Key takeaways:
- Thiamine (vitamin B1) is a water-soluble vitamin that serves as a cofactor in the metabolism of carbohydrates, branch-chain amino acids, and fatty acids.
- A lack of thiamine can cause decreased mitochondrial energy production, causing neurological issues such as fatigue, brain fog, irritability, slowed bowel function (constipation, IBS), and even long Covid.
- Genetic variants can affect how thiamine is absorbed and transported, impacting how much thiamine you need each day.
- Understanding your genetic variants may help you pinpoint how much thiamine you need and in what form.
Members will see their genotype report below, plus additional solutions in the Lifehacks section. Consider joining today.
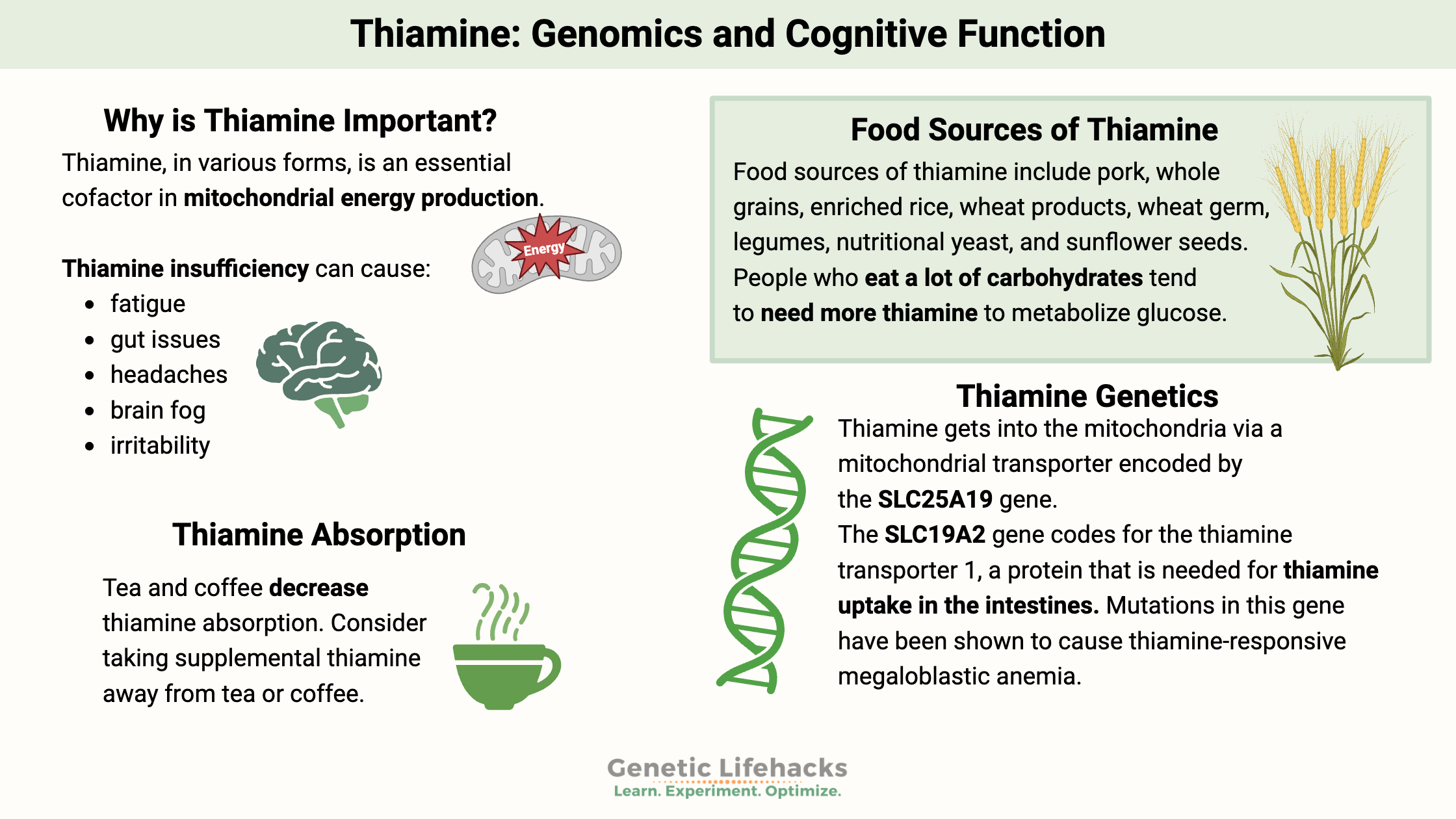
Why is thiamine important?
Thiamine (vitamin B1) is vital for energy production and neurological health. Genetic differences can influence how much thiamine you need, making it important to understand both dietary sources and your personal risk factors.
Fundamental for cellular energy:
Thiamine, in various forms, is an essential cofactor in mitochondrial energy production. Thus, a deficiency in this vitamin can cause various symptoms. New research also points to a role for thiamine in neurodegenerative diseases and cataracts.
Thiamine (also spelled thiamin) is vitamin B1. It is essential, meaning that the body doesn’t make it and you have to get it from food.
Why is it essential? ATP production, used in every cell for energy, requires thiamine. An insufficient amount of thiamine can cause problems with cellular energy.
Thiamine deficiency:
Severe deficiency of thiamine leads to a disease called beriberi. It is uncommon in our modern era to have beriberi except in cases of chronic alcohol abuse or a rare genetic mutation. However, understanding the symptoms of beriberi explains some of the conditions associated with milder deficiency.
Both severe thiamine deficiency and mild insufficiency can cause a range of neurological and physical symptoms, including the following:[ref][ref][ref]
| Condition | Symptoms |
|---|---|
| Severe Deficiency (Beriberi) | Emotional problems, weakness, confusion, muscle pain, irregular heartbeat, edema, neuropathy, cardiomyopathy, lactic acidosis |
| Thiamine Insufficiency | Fatigue, drowsiness, gut issues, headaches, brain fog, irritability |
Notice that all of these symptoms are related to a lack of cellular energy in the brain and nervous system.
Thiamine insufficiency misdiagnosed in aging:
Mild thiamine deficiency is fairly common in the elderly and can lead to misdiagnoses for dementia-related illnesses and increase the risk of these diseases.[ref][ref]
- In animal studies, supplementing with thiamine “rescued cognitive deficits and reduced Aβ burden in amyloid precursor protein”.[ref]
- Research on a bioavailable form of thiamine called benfotiamine shows promise for Alzheimer’s disease.[ref][ref]
Food sources and dietary interactions:
Food sources of thiamine include pork, whole grains, enriched rice, wheat products, wheat germ, legumes, nutritional yeast, and sunflower seeds. The daily recommended intake (RDA) for thiamine is 1.2 – 2 mg per day for adults.
Food processing often depletes nutrients, including thiamine. Some foods, especially rice and wheat products, have thiamine added to them (check the labels).
One study of obese people who were getting plenty of calories found that 29% were thiamine-deficient. People who eat a lot of carbohydrates tend to need more thiamine to metabolize glucose. Similarly, people who exercise a lot also need more thiamine.[ref]
How can you know if you are getting enough thiamine in your diet?
Most people eating a varied diet with whole foods are likely to reach the RDA. For someone on a grain-free diet who doesn’t eat a lot of pork, it may be worth tracking your intake for a week or so to ensure that you get enough thiamine. Cronometer.com is a free online app to keep track of your nutrient intake.
Thiamine absorption and genetics:
Thiamine is a water-soluble vitamin that isn’t stored in large amounts in the body, although the liver can store a little bit. The half-life of thiamine is short, and thus, we need to regularly consume it.[ref]
Thiamine is absorbed in the upper part of the small intestine. Most dietary thiamine is in a phosphorylated form, which is converted in the intestines to free thiamine before being absorbed. At low doses (below 5 mg), thiamine is absorbed using the thiamine transporters SLC19A2 and SLC19A3.[ref] Thiamine produced by the gut microbiome is absorbed in the colon through the SLC44A4 transporter.[ref] SLC35F3 is an intracellular thiamine transporter, and once inside the cells, SLC25A19 moves thiamine into the mitochondria.
Genetic variants in all these genes can influence your need for thiamine (See your genotypes in the Genotype Report section.)
| Gene | Role in Thiamine Metabolism | Effect of Variants |
|---|---|---|
| SLC19A2 | Intestinal thiamine transporter | Reduced absorption |
| SLC19A3 | Intestinal thiamine transporter | Reduced absorption |
| SLC44A4 | Colonic thiamine transporter | Affects microbiome-derived thiamine |
| TPK1 | Converts thiamine in enterocytes | Impaired conversion |
| SLC35F3 | Intracellular transport of thiamine | Metabolic syndrome, IBS, constipation |
| SLC22A1 (OCT1) | Liver thiamine transporter | Alters hepatic uptake |
| SLC25A19 | Mitochondrial thiamine transporter | Impaired mitochondrial import |
Changes and selection of variants in response to agriculture:
Interestingly, researchers think that some of the genetic variants in the thiamine transporter genes have arisen in different population ancestry groups over history in response to changes in agriculture. For example, there are variants more common in East Asian populations that date back to when there was a shift toward consuming more polished rice.[ref]
Gut microbiome influences thiamine:
Thiamine is intricately related to your gut microbiome. Certain bacteria in the gut can degrade thiamine from food or supplements, decreasing the amount available to be absorbed. E. coli overgrowth is one bacterium that is linked to reduced thiamine uptake. Certain gut microbes can also produce thiamine.
Studies show the following interactions:[ref][ref]
| Bacteria Type | Effect on Thiamine |
|---|---|
| E. coli (overgrowth) | Decreases absorption |
| Bifidobacterium, Lactobacillus, Enterococcus | Increases absorption |
| Various (producers) | Synthesize thiamine |
Keep in mind when evaluating your dietary intake and your genetic variants that the third player here is your gut microbiome. If you have an overgrowth of certain bacteria, it may decrease your thiamine transport.
Thiamine insufficiency symptoms supported by research:
While full-blown beriberi is rare in the modern world due to thiamine-fortified foods, symptoms from thiamine insufficiency are possible due to restrictive diets, impaired absorption, anorexia, or bariatric surgery.
People who drink a lot of alcohol can develop a form of thiamine deficiency, known as Wernicke’s encephalopathy, due to liver problems and reduced uptake of thiamine in the intestines. Thiamine supplementation is often recommended for anyone with alcohol dependency issues.[ref]
What does research show on thiamine insufficiency?
- Thiamine insufficiency is linked to an increased risk of heart failure in older people.[ref]
- Clinical trial data show that oral benfotiamine (a type of thiamine) helps with peripheral neuropathy in alcoholics.[ref]
- A phase II clinical trial of benfotiamine shows that it is safe and possibly effective for improving cognitive outcomes in Alzheimer’s patients.[ref]
- Thiamine may help reduce the production of advanced glycation end products.[ref]
- Benfotiamine has also been shown to reduce the inflammatory response in microglia. It could potentially be important in cognitive function and neurodegenerative diseases.[ref]
- Animal studies show that thiamine could improve cognition via modulating GSK-3B expression.[ref]
- In hospitalized patients with sepsis, a combination of IV thiamine, vitamin C, and hydrocortisone vastly improved the mortality rate compared to a control group.[ref]
- An animal study of induced myocardial infarction (heart attack) showed that benfotiamine administered before and after the heart attack had improved cardiac markers.[ref]
- Thiamine deficiency (mild or severe) is significantly associated with cognitive impairment and mood changes in cancer patients.[ref]
- A 2026 genome-wide association study paired with Mendelian randomization found that thiamine transport and activation are key in intestinal motility and IBS-C.[ref] Other studies show that increasing thiamine intake reduces the risk of constipation.[ref]
Thiamine in Energy Metabolism:
Cellular uptake:
Thiamine is absorbed in the region of the small intestine called the jejunum. The food you eat breaks down throughout the digestive tract, and then thiamine absorption occurs in the intestinal wall. At higher levels, thiamine passes into the intestinal wall via passive diffusion; at lower levels, thiamine uses a transporter (encoded by the SLC19A2 gene) to enter cells.[ref]
Within the cells lining the intestines, thiamine is transformed by an enzyme encoded by the TPK1 gene. Then it is moved into the bloodstream to be transported through the body. If you have gastrointestinal issues that impact nutrient absorption, this could also decrease your ability to absorb thiamine.

Transport into cells throughout the body: SLC35F3
SLC35F3 is involved in transporting thiamine into cells, and variants in the gene affect carbohydrate metabolism and metabolic syndrome. Variants are also associated with increased blood pressure and visceral fat levels.
Thiamine in gut motility:
A 2026 genetics study identified thiamine and SLC35F3 as key in gut motility and stool frequency. The study also found that XPR1 may play a role in slowed motility. XPR1 exports inorganic phosphate, which is essential for activating thiamine. For anyone dealing with IBS and constipation, thiamine transport may be important.[ref]
Thiamine in the liver:
OCT1 (organic cation transporter 1) is important in how drugs, including metformin, work in the body. Genetic variants in the gene that encodes OCT1 (SLC22A1 gene) are linked to increased or decreased function and also impact metformin efficacy. Recent research shows that OCT1 is also a major thiamine transporter in the liver.[ref]
Recent animal studies show that high doses of thiamine improve fatty liver disease.[ref]
Energy production:
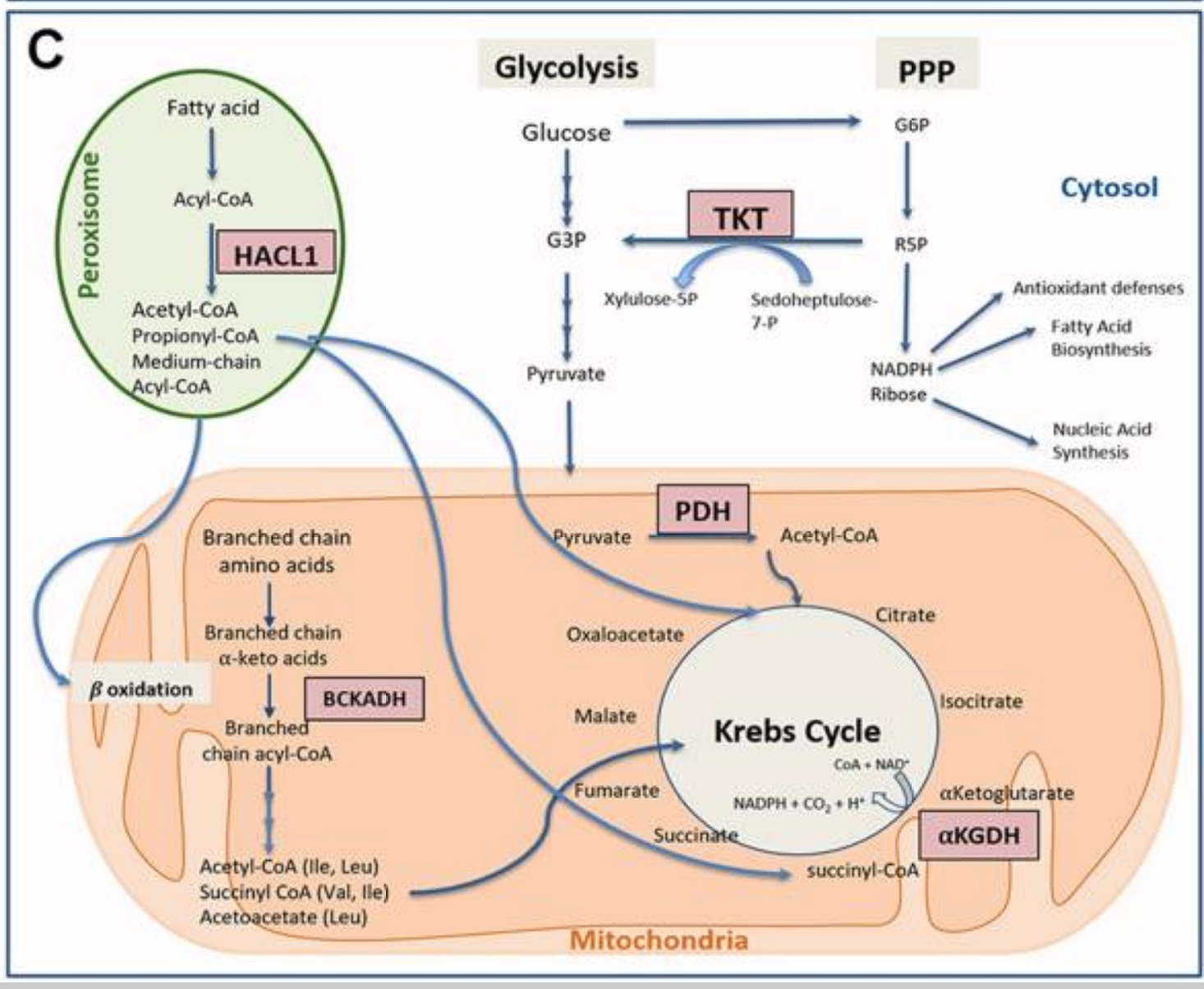
Thiamine is a cofactor essential for mitochondrial energy production. Within the Krebs cycle, thiamine is a cofactor for pyruvate dehydrogenase, α-ketoglutarate dehydrogenase, and branched-chain α-ketoacid dehydrogenase complexes.[ref]
Animal studies show that a thiamine derivative called thiamine tetrahydrofurfuryl disulfide increases physical activity by interacting with dopamine in the prefrontal cortex. Essentially, this form of thiamine increases wakefulness and arousal.[ref]
Keto diets:
Researchers believe that thiamine deficiency decreases the cell’s ability to switch easily between burning fat for fuel vs. using glucose.[ref] This is important for people doing intermittent fasting or a ketogenic diet. Fat doesn’t contain thiamine, so a high-fat diet will naturally be low in thiamine. While thiamine isn’t as essential for someone in ketosis, if you are switching between a ketogenic diet and one with carbohydrates, you may need more thiamine (or prioritize thiamine-rich carbohydrates).
Mitochondrial transporter:
For thiamine to enter the mitochondria, a transporter is needed. Thiamine gets into the mitochondria via a mitochondrial transporter encoded by the SLC25A19 gene.
In addition to its role as a cofactor for Krebs cycle enzymes, thiamine is also a cofactor for a key enzyme in the pentose phosphate pathway. It is important in generating NADPH and ribose 5-phosphate and inhibiting NADPH oxidase.[ref]
Related article: Fatigue: Genetics and Causes
High cholesterol:
A Mendelian randomization study shows that there is a link between familial hypercholesterolemia (very high cholesterol due to genetics) and thiamine deficiency.[ref] If you have hypercholesterolemia, you may want to consider checking your thiamine intake.
Long Covid and thiamine:
Anecdotally, thiamine supplementation in the form of benfotiamine helps some people significantly with long covid symptoms.
Let’s take a look at the studies on this topic:
- A randomized, controlled trial of long Covid patients showed that thiamine significantly shortened the duration of symptoms, compared to a control group.[ref]
- Several publications lay out the rationale for why thiamine supplementation may help patients suffering from long Covid.[ref]
- In patients with severe COVID-19, Wernicke encephalopathy (due to a lack of thiamine) is frequent.[ref] Thiamine supplementation in the hospital significantly decreases mortality in severe COVID-19 patients.[ref]
In addition to thiamine’s role in mitochondrial energy production, it also modulates the immune response:
- Benfotiamine has been shown to reduce endotoxin-induced cytokine release. It shows that it has a modulating effect on the immune response. While we want a robust immune response against a pathogen, an overactive immune response causes damage.[ref]
Thiamine Genotype Report:
Access this content:
An active subscription is required to access this content.
Lifehacks:
Thiamine insufficiency is easy to overlook as a cause of symptoms that mimic other conditions. Fatigue, drowsiness, brain fog, infrequent bowel movements, IBS-C, irritability, memory problems, confusion, loss of appetite, heart problems, and balance issues: all can be caused by thiamine deficiency — or by other things.
How much thiamine do you need?
At a glance:
- RDA: Men: 1.2 mg/day | Women: 1.1 mg/day
- Upper Limit: Not established (insufficient evidence to set a tolerable upper limit, but you likely won’t absorb more than 5 mg/day)[ref]
Talk to your doctor or a registered dietician if you have any concerns about your nutritional needs or about whether a supplement is right for you.
Diet:
Foods that are high in thiamine include[ref][ref][ref]
| Food with thiamine (vitamin B1) | Milligrams (mg) per serving |
Percent DV* |
|---|---|---|
| Breakfast cereals, fortified with 100% of the DV for thiamin, 1 serving | 1.2 | 100 |
| Egg noodles, enriched, cooked, 1 cup | 0.5 | 42 |
| Pork chop, bone in, broiled, 3 ounces | 0.4 | 33 |
| Trout, cooked, dry heat, 3 ounces | 0.4 | 33 |
| Black beans, boiled, ½ cup | 0.4 | 33 |
| English muffin, plain, enriched, 1 | 0.3 | 25 |
| Mussels, blue, cooked, moist heat, 3 ounces | 0.3 | 25 |
| Tuna, bluefin, cooked, dry heat, 3 ounces | 0.2 | 17 |
| Macaroni, whole wheat, cooked, 1 cup | 0.2 | 17 |
| Acorn squash, cubed, baked, ½ cup | 0.2 | 17 |
| Rice, brown, long grain, not enriched, cooked, ½ cup | 0.2 | 17 |
The recommended daily thiamine intake is 1.2 mg/day of thiamin for men and 1.1 mg/day for women.
Keep in mind that it is a water-soluble vitamin that isn’t stored for long in the body. So you need a constant, low-level intake.
Factors affecting thiamine absorption:
| Factor | Effect on Thiamine Status |
|---|---|
| Tea, coffee | Decrease absorption |
| Mold/mycotoxins | Anti-thiamine effect |
| Yogurt/probiotics | Increase plasma thiamine |
| Resistant starch | Increases microbially produced thiamine |
Increasing thiamine absorption:
Some foods contain anti-thiamine factors. For example, tea and coffee decrease thiamin absorption, and certain mycotoxins (mold toxins) can act as anti-thiamine factors.
Maximizing Gut Microbiome Production:
Thiamine from foods is absorbed primarily in the small intestine. However, we also get some thiamine from our gut bacteria. In the colon, certain bacteria in the gut microbiome can synthesize thiamine, but other types of bacteria can degrade thiamine. Thus, the balance of producers to degraders in the gut microbiome can alter your thiamine levels. In addition, the pH of the gut, which can be affected by lactic acid producing bacteria, also plays a role in how much thiamine is absorbed in the gut.[ref]
Yogurt and probiotics:
A study on yogurt consumption showed that both regular yogurt and yogurt with extra probiotics added increase plasma thiamine levels in healthy adults.[ref]
Resistant starch:
Cooking and then cooling rice and potatoes increases the resistant starch in them. A clinical trial showed that resistant starch increases microbially produced thiamine.[ref]
Tip: Try cooking your rice or potatoes a day ahead and cooling them in the refrigerator overnight. You can then reheat them the next day, and they will still have the resistant starch in them.
Safety and testing:
Testing:
Blood tests for thiamine levels are readily available. Talk with your doctor about testing, or order it on your own. Shop around if you’re ordering lab tests on your own – prices vary quite a bit on online lab test websites.
Safety: Caution with cancer
Thiamine is a water-soluble vitamin with no known toxicity.
If you have a rapidly growing tumor, talk with your doctor before supplementing with thiamine. Rapidly dividing cells need a lot of thiamine, which may be contraindicated with your treatment.
Thiamine: three types of supplements
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
References:
Díaz-Muñoz C, Bozzarelli I, Lopera-Maya EA, et al
Genetic dissection of stool frequency implicates vitamin B1 metabolism and other actionable pathways in the modulation of gut motility
Gut Published Online First: 20 January 2026. doi: 10.1136/gutjnl-2025-337059
Ahmed, Lamiaa A., et al. “Beneficial Effects of Benfotiamine, a NADPH Oxidase Inhibitor, in Isoproterenol-Induced Myocardial Infarction in Rats.” PloS One, vol. 15, no. 5, 2020, p. e0232413. PubMed, https://doi.org/10.1371/journal.pone.0232413.
———. “Beneficial Effects of Benfotiamine, a NADPH Oxidase Inhibitor, in Isoproterenol-Induced Myocardial Infarction in Rats.” PLoS ONE, vol. 15, no. 5, May 2020, p. e0232413. PubMed Central, https://doi.org/10.1371/journal.pone.0232413.
Al Sulaiman, Khalid, et al. “Evaluation of Thiamine as Adjunctive Therapy in COVID-19 Critically Ill Patients: A Two-Center Propensity Score Matched Study.” Critical Care (London, England), vol. 25, no. 1, June 2021, p. 223. PubMed, https://doi.org/10.1186/s13054-021-03648-9.
Ang, Cynthia D., et al. “Vitamin B for Treating Peripheral Neuropathy.” The Cochrane Database of Systematic Reviews, vol. 2008, no. 3, July 2008, p. CD004573. PubMed, https://doi.org/10.1002/14651858.CD004573.pub3.
Ao, Misora, et al. “Possible Involvement of Thiamine Insufficiency in Heart Failure in the Institutionalized Elderly.” Journal of Clinical Biochemistry and Nutrition, vol. 64, no. 3, May 2019, pp. 239–42. PubMed, https://doi.org/10.3164/jcbn.18-85.
Bhawal, Ruchika, et al. “Serum Metabolomic and Lipidomic Profiling Reveals Novel Biomarkers of Efficacy for Benfotiamine in Alzheimer’s Disease.” International Journal of Molecular Sciences, vol. 22, no. 24, Dec. 2021, p. 13188. PubMed Central, https://doi.org/10.3390/ijms222413188.
Bozic, Iva, et al. “Benfotiamine Attenuates Inflammatory Response in LPS Stimulated BV-2 Microglia.” PloS One, vol. 10, no. 2, 2015, p. e0118372. PubMed, https://doi.org/10.1371/journal.pone.0118372.
Branco de Oliveira, Marcus Vinicius, et al. “Encephalopathy Responsive to Thiamine in Severe COVID-19 Patients.” Brain, Behavior, & Immunity – Health, vol. 14, July 2021, p. 100252. PubMed, https://doi.org/10.1016/j.bbih.2021.100252.
Dhir, Shibani, et al. “Neurological, Psychiatric, and Biochemical Aspects of Thiamine Deficiency in Children and Adults.” Frontiers in Psychiatry, vol. 10, Apr. 2019, p. 207. PubMed Central, https://doi.org/10.3389/fpsyt.2019.00207.
Fabian, Elisabeth, et al. “Influence of Probiotic and Conventional Yoghurt on the Status of Vitamins B1, B2 and B6 in Young Healthy Women.” Annals of Nutrition & Metabolism, vol. 52, no. 1, 2008, pp. 29–36. PubMed, https://doi.org/10.1159/000114408.
Fattal-Valevski, Aviva. “Thiamine (Vitamin B1 ).” Journal of Evidence-Based Complementary & Alternative Medicine, vol. 16, no. 1, Jan. 2011, pp. 12–20. DOI.org (Crossref), https://doi.org/10.1177/1533210110392941.
Gibson, Gary E., et al. “Benfotiamine and Cognitive Decline in Alzheimer’s Disease: Results of a Randomized Placebo-Controlled Phase IIa Clinical Trial.” Journal of Alzheimer’s Disease: JAD, vol. 78, no. 3, 2020, pp. 989–1010. PubMed, https://doi.org/10.3233/JAD-200896.
Glasø, M., et al. “Reduced Concentrations of Several Vitamins in Normal Weight Patients with Late-Onset Dementia of the Alzheimer Type without Vascular Disease.” The Journal of Nutrition, Health & Aging, vol. 8, no. 5, 2004, pp. 407–13. PubMed.
Iimura, Yohei, et al. “Thiamine Deficiency and Neurological Symptoms in Patients with Hematological Cancer Receiving Chemotherapy: A Retrospective Analysis.” Journal of Neurosciences in Rural Practice, vol. 12, no. 4, Oct. 2021, pp. 726–32. PubMed, https://doi.org/10.1055/s-0041-1735825.
Jankowska-Kulawy, Agnieszka, et al. “Acetyl-CoA Deficit in Brain Mitochondria in Experimental Thiamine Deficiency Encephalopathy.” Neurochemistry International, vol. 57, no. 7, Dec. 2010, pp. 851–56. PubMed, https://doi.org/10.1016/j.neuint.2010.09.003.
Marik, Paul E., et al. “Hydrocortisone, Vitamin C, and Thiamine for the Treatment of Severe Sepsis and Septic Shock: A Retrospective Before-After Study.” Chest, vol. 151, no. 6, June 2017, pp. 1229–38. PubMed, https://doi.org/10.1016/j.chest.2016.11.036.
Markova, Nataliia, et al. “Thiamine and Benfotiamine Improve Cognition and Ameliorate GSK-3β-Associated Stress-Induced Behaviours in Mice.” Progress in Neuro-Psychopharmacology & Biological Psychiatry, vol. 75, Apr. 2017, pp. 148–56. PubMed, https://doi.org/10.1016/j.pnpbp.2016.11.001.
Office of Dietary Supplements – Thiamin. https://ods.od.nih.gov/factsheets/Thiamin-HealthProfessional/. Accessed 9 Dec. 2025.
Oliver, Andrew, et al. “Diet, Microbiome, and Inflammation Predictors of Fecal and Plasma Short-Chain Fatty Acids in Humans.” The Journal of Nutrition, vol. 154, no. 11, Nov. 2024, pp. 3298–311. PubMed, https://doi.org/10.1016/j.tjnut.2024.08.012.
Raj, V., et al. “Therapeutic Potential of Benfotiamine and Its Molecular Targets.” European Review for Medical and Pharmacological Sciences, vol. 22, no. 10, May 2018, pp. 3261–73. PubMed, https://doi.org/10.26355/eurrev_201805_15089.
Tapias, Victor, et al. “Benfotiamine Treatment Activates the Nrf2/ARE Pathway and Is Neuroprotective in a Transgenic Mouse Model of Tauopathy.” Human Molecular Genetics, vol. 27, no. 16, Aug. 2018, pp. 2874–92. PubMed, https://doi.org/10.1093/hmg/ddy201.
The Most Accurate Nutrition Tracking App | Cronometer. https://cronometer.com/index.html. Accessed 9 Dec. 2025.
Thiamin | Linus Pauling Institute | Oregon State University. 12 Oct. 2021, https://lpi.oregonstate.edu/mic/vitamins/thiamin.
Thiamin – Vitamin B1 • The Nutrition Source. 8 July 2019, https://nutritionsource.hsph.harvard.edu/vitamin-b1/.
Ungur, Alexander Lavinius, et al. “Perioperative Management of Alcohol Withdrawal Syndrome.” Visceral Medicine, vol. 36, no. 3, June 2020, pp. 160–66. PubMed, https://doi.org/10.1159/000507595.
Vatsalya, Vatsalya, et al. “Therapeutic Prospects for Th-17 Cell Immune Storm Syndrome and Neurological Symptoms in COVID-19: Thiamine Efficacy and Safety, In-Vitro Evidence and Pharmacokinetic Profile.” medRxiv: The Preprint Server for Health Sciences, Aug. 2020, p. 2020.08.23.20177501. PubMed, https://doi.org/10.1101/2020.08.23.20177501.
Viel, Christian, et al. “Effects of Magnesium Orotate, Benfotiamine and a Combination of Vitamins on Mitochondrial and Cholinergic Function in the TgF344-AD Rat Model of Alzheimer’s Disease.” Pharmaceuticals (Basel, Switzerland), vol. 14, no. 12, Nov. 2021, p. 1218. PubMed, https://doi.org/10.3390/ph14121218.
Wan, Zhijie, et al. “Intermediate Role of Gut Microbiota in Vitamin B Nutrition and Its Influences on Human Health.” Frontiers in Nutrition, vol. 9, Dec. 2022. Frontiers, https://doi.org/10.3389/fnut.2022.1031502.
———. “Intermediate Role of Gut Microbiota in Vitamin B Nutrition and Its Influences on Human Health.” Frontiers in Nutrition, vol. 9, Dec. 2022. Frontiers, https://doi.org/10.3389/fnut.2022.1031502.
Wang, Changpeng, et al. “High Thiamine Diphosphate Level as a Protective Factor for Alzheimer’s Disease.” Neurological Research, vol. 40, no. 8, Aug. 2018, pp. 658–65. PubMed, https://doi.org/10.1080/01616412.2018.1460704.
Yadav, Umesh C. S., et al. “Prevention of Endotoxin-Induced Uveitis in Rats by Benfotiamine, a Lipophilic Analogue of Vitamin B1.” Investigative Ophthalmology & Visual Science, vol. 50, no. 5, May 2009, pp. 2276–82. PubMed Central, https://doi.org/10.1167/iovs.08-2816.
Zhang, Cheng, et al. “Causal Relationships of Familial Hypercholesterolemia with the Risk of Multiple Vitamin Deficiencies: A Mendelian Randomization Study.” Frontiers in Endocrinology, vol. 15, Oct. 2024, p. 1401260. PubMed Central, https://doi.org/10.3389/fendo.2024.1401260.