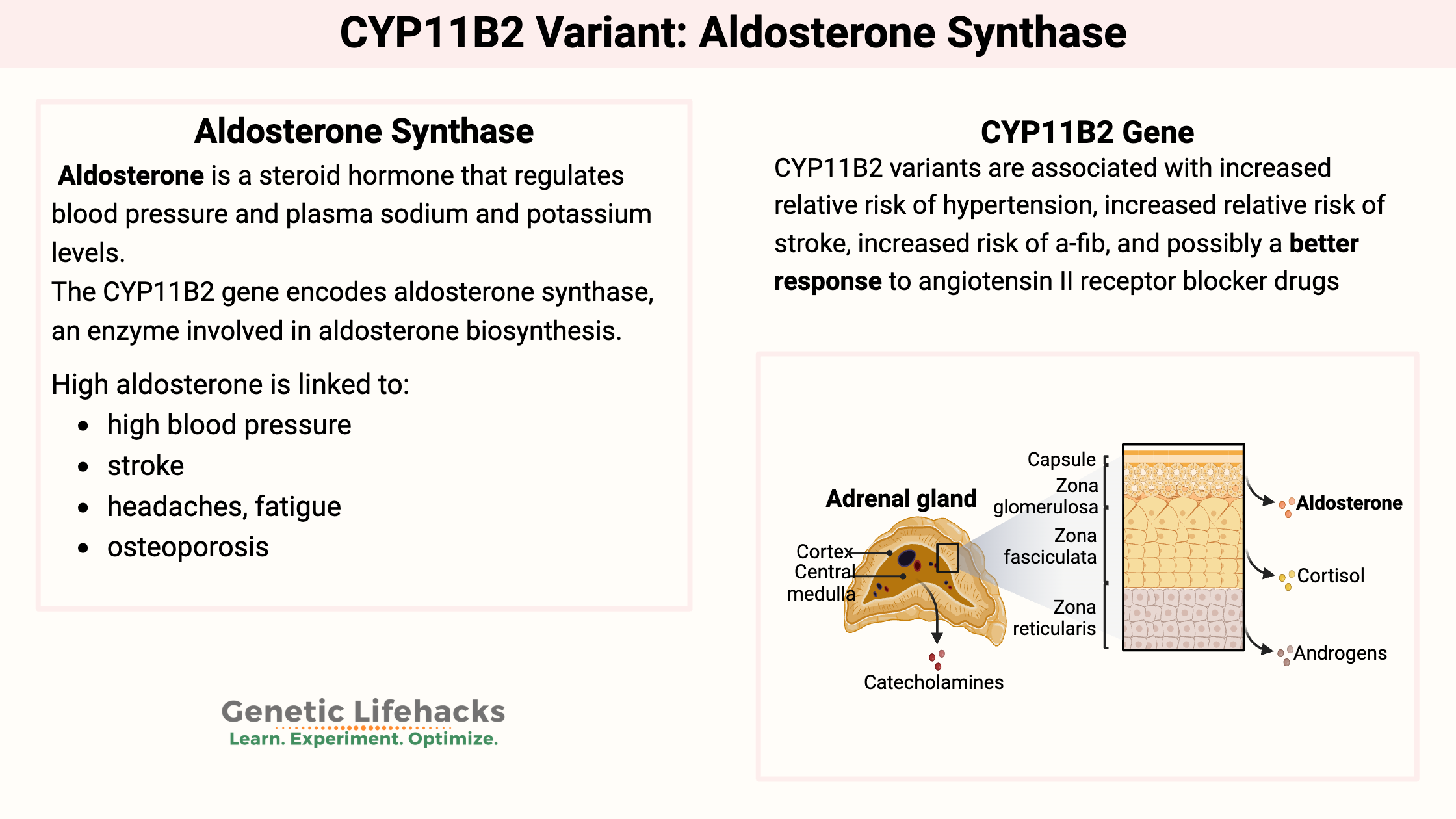
~ Aldosterone synthase, encoded by the CYP11B2 gene, is essential for regulating blood pressure by controlling aldosterone production, which balances sodium and potassium levels in the kidneys.
~ Genetic variants in the CYP11B2 gene increase the risk of hypertension, stroke, atrial fibrillation, and other cardiovascular conditions.
What is aldosterone synthase?
The CYP11B2 gene encodes aldosterone synthase, an enzyme involved in aldosterone biosynthesis.
First, what is aldosterone? Aldosterone is a steroid hormone that regulates blood pressure and plasma sodium and potassium levels. It works on the kidneys to balance out electrolytes (Na+ and K+) as a way of influencing blood pressure and blood volume.
In the body, think of ‘water follows sodium’. We all know a meal high in salt can leave us feeling ‘puffy’, and the action here is of the same concept. Aldosterone influences the kidneys to retain sodium while also excreting potassium. This shift in the electrolyte balance that allows for sodium retention directly increases water retention…by water following sodium. This action allows for an increase in blood volume and pressure.[ref]
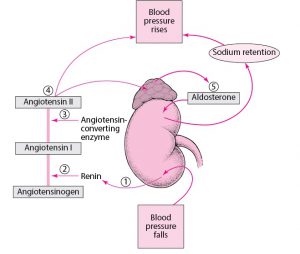
Aldosterone synthase is produced in the adrenal cortex and regulated by the RAAS pathway (renin-angiotensin-aldosterone system). As the limiting factor in aldosterone production, the aldosterone synthase enzyme is crucial for blood pressure regulation and electrolyte balance.
The aldosterone synthase enzyme catalyzes a reaction involving deoxycorticosterone in a multistep process to form aldosterone. Deoxycorticosterone is a steroid hormone produced by the adrenal glands, and it can also regulate potassium, similar to aldosterone. The key, though, is that deoxycorticosterone doesn’t usually decrease sodium levels much compared to potassium.
The precursor for deoxycorticosterone (and thus, aldosterone) is progesterone. The hormone progesterone is synthesized in the body from cholesterol.
What does high aldosterone cause?
High aldosterone is linked to:[ref][ref]
- high blood pressure
- stroke
- headaches, fatigue (related to low potassium known as ‘hypokalemia’)
- osteoporosis
Related Article: Osteoporosis Genes and Prevention Strategies
What does very low aldosterone cause?
Very low aldosterone levels are a serious problem. Lack of aldosterone, often found in Cushing’s disease, is linked to salt-wasting, growth retardation in children, and failure to thrive. Altered potassium levels and low blood pressure can also occur.[ref][ref]
CYP11B2 Genotype Report
Access this content:
An active subscription is required to access this content.
Lifehacks:
Medications for high blood pressure:
People with hypertension along with the G/G genotype for rs1799998, may respond well to certain blood pressure-reducing drugs ending in -sartans. The -sartan drugs are angiotensin II receptor blockers.[ref][ref] These agents block the action of angiotensin II thus causing vasodilation and increased sodium and water excretion. (Talk with your doctor, of course!)
Aldosterone synthase inhibitors are a new class of hypertensive drugs under development. Inhibiting CYP11B2 decreases aldosterone and subsequently increases deoxycorticosterone, thus changing the sodium-potassium balance.[ref]
Researchers are also looking at ways to reduce aldosterone by stopping the stimulation of aldosterone in the adrenals. Substance P binds to a receptor to increase aldosterone synthase and blocking that receptor with drugs (in clinical trials) reduces aldosterone.[ref] If Substance P sounds familiar, it is a neuropeptide released in pain, vomiting, and hot flashes. In the adrenal glands, substance P partly influences aldosterone stimulation.
Natural ways to reduce high blood pressure with CYP11B2:
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
References:
Antonov, E. V., et al. “Aldosterone and Stress-Dependent Arterial Hypertension.” Bulletin of Experimental Biology and Medicine, vol. 152, no. 2, Dec. 2011, pp. 188–91. PubMed, https://doi.org/10.1007/s10517-011-1484-8.
Azimi-Nezhad, Mohsen, et al. “Association of CYP11B2 Gene Polymorphism with Preeclampsia in North East of Iran (Khorasan Province).” Gene, vol. 733, Apr. 2020, p. 144358. PubMed, https://doi.org/10.1016/j.gene.2020.144358.
Azizi, Michel, et al. “Aldosterone Synthase Inhibition in Humans.” Nephrology, Dialysis, Transplantation: Official Publication of the European Dialysis and Transplant Association – European Renal Association, vol. 28, no. 1, Jan. 2013, pp. 36–43. PubMed, https://doi.org/10.1093/ndt/gfs388.
“Figure: Regulating Blood Pressure: The Renin-Angiotensin-Aldosterone System.” Merck Manuals Consumer Version, https://www.merckmanuals.com/home/multimedia/figure/cvs_regulating_blood_pressure_renin. Accessed 20 Sept. 2021.
Gideon, Angelina, et al. “Kinetics and Interrelations of the Renin Aldosterone Response to Acute Psychosocial Stress: A Neglected Stress System.” The Journal of Clinical Endocrinology and Metabolism, vol. 105, no. 3, Mar. 2020, p. dgz190. PubMed, https://doi.org/10.1210/clinem/dgz190.
Gong, Hongtao, et al. “Association of Polymorphisms of CYP11B2 Gene -344C/T and ACE Gene I/D with Antihypertensive Response to Angiotensin Receptor Blockers in Chinese with Hypertension.” Journal of Genetics, vol. 98, Mar. 2019, p. 1.
Gorini, Stefania, et al. “Mineralocorticoid Receptor and Aldosterone-Related Biomarkers of End-Organ Damage in Cardiometabolic Disease.” Biomolecules, vol. 8, no. 3, Sept. 2018, p. 96. PubMed Central, https://doi.org/10.3390/biom8030096.
Jia, En-Zhi, et al. “Renin-Angiotensin-Aldosterone System Gene Polymorphisms and Coronary Artery Disease: Detection of Gene-Gene and Gene-Environment Interactions.” Cellular Physiology and Biochemistry: International Journal of Experimental Cellular Physiology, Biochemistry, and Pharmacology, vol. 29, no. 3–4, 2012, pp. 443–52. PubMed, https://doi.org/10.1159/000338498.
—. “Renin-Angiotensin-Aldosterone System Gene Polymorphisms and Coronary Artery Disease: Detection of Gene-Gene and Gene-Environment Interactions.” Cellular Physiology and Biochemistry: International Journal of Experimental Cellular Physiology, Biochemistry, and Pharmacology, vol. 29, no. 3–4, 2012, pp. 443–52. PubMed, https://doi.org/10.1159/000338498.
Kurland, Lisa, et al. “Aldosterone Synthase (CYP11B2) -344 C/T Polymorphism Is Related to Antihypertensive Response: Result from the Swedish Irbesartan Left Ventricular Hypertrophy Investigation versus Atenolol (SILVHIA) Trial.” American Journal of Hypertension, vol. 15, no. 5, May 2002, pp. 389–93. PubMed, https://doi.org/10.1016/s0895-7061(02)02256-2.
Li, Xinghui, et al. “CYP11B2 Gene Polymorphism and Essential Hypertension among Tibetan, Dongxiang and Han Populations from Northwest of China.” Clinical and Experimental Hypertension (New York, N.Y.: 1993), vol. 38, no. 4, 2016, pp. 375–80. PubMed, https://doi.org/10.3109/10641963.2015.1131287.
Li, Yan-yan, et al. “CYP11B2 T-344C Gene Polymorphism and Atrial Fibrillation: A Meta-Analysis of 2,758 Subjects.” PloS One, vol. 7, no. 11, 2012, p. e50910. PubMed, https://doi.org/10.1371/journal.pone.0050910.
Mulatero, Paolo, et al. “CYP11B2 -344T/C Gene Polymorphism and Blood Pressure in Patients with Acromegaly.” The Journal of Clinical Endocrinology and Metabolism, vol. 91, no. 12, Dec. 2006, pp. 5008–12. PubMed, https://doi.org/10.1210/jc.2006-0049.
Munshi, Anjana, et al. “Association of the −344C/T Aldosterone Synthase (CYP11B2) Gene Variant with Hypertension and Stroke.” Journal of the Neurological Sciences, vol. 296, no. 1, Sept. 2010, pp. 34–38. ScienceDirect, https://doi.org/10.1016/j.jns.2010.06.013.
Peter, M., et al. “Hereditary Defect in Biosynthesis of Aldosterone: Aldosterone Synthase Deficiency 1964-1997.” The Journal of Clinical Endocrinology and Metabolism, vol. 82, no. 11, Nov. 1997, pp. 3525–28. PubMed, https://doi.org/10.1210/jcem.82.11.4399.
“ClinPGx.” ClinPGx, https://www.clinpgx.org/summaryAnnotation/1183614600. Accessed 20 Sept. 2021.
“—.” ClinPGx, https://www.clinpgx.org/summaryAnnotation/1183614600. Accessed 20 Sept. 2021.
“Primary Aldosteronism – Symptoms and Causes.” Mayo Clinic, https://www.mayoclinic.org/diseases-conditions/primary-aldosteronism/symptoms-causes/syc-20351803. Accessed 20 Sept. 2021.
Rs28931609 RefSNP Report – DbSNP – NCBI. https://www.ncbi.nlm.nih.gov/snp/rs28931609?horizontal_tab=true#clinical_significance. Accessed 20 Sept. 2021.
Rs61757294 RefSNP Report – DbSNP – NCBI. https://www.ncbi.nlm.nih.gov/snp/rs61757294#clinical_significance. Accessed 20 Sept. 2021.
Rs104894072 RefSNP Report – DbSNP – NCBI. https://www.ncbi.nlm.nih.gov/snp/rs104894072?horizontal_tab=true#clinical_significance. Accessed 20 Sept. 2021.
Schoppen, Stefanie, et al. “Sodium-Bicarbonated Mineral Water Decreases Aldosterone Levels without Affecting Urinary Excretion of Bone Minerals.” International Journal of Food Sciences and Nutrition, vol. 59, no. 4, June 2008, pp. 347–55. PubMed, https://doi.org/10.1080/09637480701560308.
Toxqui, Laura, and M. Pilar Vaquero. “Aldosterone Changes after Consumption of a Sodium-Bicarbonated Mineral Water in Humans. A Four-Way Randomized Controlled Trial.” Journal of Physiology and Biochemistry, vol. 72, no. 4, Dec. 2016, pp. 635–41. PubMed, https://doi.org/10.1007/s13105-016-0502-8.
Wang, Lijuan, et al. “Association between Single-Nucleotide Polymorphisms in Six Hypertensive Candidate Genes and Hypertension among Northern Han Chinese Individuals.” Hypertension Research: Official Journal of the Japanese Society of Hypertension, vol. 37, no. 12, Dec. 2014, pp. 1068–74. PubMed, https://doi.org/10.1038/hr.2014.124.
Wils, Julien, et al. “The Neuropeptide Substance P Regulates Aldosterone Secretion in Human Adrenals.” Nature Communications, vol. 11, no. 1, May 2020, p. 2673. PubMed, https://doi.org/10.1038/s41467-020-16470-8.
Young, William F. “Primary Aldosteronism: Renaissance of a Syndrome.” Clinical Endocrinology, vol. 66, no. 5, May 2007, pp. 607–18. PubMed, https://doi.org/10.1111/j.1365-2265.2007.02775.x.