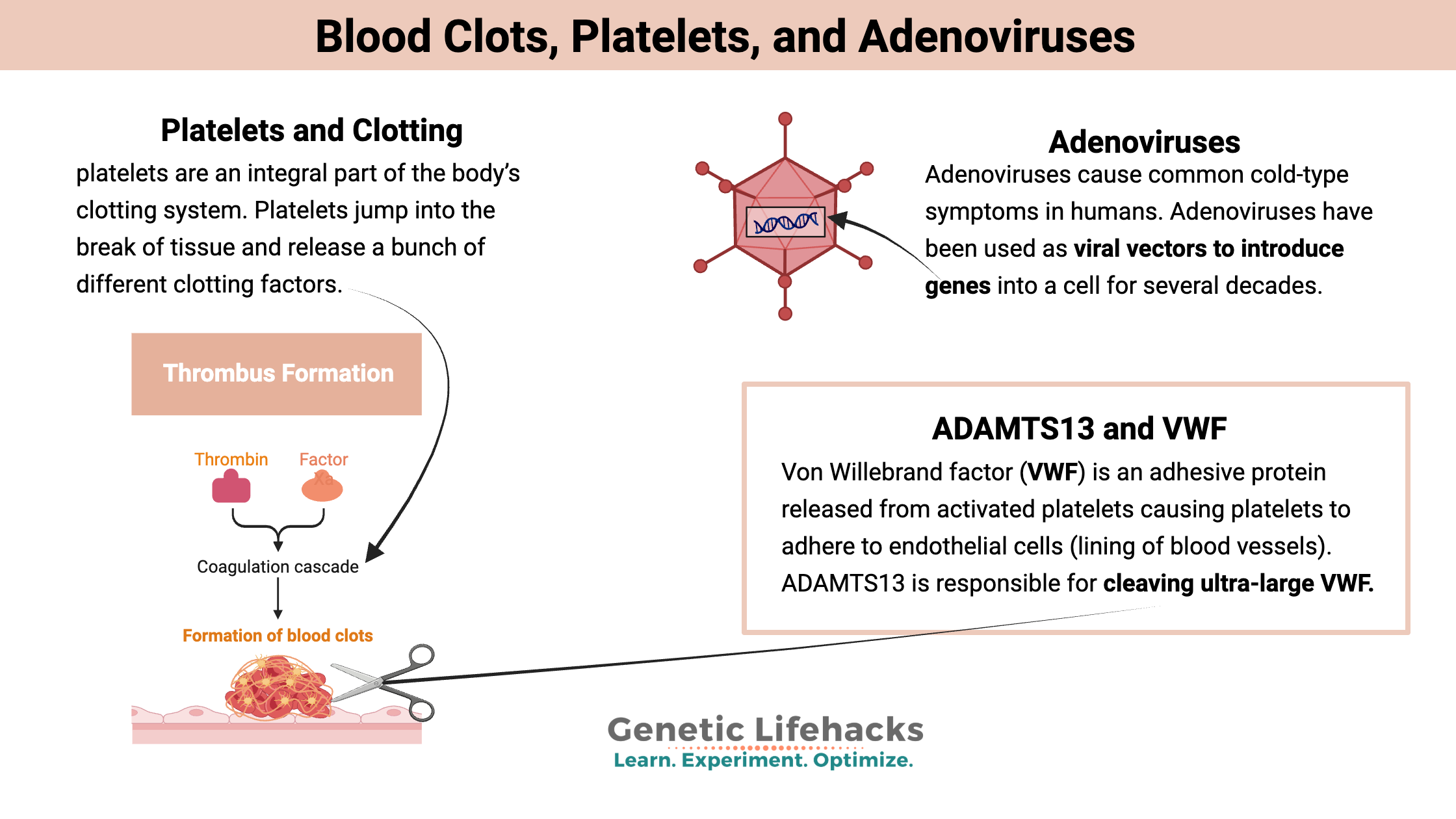
Most people think of platelets forming a clot after they’ve cut themselves — and this is a vital role of platelets. But platelets also do a lot to protect us from getting sick from bacteria and viruses.
In this article, I’ll explain how platelets interact with the immune system – and how a low platelet count (thrombocytopenia) relates to blood clots (thrombosis) in some instances. Then I’ll explain prior research on adenovirus-vector therapy, thrombocytopenia, and platelet reactions.
Recently, governments around the world have paused adenovirus-vector COVID-19 vaccines due to a really rare occurrence of blood clots. Let me be clear upfront: The current stoppage of injecting adenovirus-based vaccinations is due to an undetermined cause of blood clots. The information presented in this article is for background and educational purposes.
Abbink, Peter, et al. “Comparative Seroprevalence and Immunogenicity of Six Rare Serotype Recombinant Adenovirus Vaccine Vectors from Subgroups B and D.” Journal of Virology, vol. 81, no. 9, May 2007, pp. 4654–63. PubMed, https://doi.org/10.1128/JVI.02696-06.
Aldrighi, José M., et al. “Platelet Activation Status Decreases after Menopause.” Gynecological Endocrinology: The Official Journal of the International Society of Gynecological Endocrinology, vol. 20, no. 5, May 2005, pp. 249–57. PubMed, https://doi.org/10.1080/09513590500097549.
Assinger, Alice. “Platelets and Infection – An Emerging Role of Platelets in Viral Infection.” Frontiers in Immunology, vol. 5, Dec. 2014, p. 649. PubMed Central, https://doi.org/10.3389/fimmu.2014.00649.
Atasheva, Svetlana, et al. “Innate Immunity to Adenovirus: Lessons from Mice.” FEBS Letters, vol. 593, no. 24, Dec. 2019, pp. 3461–83. PubMed Central, https://doi.org/10.1002/1873-3468.13696.
Banerjee, Meenakshi, et al. “Platelets Endocytose Viral Particles and Are Activated via TLR (Toll-Like Receptor) Signaling.” Arteriosclerosis, Thrombosis, and Vascular Biology, vol. 40, no. 7, July 2020, pp. 1635–50. DOI.org (Crossref), https://doi.org/10.1161/ATVBAHA.120.314180.
Berlin, G., et al. “Effects of Age, Gender and Menstrual Cycle on Platelet Function Assessed by Impedance Aggregometry.” Platelets, vol. 30, no. 4, 2019, pp. 473–79. PubMed, https://doi.org/10.1080/09537104.2018.1466387.
———. “Effects of Age, Gender and Menstrual Cycle on Platelet Function Assessed by Impedance Aggregometry.” Platelets, vol. 30, no. 4, May 2019, pp. 473–79. DOI.org (Crossref), https://doi.org/10.1080/09537104.2018.1466387.
Custers, Jerome, et al. “Vaccines Based on Replication Incompetent Ad26 Viral Vectors: Standardized Template with Key Considerations for a Risk/Benefit Assessment.” Vaccine, vol. 39, no. 22, May 2021, pp. 3081–101. ScienceDirect, https://doi.org/10.1016/j.vaccine.2020.09.018.
Excoffon, Katherine J. D. A. “The Coxsackievirus and Adenovirus Receptor: Virological and Biological Beauty.” FEBS Letters, vol. 594, no. 12, June 2020, pp. 1828–37. DOI.org (Crossref), https://doi.org/10.1002/1873-3468.13794.
Gupalo, Elena, et al. “Platelet-Adenovirus vs. Inert Particles Interaction: Effect on Aggregation and the Role of Platelet Membrane Receptors.” Platelets, vol. 24, no. 5, 2013, pp. 383–91. PubMed, https://doi.org/10.3109/09537104.2012.703792.
Hendrie, Paul C., and David W. Russell. “Gene Targeting with Viral Vectors.” Molecular Therapy, vol. 12, no. 1, July 2005, pp. 9–17. ScienceDirect, https://doi.org/10.1016/j.ymthe.2005.04.006.
Jin, Ying-Yu, et al. “Adenovirus Type 3 Induces Platelet Activation in Vitro.” Molecular Medicine Reports, vol. 9, no. 1, Jan. 2014, pp. 370–74. www.spandidos-publications.com, https://doi.org/10.3892/mmr.2013.1805.
Lichtenstein, Drew L., and William S. M. Wold. “Experimental Infections of Humans with Wild-Type Adenoviruses and with Replication-Competent Adenovirus Vectors: Replication, Safety, and Transmission.” Cancer Gene Therapy, vol. 11, no. 12, Dec. 2004, pp. 819–29. www.nature.com, https://doi.org/10.1038/sj.cgt.7700765.
Lopez-Gordo, Estrella, et al. “Circumventing Antivector Immunity: Potential Use of Nonhuman Adenoviral Vectors.” Human Gene Therapy, vol. 25, no. 4, Apr. 2014, pp. 285–300. PubMed Central, https://doi.org/10.1089/hum.2013.228.
Merchant, Hamid A. “CoViD Vaccines and Thrombotic Events: EMA Issued Warning to Patients and Healthcare Professionals.” Journal of Pharmaceutical Policy and Practice, vol. 14, no. 1, Mar. 2021, p. 32. Springer Link, https://doi.org/10.1186/s40545-021-00315-w.
Mitani, K., and S. Kubo. “Adenovirus as an Integrating Vector.” Current Gene Therapy, vol. 2, no. 2, May 2002, pp. 135–44. PubMed, https://doi.org/10.2174/1566523024605591.
Nathwani, Amit C., et al. “Adenovirus-Associated Virus Vector-Mediated Gene Transfer in Hemophilia B.” The New England Journal of Medicine, vol. 365, no. 25, Dec. 2011, pp. 2357–65. PubMed, https://doi.org/10.1056/NEJMoa1108046.
Othman, Maha, et al. “Adenovirus-Induced Thrombocytopenia: The Role of von Willebrand Factor and P-Selectin in Mediating Accelerated Platelet Clearance.” Blood, vol. 109, no. 7, Apr. 2007, pp. 2832–39. ScienceDirect, https://doi.org/10.1182/blood-2006-06-032524.
Page, Martin J., and Etheresia Pretorius. “A Champion of Host Defense: A Generic Large-Scale Cause for Platelet Dysfunction and Depletion in Infection.” Seminars in Thrombosis and Hemostasis, vol. 46, no. 3, Apr. 2020, pp. 302–19. PubMed Central, https://doi.org/10.1055/s-0040-1708827.
Planty, Camille, et al. “Nonclinical Safety Assessment of Repeated Administration and Biodistribution of ChAd3‐EBO‐Z Ebola Candidate Vaccine.” Journal of Applied Toxicology, vol. 40, no. 6, June 2020, pp. 748–62. PubMed Central, https://doi.org/10.1002/jat.3941.
Potter, Robert N., et al. “Adenovirus-Associated Deaths in US Military during Postvaccination Period, 1999–2010.” Emerging Infectious Diseases, vol. 18, no. 3, Mar. 2012, pp. 507–09. PubMed Central, https://doi.org/10.3201/eid1803.111238.
Rajagopalan, S., et al. “Adenovirus-Mediated Gene Transfer of VEGF(121) Improves Lower-Extremity Endothelial Function and Flow Reserve.” Circulation, vol. 104, no. 7, Aug. 2001, pp. 753–55. PubMed, https://doi.org/10.1161/hc3201.095192.
Raper, Steven E., et al. “Fatal Systemic Inflammatory Response Syndrome in a Ornithine Transcarbamylase Deficient Patient Following Adenoviral Gene Transfer.” Molecular Genetics and Metabolism, vol. 80, nos. 1–2, 2003, pp. 148–58. PubMed, https://doi.org/10.1016/j.ymgme.2003.08.016.
Schultz, Nina H., et al. “Thrombosis and Thrombocytopenia after ChAdOx1 nCoV-19 Vaccination.” New England Journal of Medicine, vol. 384, no. 22, June 2021, pp. 2124–30. DOI.org (Crossref), https://doi.org/10.1056/NEJMoa2104882.
Shirley, Jamie L., et al. “Immune Responses to Viral Gene Therapy Vectors.” Molecular Therapy, vol. 28, no. 3, Mar. 2020, pp. 709–22. ScienceDirect, https://doi.org/10.1016/j.ymthe.2020.01.001.
Stanley, Michael, et al. “Thrombotic Thrombocytopenic Purpura.” StatPearls, StatPearls Publishing, 2025. PubMed, http://www.ncbi.nlm.nih.gov/books/NBK430721/.
Tapia, Milagritos D., Samba O. Sow, Birahim P. Ndiaye, et al. “Safety, Reactogenicity, and Immunogenicity of a Chimpanzee Adenovirus Vectored Ebola Vaccine in Adults in Africa: A Randomised, Observer-Blind, Placebo-Controlled, Phase 2 Trial.” The Lancet. Infectious Diseases, vol. 20, no. 6, June 2020, pp. 707–18. PubMed, https://doi.org/10.1016/S1473-3099(20)30016-5.
Tapia, Milagritos D., Samba O. Sow, Khardiata D. Mbaye, et al. “Safety, Reactogenicity, and Immunogenicity of a Chimpanzee Adenovirus Vectored Ebola Vaccine in Children in Africa: A Randomised, Observer-Blind, Placebo-Controlled, Phase 2 Trial.” The Lancet. Infectious Diseases, vol. 20, no. 6, June 2020, pp. 719–30. PubMed, https://doi.org/10.1016/S1473-3099(20)30019-0.
Wang, Jong-Shyan, et al. “Effects of Short-Term Exercise on Female Platelet Function During Different Phases of the Menstrual Cycle.” Arteriosclerosis, Thrombosis, and Vascular Biology, vol. 17, no. 9, Sept. 1997, pp. 1682–86. DOI.org (Crossref), https://doi.org/10.1161/01.ATV.17.9.1682.
Wolins, N., et al. “Intravenous Administration of Replication‐incompetent Adenovirus to Rhesus Monkeys Induces Thrombocytopenia by Increasing in Vivo Platelet Clearance.” British Journal of Haematology, vol. 123, no. 5, Dec. 2003, pp. 903–05. DOI.org (Crossref), https://doi.org/10.1046/j.1365-2141.2003.04719.x.
Xiao, Shao Wen, et al. “Recombinant Adenovirus-P53 Gene Therapy for Advanced Unresectable Soft-Tissue Sarcomas.” Human Gene Therapy, vol. 29, no. 6, June 2018, pp. 699–707. PubMed, https://doi.org/10.1089/hum.2017.103.