Key takeaways:
- Chronically elevated inflammation causes depression and anxiety, for some people…
- Genetics may help point to whether this is an underlying cause for you.
- Finding your underlying physiological cause of depression or anxiety may lead you to the solutions that are right for you.
This article explains why inflammation causes depression and how your genetic variants in inflammatory genes may play a role in depression or anxiety. Members will see their genotype report below, plus additional solutions in the Lifehacks section. Join today.
Depression and Anxiety: Inflammation as a root cause
Research over the past two decades clearly shows a causal link between increased inflammatory markers and depression. This means that inflammation – elevated levels of inflammatory cytokines – can CAUSE mood disorders, for at least a portion of depressed patients. Addressing inflammation as an underlying factor helps to resolve depression or anxiety for many, but not all. And your genes may hold the key here.[ref][ref]
There’s currently a push in psychiatry to include in the DSM a specific subtype of depression linked to inflammation.[ref] Inflammation-driven depression is characterized by:[ref]
- sleep problems
- tiredness, lack of energy
- changes in appetite
- anhedonia (meh, feeling like nothing matters)
Genetic studies show causality:
One way to determine if inflammation is a root cause of mood disorders is to look at the genetic variants that increase susceptibility to depression or anxiety. In this case, research shows that a bunch of genetic risk factors for mood disorders are in inflammation-related genes.[ref][ref] Deleting certain inflammation genes causes animals to be resistant to stress-induced depression.[ref]
Animal models of depression let researchers experiment to determine causality. Increasing inflammatory cytokines clearly causes depression and anxiety symptoms in animals, and decreasing the inflammation takes away the symptoms.[ref] Additionally, human studies show that drugs that decrease inflammation significantly improve depression in some people.[ref]
Disclaimer: This article is a presentation of the research on depression, anxiety, and inflammation. It is not to be taken as medical advice. If you are under the care of a physician for a mood disorder, please talk with your provider for medical advice.
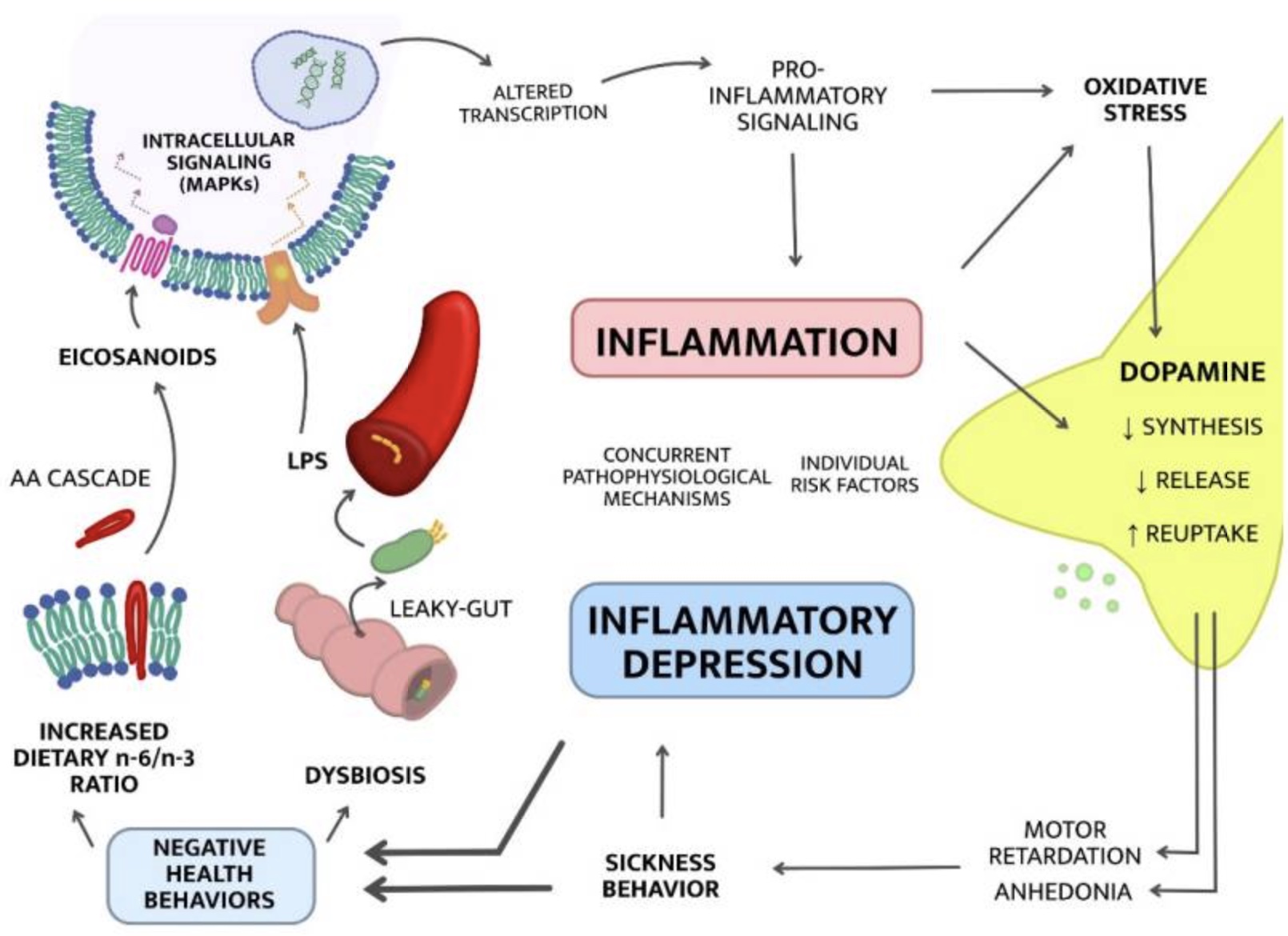
Inflammatory cytokines cause behavior changes:
Behavior changes when you get sick. You’re tired, irritable, and don’t want to interact with people. If you have kids, you learn to spot these signs they’re fighting off ‘a bug’ very quickly. Even in pets, you will see that they withdraw, sleep, and avoid others when they are sick. Appetite changes, and they often are hyper-vigilant as well.
Researchers call this ‘sickness behavior‘, and it’s apparent across all species of animals. It’s instinctive. The best way not to infect your family (or herd, pack, tribe, flock, group) is to curl up in a corner and avoid everyone.
What drives this response? Researchers in the 1980s made a key discovery. Interferon-alpha, an inflammatory cytokine that fights viruses, was first used in the 80s as a treatment for chronic hepatitis. Some patients reacted immediately to the interferon-alpha with psychiatric side effects, including depression, emotional instability, paranoia, agitation, and suicidal potential. These side effects occurred in patients on the highest doses of interferon-alpha, and reducing the dose took away the psychiatric problems.[ref][ref][ref]
5 common causes of inflammation in depression and anxiety:
While this is a huge topic, here are five of the factors that explain why depression and anxiety are so prevalent today.
#1 Gut dysbiosis and increased permeability:
Your intestinal barrier keeps the microbes in your gut separate from the rest of your body. Lining your intestines is a mucosal barrier that keeps the gut microbes away from the epithelial cells, which keep out microbes by being tightly joined together. Reducing the mucosal lining and/or loosening the tight junctions in your intestinal barrier can increase your inflammatory response to your gut bacteria. A study on patients with major depressive disorder showed that patients have higher than average activity of the inflammatory markers for dealing with bacteria from the gut.[ref]
What can cause gut dysbiosis and increased intestinal permeability? A diet that includes a lot of junk food. (read about emulsifiers, food additives, and inflammation)
#2 Autoimmune diseases and allergies:
Many autoimmune diseases, including lupus, multiple sclerosis, psoriasis, and rheumatoid arthritis, have links to an increase in inflammation. People with these autoimmune issues are also at an increased risk of depression. Allergies and asthma also have links to an increased risk of depression. For example, a meta-analysis calculated that the prevalence of depression in people with rheumatoid arthritis is ~39%.[ref][ref][ref][ref][ref][ref]
#3 Stress:
The body undergoes a series of physical changes when stressed. Major life events, such as losing a job, the death of a loved one, moving, or divorce, cause stress in the body. The stress response initiates pro-inflammatory cytokine production to enhance wound healing and improve physical recovery. In fact, to create an animal model of anxiety or depression, researchers simply subject animals to mild, unpredictable stress (tilting the cage, intermittent light, noise).[ref]
#4 Omega 6: Omega 3 ratio:
Consuming a higher ratio of omega-6 to omega-3 fats increases inflammation. Omega-6 fats are found in oils such as soybean, corn, vegetable oil, sunflower, and other seed oils, which are all prevalent in a modern diet. An excess of omega-6 oils causes an increase in overall inflammation.[ref]
A 2026 placebo-controlled clinical trial showed that supplementing with DHA and EPA, omega-3 fatty acids, significantly reduced depression, stress, and anxiety symptoms (compared to a placebo group taking corn oil).[ref]
Why would DHA and EPA help? The resolution of inflammation is an active process that involves specific lipid mediators. The omega-3 fatty acids DHA and EPA are the biological starting points for the cells to create lipid mediators that resolve inflammation.[ref]
Related article: Resolution of inflammation and specialized pro-resolving mediators
#5 Obesity:
Higher overall inflammation goes hand-in-hand with weight gain. Adipose tissue (fat) gives off inflammatory cytokines, increasing the overall burden of inflammation. People who are overweight and have depression also have higher inflammatory markers, on average, than normal-weight people with depression.[ref][ref]
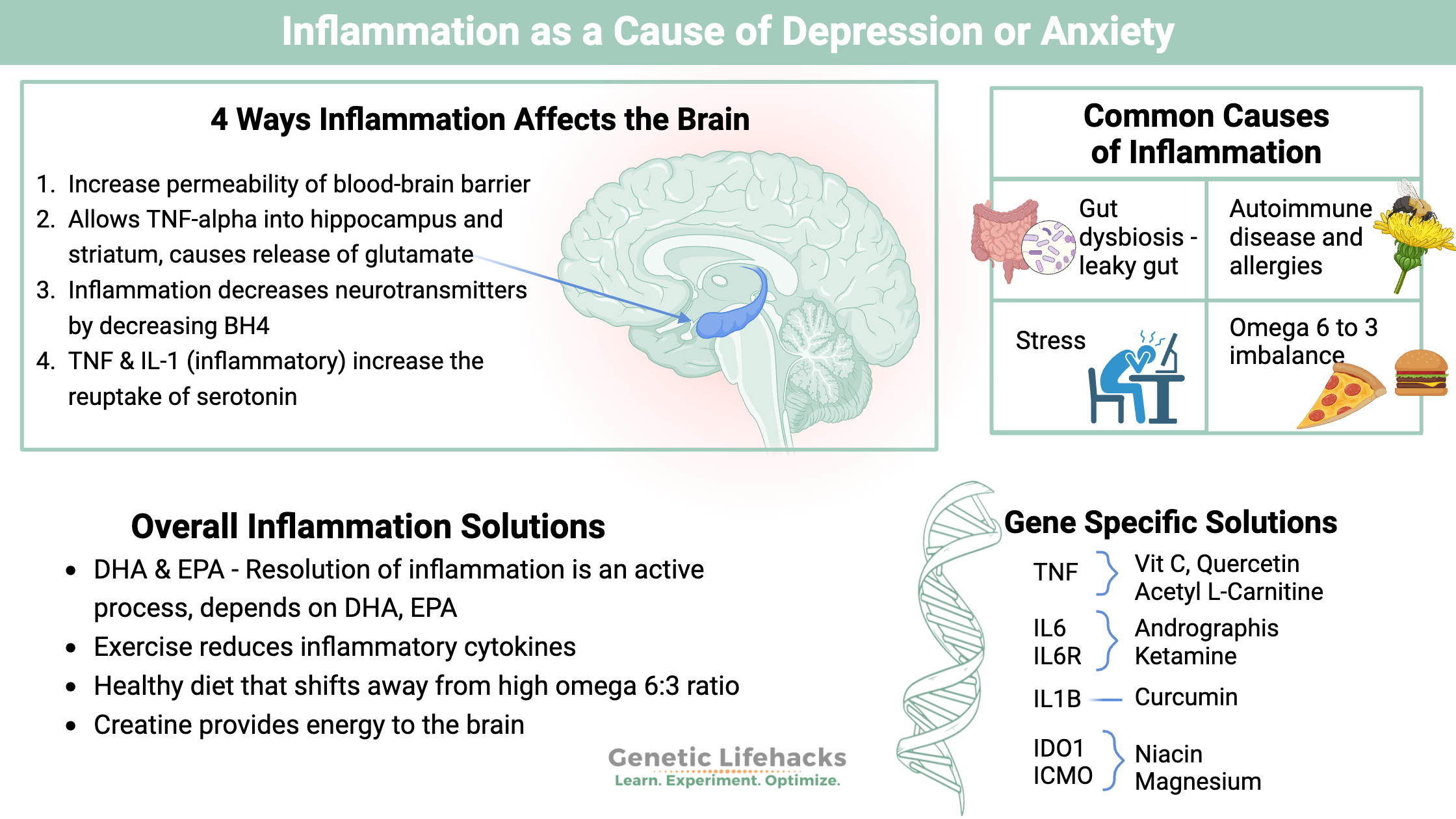
Why does inflammation affect the brain and mood?
There are a couple of ways that inflammation directly affects the brain:
Increased inflammatory cytokines throughout the body will directly increase the permeability of the blood-brain barrier. This allows the inflammatory cytokines to enter the brain, triggering functional changes in certain brain regions.
- Higher levels of TNF-alpha in the hippocampus and striatum (brain regions) have links to anxiety and depression in animal studies. TNF-alpha directly causes the release of glutamate in the hippocampus, causing depressive behavior in animals. (Read about TNF genes)
- IL-1β decreases neurogenesis via the kynurenine pathway.[ref] (read about kynurenine genes)
- Inflammation decreases the synthesis of neurotransmitters through the disruption of BH4, which is needed for dopamine and serotonin synthesis.[ref] (Read about BH4 genes)
- TNF-alpha and IL-1 increase the reuptake of serotonin, leaving less serotonin available.[ref] (read about serotonin genes)
- Increased inflammation shifts the body from converting tryptophan into serotonin and instead produces kynurenine, which has a neurotoxic metabolite called quinolinic acid.[ref] We’ll come back to this in more detail below.
Which inflammatory cytokines are linked to depression or anxiety?
Depression: Research shows that some people with depression have higher levels of TNFα, IL-6, IL-13, IL-18, IL-12, and IL-1RA.[ref] The “IL” stands for interleukin, which is a whole family of inflammatory molecules.
Anxiety: Studies also show that TNF-alpha, IL-6, and C-reactive protein (CRP) have links to anxiety disorders in some people.[ref]
PTSD: Studies on PTSD, which is grouped with anxiety disorders, show that trauma elevates IL-1B, IL-6, TNF-alpha, and C-reactive protein. Generalized anxiety disorder (GAD) has links to increased levels of TNF-alpha and CRP.[ref]
Brain imaging studies point to higher levels of CRP (C-reactive protein), which is a general marker of inflammation and is predictive of psychosis. Additionally, increasing IL-10, which is anti-inflammatory, is linked with greater symptom improvement in depression.[ref]
Scientists have also narrowed down that stress-induced depression is more likely to have pro-inflammatory cytokines as a cause.[ref]
Details on TNF-alpha
TNF-alpha (tumor necrosis factor alpha) is one of the body’s primary inflammatory cytokines. Its production occurs by activated immune cells, such as macrophages, mast cells, B cells, and lymphocytes. It can also be produced in other cells, such as smooth muscle cells, in response to an injury. Adipose tissue (fat tissue) also secretes TNF-alpha.[ref][ref]
When TNF-alpha is elevated in depressed people, blocking TNF-alpha can ameliorate depressive symptoms. A 2008 genome-wide association study found that a TNF genetic variant increases the risk of depression.[ref]
People with rheumatoid arthritis are often given drugs to decrease TNF-alpha. The anti-TNF drugs decrease the frequency of anxiety disorders and depression in people with RA.[ref]
Researchers think that inflammation, specifically high TNF-alpha levels, impacts the HPA axis and elevates cortisol as well. HPA axis dysfunction is strongly linked to depression.
Related article: HPA axis dysfunction: genes and environment
Additionally, higher TNF-alpha levels can cause increased uptake of serotonin into cells, causing depressive symptoms.[ref]
The phrase ‘it’s all in your head’ could be literally true for some people with TNF-alpha variants. A brain-imaging study found that a TNF-alpha genetic variant associated with higher TNF levels was linked to anatomical brain changes in people with major depressive disorder.[ref]
Elevated Interleukin-6
Encoded by the IL6 gene, interleukin-6 (IL-6) is another inflammatory cytokine shown to be consistently elevated in depression.
Animal studies show that inducing an elevated level of IL-6 causes depressive behavior. This can be done using stressful situations or by injecting lipopolysaccharide, an endotoxin from bacteria.[ref] Animals bred not to produce IL-6 are resistant to despair behavior usually induced by stress.[ref]
Genetic studies in humans also link higher levels of IL-6, stressful life events, and an increased risk of depression.[ref]
Interleukin-6 is one of the major cytokines released by immune cells in response to a pathogen or toxin. Chronic or acute stress can also cause the release of IL-6. In the brain, increased levels of IL-6 cause decreased neurotransmitters, including serotonin. Additionally, IL-6 causes a decrease in brain-derived neurotrophic factor (BDNF) and impacts the HPA axis. All of these pathways are implicated in mood disorders.[ref]
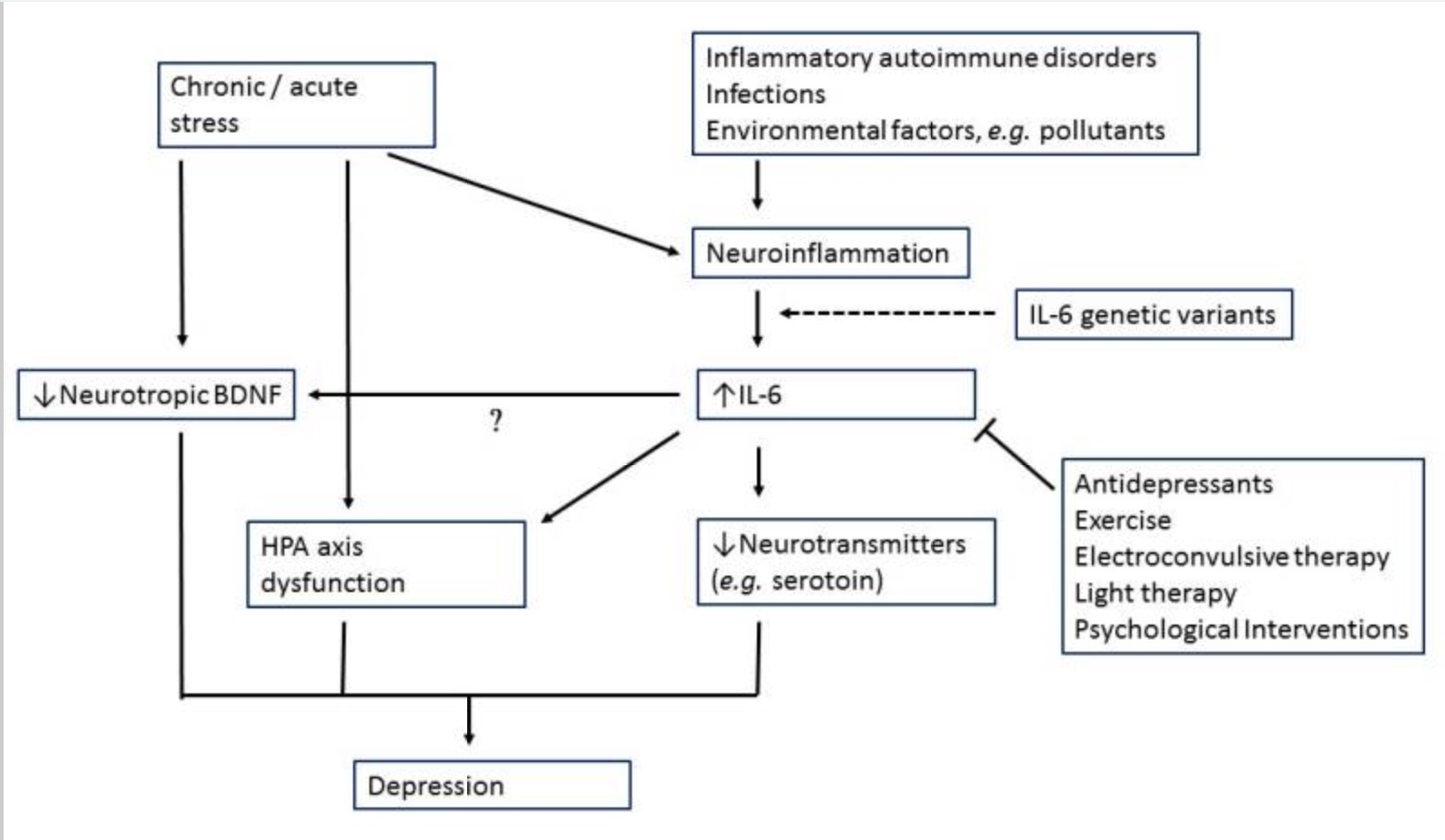
IL-6 also impacts the amygdala’s response to socially threatening situations linking the increased immune response to greater social disconnect, fatigue, and cognitive disturbance.[ref]
Kynurenine pathway:
Tryptophan is an essential amino acid found in many foods that contain protein.
In the body, tryptophan can go down two different pathways:
- tryptophan can be used to make serotonin and melatonin
- tryptophan can convert into kynurenine and then into several other metabolites, including quinolinic acid.
Kynurenine can form quinolinic acid, which is a neurotoxin. This is part of the pathway, though, that eventually produces niacin (vitamin B3).
Tryptophan metabolism needs to balance out. The body and brain need serotonin as an important neurotransmitter. Melatonin, produced from serotonin, is an integral part of the circadian system as well as a vital anti-inflammatory molecule.
Increased inflammation causes a shift from tryptophan being used for serotonin to being used for the kynurenine pathway. This causes a double whammy – lower serotonin and higher quinolinic acid. Quinolinic acid acts on NMDA receptors, and too much can kill off brain cells. Studies show that depression scores correlate to higher quinolinic acid in the blood, and postmortem studies show more cells in the brain producing quinolinic acid in suicide victims.[ref]
Hepatitis C patients treated with interferon-alpha have depression and higher kynurenine metabolites.[ref]
Another reason tryptophan metabolism could tilt more towards kynurenine is when the body is lacking in niacin. Providing enough niacin through the diet is important here.
Genetic variants in the IDO1 gene also can skew the body towards producing kynurenine instead of serotonin. (Read more about tryptophan, kynurenine, serotonin, and genetics.)
Allergies, COVID-19, and depression and anxiety:
A study of the psychological impact of COVID-19 revealed that people with allergies, which chronically raise inflammatory cytokine levels, were more likely to have PTSD and depression after COVID-19.[ref]
Another study found that people with systemically elevated inflammation after COVID-19 illness were at an increased risk of depression, anxiety, or PTSD.[ref]
While there is a lot of swirl and science about long-haul COVID symptoms, previous research on other viruses that remain active or reactivate (e.g., Epstein-Barr virus, cytomegalovirus) also has links between infection, inflammatory cytokines, and depression.[ref]
Inflammation, Depression, and Anxiety: Genotype Report
Access this content:
An active subscription is required to access this content.
Lifehacks:
Talk with your healthcare provider about any supplements or changes – especially if you are on medications.
The lifehacks below deal with ways to suppress inflammatory cytokines, but the obvious solution is to take away the source of the inflammation. Thus, these lifehacks may help in the immediate timeframe, but for a long-term solution, you need to figure out the source of your inflammation (e.g., obesity, stress, diet, gut dysbiosis).
You may find that multiple solutions stacked together work best. For example, curcumin + exercise + an anti-inflammatory diet may be more effective than just popping a curcumin supplement.
Anti-inflammatory OTC medications:
- Aspirin: Aspirin is a COX-1 and COX-2 inhibitor, which reduces TNF-alpha, IL-6, and CRP. Human studies on aspirin for depression have been small and inconclusive, but research does show that aspirin, along with an SSRI, shortened the time it took for the antidepressant to work.[ref]
- NSAIDs: Naproxen and ibuprofen reduced depression scores in people who also had osteoarthritis.[ref] Studies on NSAIDs in people without an inflammatory condition show no results for depression.
Who might this work best for? Individuals with genetic variants in TNF, IL6, IL6R
Antibiotics:
Antibiotics can be effective for reducing inflammation in specific circumstances. Talk with your doctor.
- Minocycline: Minocycline is a broad-spectrum antibiotic that easily crosses the blood-brain barrier. A recent clinical trial shows that it is effective as an adjunctive treatment along with immunomodulatory medications for treatment-resistant major depressive disorder.[ref] Minocycline has also been shown in animal studies to work on anxiety.[ref] A 2021 study showed that adding minocycline to antidepressant treatment in people with elevated C-reactive protein significantly reduced depression levels. The results showed that study participants with high IL-6 levels were much more likely to respond to minocycline.[ref] Minocycline has been shown to reduce IL6[ref]
Who might this work best for? Individuals with genetic variants in IL6, IL6R (or with a chronic infection, such as Lyme)
IL-6 inhibitors:
- Ketamine: Ketamine decreases IL-6 in depression in the prefrontal cortex and hippocampus.[ref]
- Andrographis: Andrographolide, a component of the traditional Chinese medicine plant Andrographis, inhibits IL-6.[ref] Andrographis is readily available in health stores and online as a supplement.
Who might this work best for? Individuals with genetic variants in IL6, IL6R
Targeting the kynurenine pathway:
- Niacin, magnesium: In addition to responding to inflammation or stress, the body also shunts more tryptophan towards the kynurenine pathway when niacin is low.[ref] Additionally, magnesium is needed to convert quinolinic acid into niacin.
- Omeprazole (PPI) avoidance: Omeprazole inhibits the conversion of tryptophan to serotonin and could thus cause increased kynurenine in addition to lower levels of serotonin. This seems to be dose-dependent, though. At higher doses, omeprazole also inhibits MAO-A, the enzyme that breaks down serotonin, and thus consequently increases brain serotonin.[ref]
Related article: MAO-A and MAO-B polymorphisms
Who might this work best for? Individuals with genetic variants in IDO, KMO
Exercise:
- Long-term exercise: Physical activity has been shown in a number of solid research studies to work as an alternative to antidepressants.[ref] Specifically, exercise has been shown to decrease depressive symptoms over a 12-week period in people who have high baseline TNF-alpha or IL-1B levels. The people in the clinical trial were previously unresponsive to SSRIs, and the exercise was aerobic exercise, such as a treadmill or cycling. Also, an important note in this study is that depression scores continued to get better over the course of the 3-month long intervention. So this isn’t an instant cure-all, but rather something to stick with long-term.[ref]
Who might this work best for? Individuals with genetic variants in TNF or IL1B
Dietary changes to reduce inflammation:
Healthy dietary changes may help a lot in combating depression, especially if you currently eat a lot of fast food or junk food.
- Decreasing your omega-6 to omega-3 ratio by cutting out junk food should help to reduce inflammation.
- Avoiding foods that you are allergic to or sensitive to can also help.
- Ditch the processed food with preservatives and food dyes.
Related article: Which diet is right for your genes?
Who might this work best for? Everyone. This is a basic change that should help everyone.
Keto diet: A low-carb, ketogenic diet has now been shown in several clinical trials to have efficacy for depression and mood disorders. A 2023 animal study specifically showed the mechanism: reduced neuroinflammation. The ketogenic diet decreased IL-1β, TNF-α, and COX-2 levels.[ref]
Natural anti-inflammatory supplements:
Again, check with your doctor or pharmacist for any interactions with medications before starting new supplements.
- Quercetin: Quercetin is another natural anti-inflammatory supplement that reduces TNF-alpha and other inflammatory cytokines. Animal studies show that quercetin is effective in reducing depression and anxiety behavior.[ref][ref]
Related article: Quercetin, genetics, absorption and more
Your genotype for rs4680 is —, which means you have two copies of the slow COMT variant and will likely have lower COMT enzyme function. Read the full article on COMT here.
- Curcumin: Curcumin, a compound found in the spice turmeric, acts as an anti-inflammatory and decreases TNF-alpha and IL1B.[ref] Several good clinical trials have investigated the effect of curcumin on depression and anxiety. Overall, supplementation of curcumin is well tolerated and decreases both depression and anxiety scores.[ref]
Related article: Curcumin Supplements: Decreasing Inflammation
Who might this work best for? Individuals with genetic variants in TNF or IL1B
- Acetyl l-carnitine: Acetyl l-carnitine has been shown in studies to combat inflammation, including reducing TNF-alpha and IL-1B.[ref] Additionally, research shows that people diagnosed with depression have lower acetyl l-carnitine levels.[ref]
Who might this work best for? Individuals with genetic variants in TNF or IL1B
Saffron: Multiple clinical trials show that saffron may help with depression for some people.
- In one study, 128 adults with low mood but not diagnosed depression were given a saffron extract (affron) at 28mg/day, 22mg/day, or a placebo for 4 weeks. The results showed that mood improved and anxiety decreased at the 28mg/day dose.[ref]
- For patients with major depressive disorder, a study showed that the combination of curcumin (250mg) plus saffron (15mg/day) was effective in reducing depression scores, especially in people with atypical depression.[ref]
- In a comparison trial, saffron (30mg/day) was as effective as fluoxetine (Prozac) for mild to moderate depression after six weeks.[ref]
- Another study in adults with anxiety and depression looked at a 50mg saffron capsule taken twice a day. This study found that saffron was significantly more effective than placebo in reducing depression scores after 12 weeks. [ref]
- In older adults with major depressive disorder, saffron (60mg/day) was compared to sertraline (Zoloft) for depression. They were found to be equally effective at reducing depression.[ref]
- A study involving postmenopausal women showed that saffron tea significantly improved “happiness”.[ref]
- A randomized placebo-controlled clinical trial of 200 patients with depressive symptoms found that 3 months of 28mg of saffron supplementation daily was beneficial in reducing depression without adverse side effects. [ref]
Who might this work best for? Individuals with genetic variants in IL-6.[ref]
Creatine: Clinical trials show creatine may help with depression caused by a lack of brain energy or oxidative stress.[ref]
Related article: Creatine
Vitamin C: Animal studies show that supplementing their normal diet with additional ascorbic acid (vitamin C) decreases anxiety and decreases brain levels of TNF-alpha.[ref] Foods that are high in vitamin C include orange juice, red peppers, strawberries, kiwifruit, and guava.
Related article: Vitamin C genes
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
Lithium Orotate and Vitamin B12: Benefits for Mood and Cognitive Support
References:
Jha, Manish K., et al. “Should Inflammation Be a Specifier for Major Depression in the DSM-6?” JAMA Psychiatry, vol. 82, no. 6, June 2025, pp. 549–50. PubMed, https://doi.org/10.1001/jamapsychiatry.2025.0206.