Just over fifteen years ago, researchers discovered that a mutation in the PSCK9 gene caused really high cholesterol levels. This led to more discoveries about the gene and fast-tracked the development of medication for people with hypercholesterolemia.[ref]
This article looks at the PCSK9 genetic variants that are associated with either increased or decreased LDL cholesterol — along with the long-term implications of both sides of that coin. It concludes with information on natural ways to lower LDL that work specifically for people with the PCSK9 variants that increase cholesterol levels.
What does the PCSK9 gene do?
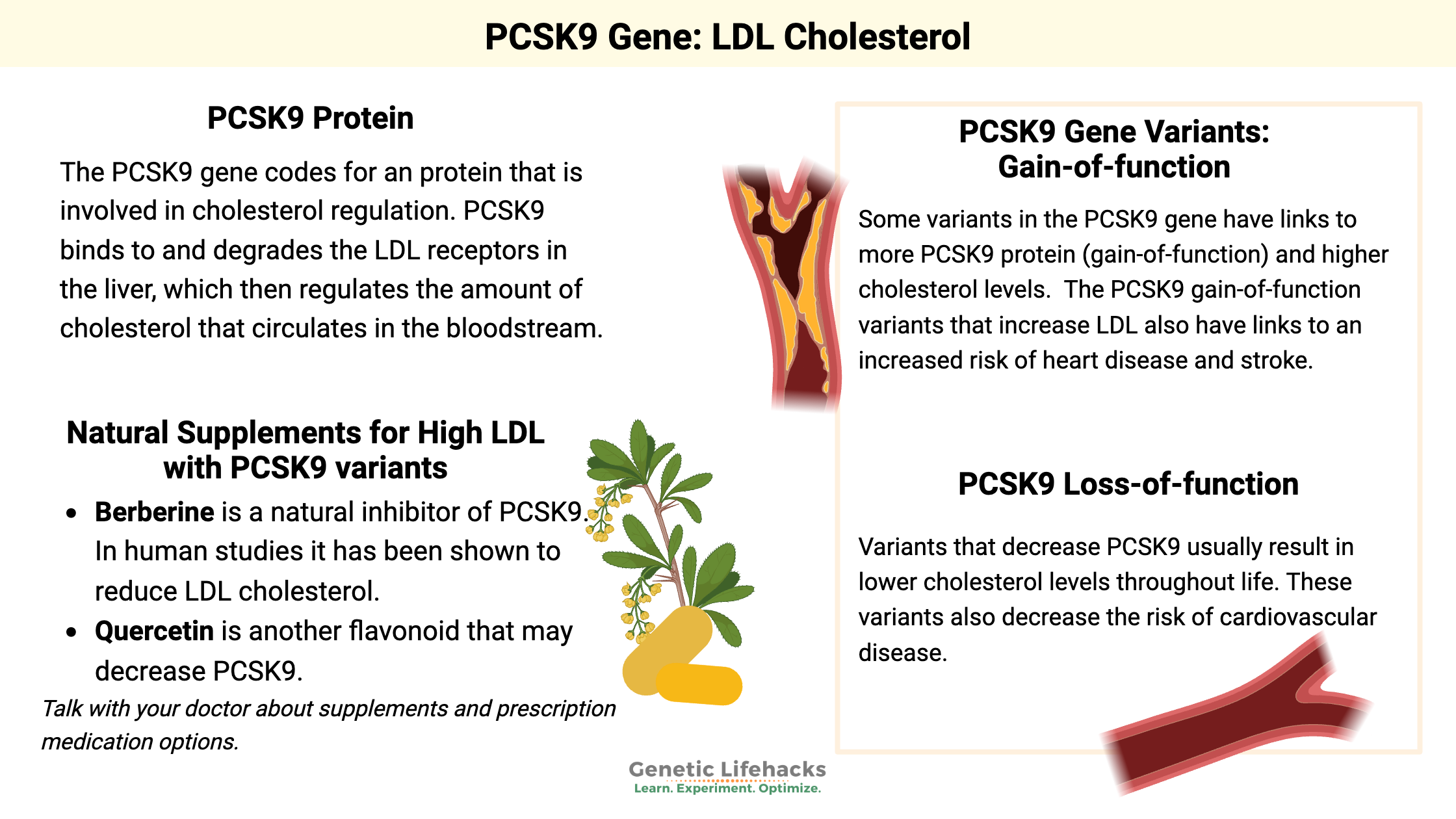
The PCSK9 gene codes for an enzyme that is involved in cholesterol regulation. PCSK9 binds to the receptor for LDL particles and degrades it so that isn’t accessible to bind to cholesterol.
PCSK9 plays a regulatory role in keeping cholesterol at the right level by controlling the number of LDL receptors in liver cells. The liver regulates cholesterol levels in the body – both by synthesis and elimination.
If you don’t have enough LDL receptors taking up cholesterol, you will have an increase in cholesterol in the bloodstream.
On the other hand, if you have more LDL receptors, your cholesterol levels in the blood will be lower.
Thus, it was quite a breakthrough when researchers figured out that blocking PCSK9 causes an increase in LDL receptors in the liver — which causes a decrease in overall cholesterol levels.
PCSK9 genetic variants and high cholesterol:
Cholesterol levels in the bloodstream are partly due to diet, however, the bigger player in cholesterol levels is genetics.
Some variants in the PCSK9 gene have links to more PCSK9 protein (gain-of-function) and higher cholesterol levels. A few of the mutations can lead to really high LDL levels, known as familial hypercholesterolemia. Other variants just increase LDL levels a little so that they are somewhat higher than normal.[ref]
PCSK9 variants and low cholesterol:
After researchers discovered the link between PCSK9 mutations and high cholesterol in 2003, other research showed that there are PCSK9 variants (loss-of-function) that lead to lower cholesterol levels throughout life. Variants with decreased function will cause more LDL receptors in the liver, thus causing more LDL particles (including cholesterol) to be removed from the bloodstream.
Therefore, loss-of-function variants are linked with lower lifetime LDL cholesterol levels and a lower risk of heart disease.[ref][ref] These variants are also associated with a decreased risk of mortality from sepsis.[ref]
PCSK9 and heart disease:
The PCSK9 gain-of-function variants that increase LDL also have links to an increased risk of heart disease and stroke.
A recent meta-analysis that included over 5,000 people found that people with two copies of a PCSK9 variant that increases LDL had more than 2-fold increased odds of having coronary artery disease.[ref]
Another study that followed participants for 18 years found that a PCSK9 variant is associated with increased arterial plaque. Higher PCSK9 levels were associated with a 2-fold risk of having arterial plaques.[ref]
PCSK9 and inflammation in atherosclerosis:
One connection between heart disease and higher PCSK9 levels is that there will be a higher circulating level of cholesterol, including oxidized cholesterol which is inflammatory. However, there is more to atherosclerosis than just a high circulating cholesterol level, and PCSK9 may be playing two roles here.
Higher PCSK9 levels also interact with macrophages and increase inflammatory cytokines. Research shows that in atherosclerosis, PCSK9 recruits inflammatory macrophages to the site of the atherosclerotic plaque. [ref]
PCSK9 and cancer prevention:
There has been quite a bit of research in the last five years or so looking at how PCSK9 levels interact with cancer risk and, more recently, the risk of cancer metastasis or recurrence.
Related article: PCSK9: Cancer Recurrence & Prevention
PCSK9 Genotype Report:
Access this content:
An active subscription is required to access this content.
Lifehacks for lowering LDL:
Natural supplements for LDL and PCSK9:
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
Diet and Blood Pressure: ACE Gene Variants and Saturated Fat
Research shows that people with the ACE deletion genotype will likely have increased blood pressure on a high-fat diet. Find out how a high-fat diet interacts with your genes.
Triglycerides- Genetic variants that impact your triglyceride levels
High triglycerides are linked with an increased risk of cardiovascular disease. Both genetics and diet combine to elevate triglyceride levels. Learn how your genes interact with what you eat to lower your triglycerides.
CYP2D6: Variants that cause reactions to common medications
CYP2D6 is responsible for the breakdown and elimination of about 25% of prescription medications. Genetic variants in CYP2D6 can significantly impact the way that you react to certain drugs.
BPIFB4 Gene Linked to Longevity and Heart Health
Discover the impact of the BPIFB4 gene on longevity and immunity, and find out if you have the longevity variant in your genotype report.
References:
Adorni, Maria Pia, et al. “Effect of a Novel Nutraceutical Combination on Serum Lipoprotein Functional Profile and Circulating PCSK9.” Therapeutics and Clinical Risk Management, vol. 13, 2017, pp. 1555–62. PubMed, https://doi.org/10.2147/TCRM.S144121.
Benn, Marianne, et al. “PCSK9R46L, Low-Density Lipoprotein Cholesterol Levels, and Risk of Ischemic Heart Disease: 3 Independent Studies and Meta-Analyses.” Journal of the American College of Cardiology, vol. 55, no. 25, June 2010, pp. 2833–42. ScienceDirect, https://doi.org/10.1016/j.jacc.2010.02.044.
—. “PCSK9R46L, Low-Density Lipoprotein Cholesterol Levels, and Risk of Ischemic Heart Disease: 3 Independent Studies and Meta-Analyses.” Journal of the American College of Cardiology, vol. 55, no. 25, June 2010, pp. 2833–42. ScienceDirect, https://doi.org/10.1016/j.jacc.2010.02.044.
Blekkenhorst, Lauren C., et al. “Cruciferous and Allium Vegetable Intakes Are Inversely Associated With 15‐Year Atherosclerotic Vascular Disease Deaths in Older Adult Women.” Journal of the American Heart Association: Cardiovascular and Cerebrovascular Disease, vol. 6, no. 10, Oct. 2017, p. e006558. PubMed Central, https://doi.org/10.1161/JAHA.117.006558.
Cao, Shijie, et al. “Berberrubine and Its Analog, Hydroxypropyl-Berberrubine, Regulate LDLR and PCSK9 Expression via the ERK Signal Pathway to Exert Cholesterol-Lowering Effects in Human Hepatoma HepG2 Cells.” Journal of Cellular Biochemistry, Oct. 2018. PubMed, https://doi.org/10.1002/jcb.27102.
Chen, Suet N., et al. “A Common PCSK9 Haplotype, Encompassing the E670G Coding Single Nucleotide Polymorphism, Is a Novel Genetic Marker for Plasma Low-Density Lipoprotein Cholesterol Levels and Severity of Coronary Atherosclerosis.” Journal of the American College of Cardiology, vol. 45, no. 10, May 2005, pp. 1611–19. PubMed, https://doi.org/10.1016/j.jacc.2005.01.051.
Chikowore, Tinashe, et al. “C679X Loss-of-Function PCSK9 Variant Lowers Fasting Glucose Levels in a Black South African Population: A Longitudinal Study.” Diabetes Research and Clinical Practice, vol. 144, Oct. 2018, pp. 279–85. PubMed, https://doi.org/10.1016/j.diabres.2018.09.012.
Cohen, Jonathan, et al. “Low LDL Cholesterol in Individuals of African Descent Resulting from Frequent Nonsense Mutations in PCSK9.” Nature Genetics, vol. 37, no. 2, Feb. 2005, pp. 161–65. PubMed, https://doi.org/10.1038/ng1509.
—. “Low LDL Cholesterol in Individuals of African Descent Resulting from Frequent Nonsense Mutations in PCSK9.” Nature Genetics, vol. 37, no. 2, Feb. 2005, pp. 161–65. PubMed, https://doi.org/10.1038/ng1509.
—. “Low LDL Cholesterol in Individuals of African Descent Resulting from Frequent Nonsense Mutations in PCSK9.” Nature Genetics, vol. 37, no. 2, Feb. 2005, pp. 161–65. PubMed, https://doi.org/10.1038/ng1509.
Dong, Bin, et al. “Inhibition of PCSK9 Transcription by Berberine Involves Down-Regulation of Hepatic HNF1α Protein Expression through the Ubiquitin-Proteasome Degradation Pathway *.” Journal of Biological Chemistry, vol. 290, no. 7, Feb. 2015, pp. 4047–58. www.jbc.org, https://doi.org/10.1074/jbc.M114.597229.
Ferreira, João Pedro, et al. “PCSK9 Protein and Rs562556 Polymorphism Are Associated With Arterial Plaques in Healthy Middle‐Aged Population: The STANISLAS Cohort.” Journal of the American Heart Association: Cardiovascular and Cerebrovascular Disease, vol. 9, no. 7, Mar. 2020, p. e014758. PubMed Central, https://doi.org/10.1161/JAHA.119.014758.
Genga, Kelly Roveran, et al. “Impact of PCSK9 Loss-of-Function Genotype on 1-Year Mortality and Recurrent Infection in Sepsis Survivors.” EBioMedicine, vol. 38, Nov. 2018, pp. 257–64. PubMed Central, https://doi.org/10.1016/j.ebiom.2018.11.032.
Kent, Shia T., et al. “PCSK9 Loss-of-Function Variants, Low-Density Lipoprotein Cholesterol, and Risk of Coronary Heart Disease and Stroke: Data From 9 Studies of Blacks and Whites.” Circulation. Cardiovascular Genetics, vol. 10, no. 4, Aug. 2017, p. e001632. PubMed, https://doi.org/10.1161/CIRCGENETICS.116.001632.
—. “PCSK9 Loss-of-Function Variants, Low-Density Lipoprotein Cholesterol, and Risk of Coronary Heart Disease and Stroke: Data From 9 Studies of Blacks and Whites.” Circulation. Cardiovascular Genetics, vol. 10, no. 4, Aug. 2017, p. e001632. PubMed, https://doi.org/10.1161/CIRCGENETICS.116.001632.
Kwon, H. J., et al. “Molecular Basis for LDL Receptor Recognition by PCSK9.” Proceedings of the National Academy of Sciences, vol. 105, no. 6, Feb. 2008, pp. 1820–25. DOI.org (Crossref), https://doi.org/10.1073/pnas.0712064105.
—. “Molecular Basis for LDL Receptor Recognition by PCSK9.” Proceedings of the National Academy of Sciences, vol. 105, no. 6, Feb. 2008, pp. 1820–25. DOI.org (Crossref), https://doi.org/10.1073/pnas.0712064105.
Li, Yan-yan, et al. “PCSK9 Gene E670G Polymorphism and Coronary Artery Disease: An Updated Meta-Analysis of 5,484 Subjects.” Frontiers in Cardiovascular Medicine, vol. 7, Nov. 2020, p. 582865. PubMed Central, https://doi.org/10.3389/fcvm.2020.582865.
—. “PCSK9 Gene E670G Polymorphism and Coronary Artery Disease: An Updated Meta-Analysis of 5,484 Subjects.” Frontiers in Cardiovascular Medicine, vol. 7, Nov. 2020, p. 582865. PubMed Central, https://doi.org/10.3389/fcvm.2020.582865.
Mbikay, Majambu, et al. “Mice Fed a High-Cholesterol Diet Supplemented with Quercetin-3-Glucoside Show Attenuated Hyperlipidemia and Hyperinsulinemia Associated with Differential Regulation of PCSK9 and LDLR in Their Liver and Pancreas.” Molecular Nutrition & Food Research, vol. 62, no. 9, May 2018, p. e1700729. PubMed, https://doi.org/10.1002/mnfr.201700729.
Postmus, Iris, et al. “PCSK9 SNP Rs11591147 Is Associated with Low Cholesterol Levels but Not with Cognitive Performance or Noncardiovascular Clinical Events in an Elderly Population.” Journal of Lipid Research, vol. 54, no. 2, Feb. 2013, pp. 561–66. PubMed Central, https://doi.org/10.1194/jlr.M033969.
Qiu, Chengfeng, et al. “What Is the Impact of PCSK9 Rs505151 and Rs11591147 Polymorphisms on Serum Lipids Level and Cardiovascular Risk: A Meta-Analysis.” Lipids in Health and Disease, vol. 16, no. 1, June 2017, p. 111. PubMed, https://doi.org/10.1186/s12944-017-0506-6.
—. “What Is the Impact of PCSK9 Rs505151 and Rs11591147 Polymorphisms on Serum Lipids Level and Cardiovascular Risk: A Meta-Analysis.” Lipids in Health and Disease, vol. 16, no. 1, June 2017, p. 111. PubMed, https://doi.org/10.1186/s12944-017-0506-6.
Shapiro, Michael D., et al. “PCSK9: From Basic Science Discoveries to Clinical Trials.” Circulation Research, vol. 122, no. 10, May 2018, pp. 1420–38. PubMed Central, https://doi.org/10.1161/CIRCRESAHA.118.311227.
Slimani, Afef, et al. “Effect of E670G Polymorphism in PCSK9 Gene on the Risk and Severity of Coronary Heart Disease and Ischemic Stroke in a Tunisian Cohort.” Journal of Molecular Neuroscience: MN, vol. 53, no. 2, June 2014, pp. 150–57. PubMed, https://doi.org/10.1007/s12031-014-0238-2.