Key takeaways:
- The APOE alleles are important in understanding your Alzheimer’s risk, and the data is found in your 23andMe raw data file (if you want to know).
- APOE is involved in carrying cholesterol and other fats in your bloodstream, and a common variant of the gene is strongly linked to a higher risk of Alzheimer’s.[ref]
- Alzheimer’s risk is influenced by both genetics and environmental factors. Genes are only one part of the equation for Alzheimer’s.
- Knowing your risk can help you prioritize lifestyle changes for the prevention of Alzheimer’s disease. Clinical trials and high-quality studies show that there are many options to help prevent Alzheimer’s.
APOE Gene Variants:
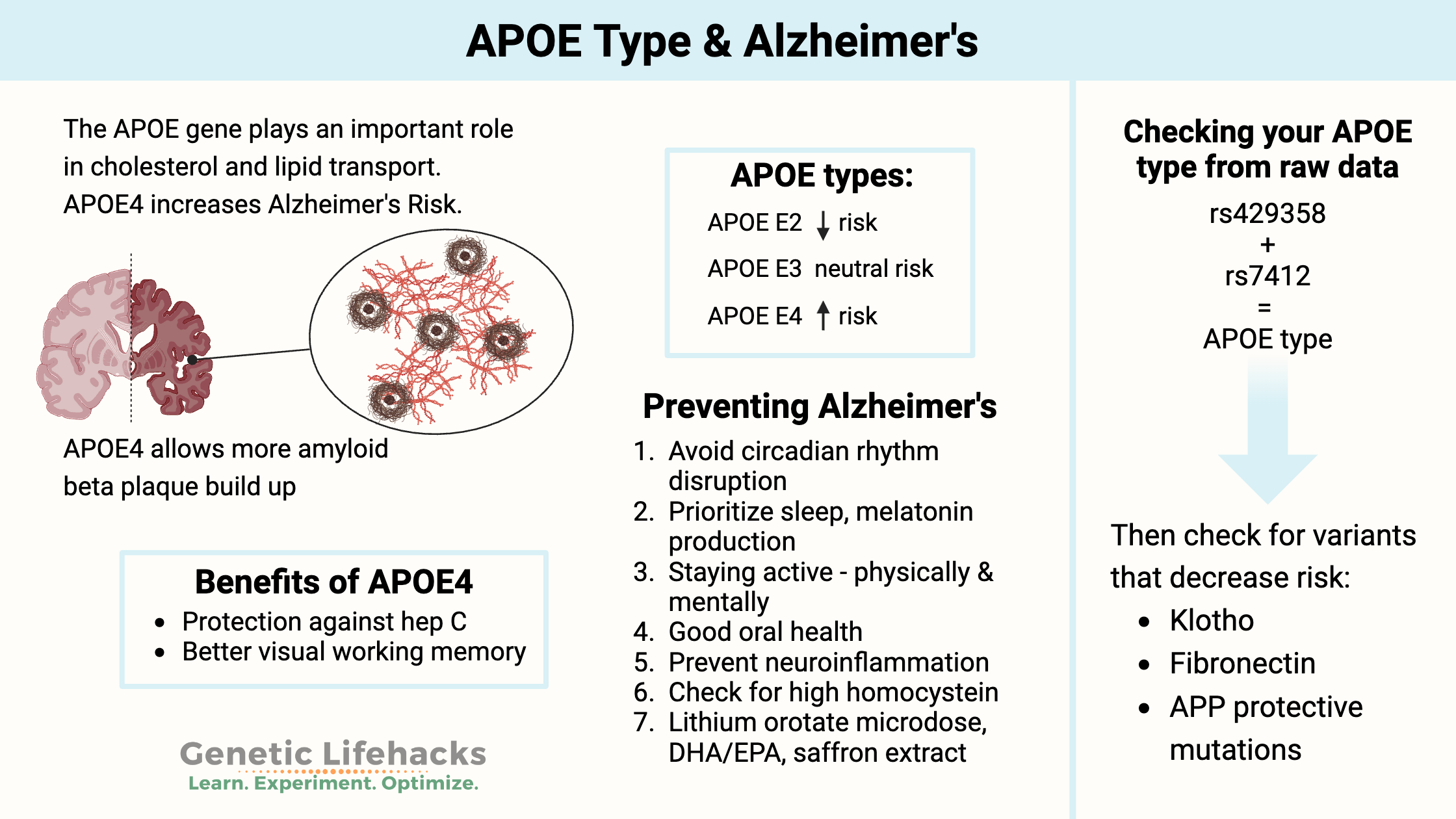
One well-known gene that influences Alzheimer’s risk is the apolipoprotein E (APOE) gene. The APOE gene encodes the APOE protein, and your APOE type is defined as a combination of three different alleles (E2, E3, or E4). You will have one APOE allele from each parent, which means your APOE type will be stated with two alleles, such as E3/E3.
The NIH website explains:
| APOE Allele | Effect on Alzheimer’s Risk |
|---|---|
| ε2 | Decreased risk (protective) |
| ε3 | Neutral (normal risk) |
| ε4 | Increased risk (>40% of Alzheimer’s cases carry this allele) |
A quick note before we start:
Learning that you carry a genetic risk factor for Alzheimer’s can be an emotional experience, and not everyone wants that information. There’s no right or wrong choice here. If you’d just want to read the research and prevention strategies without looking up your specific genotype, scroll down to the Lifehacks section for the research-backed prevention methods.
What does APOE do?
APOE (apolipoprotein E) is a protein that transports cholesterol and other fats in the body, particularly in the brain. It is primarily involved in the transport and metabolism of lipids in the blood, where it helps to carry cholesterol and other lipids (fats) to and from cells. In the brain, it helps to transport lipids across the blood-brain barrier.[ref]
In the brain, APOE helps to regulate the clearance of beta-amyloid protein, which is a key component of the plaques that form in the brains of people with Alzheimer’s disease. APOE is also involved in other processes in the body, such as inflammation, immune function, and repair of tissues after injury.
The different APOE genotypes (APOE E2, E3, and E4) result in different-sized APOE molecules. Additionally, the different APOE types are associated with the level of plasma APOE.
- APOE E2 –> higher plasma APOE levels
- APOE E3 –> typical APOE levels
- APOE E4 –> lower plasma APOE levels
As one study explains: “Low plasma levels of apoE are associated with increased risk of future Alzheimer’s disease and all dementia in the general population.”[ref]
Genotype report: Finding your APOE type
Before we go further into what the APOE research shows and options for preventing Alzheimer’s, let’s first look at how to find your APOE type.
AncestryDNA NOTE:
If you downloaded your AncestryDNA raw data in 2015 or 2016 (and haven’t re-downloaded it), it will contain the APOE information, but it may be in error. Since 2017, AncestryDNA has not contained one of the APOE rsIDs needed for determining APOE type.[ref]
Other raw data files that work with Genetic Lifehacks membership, such as 23andMe, selfdecode, sequencing.com, or MyHeritage data files, should show your APOE SNPs.
To determine your APOE type from your 23andMe data or another source, you will need to look at your genotypes for the combination of two rs IDs: rs429358 and rs7412.
Members will see their genotypes reported below and APOE type flagged if it is in their connected raw data file. Consider joining today.Check your genetic data for rs429358 (23andMe v4, v5):
- T/T: typical; look at this in combination with rs7412 (chart below)
- C/T: look at this in combination with rs7412 (chart below)
- C/C: look at this in combination with rs7412 (chart below)
Members: Your genotype for rs429358 is —.
Check your genetic data for rs7412 (23andMe v4, v5):
- C/C: typical; look at this in combination with rs429358 (chart below)
- C/T: look at this in combination with rs429358 (chart below)
- T/T: look at this in combination with rs429358 (chart below)
Members: Your genotype for rs7412 is —.
| rs429358 | rs7412 | APOE Allele | Alzheimer’s Risk |
|---|---|---|---|
| Your genotype: — | Your genotype: — | ||
| T/T | T/T | ε2/ε2 | lowest risk (rare)
🚩 This is likely your genotype, but double-check that this automated note is correct
|
| T/T | C/T | ε2/ε3 | lower risk
🚩 This is likely your genotype, but double-check that this automated note is correct
|
| C/T | C/T | ε2/ε4 | slightly higher risk than normal
🚩 This is likely your genotype, but double-check that this automated note is correct
|
| T/T | C/C | ε3/ε3 | normal risk
🚩 This is likely your genotype, but double-check that this automated note is correct
|
| C/T | C/C | ε3/ε4 | higher risk
🚩 This is likely your genotype, but double-check that this automated note is correct
|
| C/C | C/C | ε4/ε4 | highest risk[ref]
🚩 This is likely your genotype, but double-check that this automated note is correct
|
APOE types: E2, E3, and E4 alleles
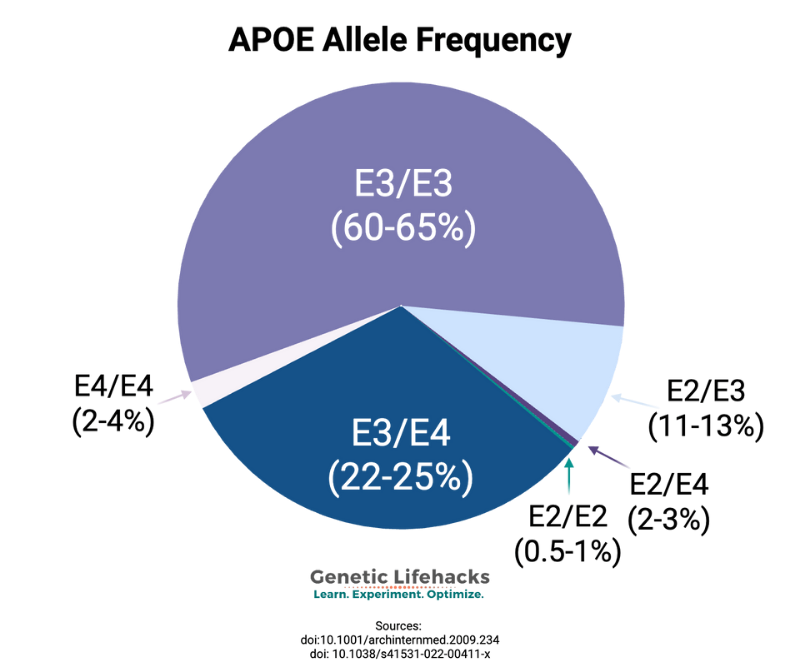
You inherit one APOE allele from your mother and one from your father. Thus, you will have two APOE allele types. There are six possible combinations (listed above): E2/E2, E2/E3, E3/E3, E3/E4, E4/E4, E2/E4.
E3/E3: The APOE E3 type is the normal and most common type. People with two copies of APOE E3 will have what is considered the typical risk allele for Alzheimer’s disease. This generally is estimated to be about a 10-12% lifetime risk by age 85, but it may be lower with lifestyle interventions.[ref]
E2/E3: APOE2 is protective against Alzheimer’s disease. This combination decreases the risk of Alzheimer’s disease significantly, but doesn’t eliminate the possibility of Alzheimer’s.
E2/E4: If you’re heterozygous at both SNPs (CT at each), standard interpretation is ε2/ε4. Theoretically, a rare ε1 haplotype could complicate this, but it is really rare. Studies vary on whether the E2/E4 combination is similar in risk to E3/E3, slightly elevated, or slightly decreased. A recent study showed that the E2/E4 combination had significantly reduced amyloid-beta levels compared to E3/E4.[ref]
E3/E4 or E4/E4: The E4 allele increases the risk of Alzheimer’s disease, with the amount of increased risk dependent on your ancestry and sex.[ref]
Let’s dig into the E4 risk allele further…
What does APOE4 mean?
The APOE E4 allele is considered the ancestral type – the original type of APOE from which the E3 and E2 alleles developed. We will come back to this idea in the lifehacks section when looking at ancestral diets.
Increased Alzheimer’s risk: The APOE E4 genotype elevates the risk of developing Alzheimer’s disease compared to the more common version of the gene, APOE3. The APOE4 allele also increases the risk of developing the disease at a slightly earlier age.[ref]
Women have a higher risk than men: A 2025 study showed that women were at a 1.48X increased relative risk of Alzheimer’s compared to men. Importantly, this seems to be primarily in the age window of 60-75 years. After that, the increase in relative risk is not as pronounced. [ref]
Risk depends on ancestry group: The increase in Alzheimer’s risk depends a lot on ancestry, with Asian E4 allele carriers being at the highest increase in risk and African E4 allele carriers being at a lesser increase in risk.[ref]
Increased Lewy body dementia risk, CAD: In addition to Alzheimer’s disease, the APOE4 allele has also been linked to an increased relative risk of other neurodegenerative conditions, including Lewy body dementia and TDP-43 pathology in Alzheimer’s disease. Additionally, APOE E4 carriers are at a slightly increased relative risk of stroke and cardiovascular disease.[ref][ref][ref]
Let’s break this out by numbers for absolute risk:
10-Year Absolute Risk of Alzheimer’s for ε4/ε4 Carriers by Age and Sex[ref]
(Rasmussen et al. 2018, n=104,537, primarily European ancestry)
| Age Range | Women (ε4/ε4) | Men (ε4/ε4) |
|---|---|---|
| 60–69 | 7% | 6% |
| 70–79 | 16% | 12% |
| 80+ | 24% | 19% |
Other factors involved in Alzheimer’s:
Importantly, APOE type is only one piece of the Alzheimer’s risk picture. Some people with the E4/E4 alleles don’t end up with Alzheimer’s; thus, APOE is not completely penetrant, meaning that other factors are involved.
- Environmental and lifestyle factors play a big role in who gets Alzheimer’s. (Covered in the Lifehacks for Alzheimer’s Prevention section below)
- New research has found that exposure to HSV-1 can contribute to Alzheimer’s risk and progression. [ref][ref]
- In people with Alzheimer’s disease, there is reduced blood glucose uptake and energy (ATP) creation in the brain. Glucose enters the brain through glucose transporters in the blood-brain barrier, and blood-glucose levels may affect risk.[ref]
Your other genes can increase or decrease your risk:
- Genetic mutations that decrease the risk of Alzheimer’s.
- Variants in the KLOTHO gene decrease the APOE E4 elevated risk.
- Fibronectin variants are protective against Alzheimer’s in APOE4 individuals.
While your APOE type isn’t the complete picture, research does show it to be the most significant common genetic risk factor for Alzheimer’s disease.
The APOE E4 allele also has benefits:
For almost every genetic variant with a negative impact common in the population, there is also a tradeoff or benefit. The reason that the allele is common is usually due to positive selection for the genotype.
This holds true even for the APOE E4 allele.
| Benefit | Description |
|---|---|
| Parasite Protection | May aid in clearing parasites and hepatitis C |
| Age-Related Macular Degeneration Protection | Protective effect against macular degeneration |
| Visual Working Memory in Older Age | Better visual working memory at age 70 in some, especially at higher β-amyloid burden |
| Infectious Disease Protection | Lower risk of liver issues in hepatitis C |
| Obesity Paradox | Obesity protective vs. dementia in APOE E4, but risk in E2/E3 alleles |
Parasite protection:
Studies in the Amazon show that older adults with the APOE E4 allele, along with high parasite burdens, were cognitively better off than those with the E3 or E2 alleles. Viral hepatitis C, Giardia, and Cryptosporidium were cleared more easily.[ref]
Protective against age-related macular degeneration:
The APOE E4 allele has been shown in several studies to protect against macular degeneration.[ref]
Visual working memory benefits at age 70:
A study showed that E4 carriers recalled locations more precisely, with the advantage more pronounced at higher amyloid-beta burden. This is evidence that some cognitive benefits of this genotype persist into older age, even in preclinical Alzheimer’s stages.[ref]
Infectious disease protection:
The APOE E4 allele is linked to a decreased risk of chronic hepatitis C and a lower risk of liver problems in people who have hepatitis C. [ref]
Obesity Paradox for APOE and Dementia:
People who are obese have been shown in studies to be at a greater risk of dementia, but not all studies show this result. It turns out that the APOE allele may be the difference.
A recent study showed that people with APOE E2 and APOE E3 alleles are at a greater risk of cognitive decline or dementia if they are obese. However, obesity was protective against dementia and Alzheimer’s when combined with the APOE E4 allele.[ref]
Diagnosis of Alzheimer’s disease can be nebulous:
One problem with Alzheimer’s disease is that it is not a clear-cut diagnosis. There are many different causes of dementia, and historically, the only way to know for sure that it was Alzheimer’s was through autopsy after death. While there are new biomarkers being looked at for diagnosis (none definitive yet), keep in mind that if you have a “family history” of Alzheimer’s, even recent studies show that up to 23% of diagnoses were likely incorrect.[ref]
What else causes the symptoms of Alzheimer’s disease?
Medications, including sleeping aids (benzodiazepines), narcotic pain relievers, antidepressants, anticholinergics, statins, warfarin, and corticosteroids, can all give some people memory and cognitive function problems that are similar to mild Alzheimer’s disease. [ref]
Lifehacks for Alzheimer’s Prevention
If you are at an increased risk for Alzheimer’s, the key is to use this knowledge to do all that you can to decrease your risk.
Download the free eBook on Alzheimer’s prevention
Research on Alzheimer’s prevention:
Here’s an overview of prevention strategies, with details below:
| Strategy | Evidence/Notes |
|---|---|
| Block blue light at night | Supports healthy melatonin/circadian rhythm, linked to prevention |
| Maintain physical activity | Exercise has been shown to lower the risk |
| Brain energy support (cell energetics) | Targeting glucose/prostaglandin-E2 pathways shows promise in mice |
| Saffron extract | As effective as standard AD drugs in trials |
| Low-dose lithium | Epidemiological & clinical trial support for protection |
| Lemon balm extract | Some evidence for improved cognition in mild/moderate AD |
| Support liver health | TUDCA & altered bile acid profile in studies |
| Use NSAIDs (cautiously) | Population studies show reduced risk, but risk varies by genotype |
| Avoid processed meat, Consume more unprocessed red meat (E4) | Higher unprocessed red meat consumption protective for APOE E4 |
| Monitor cardiovascular health/homocysteine | High homocysteine = higher risk |
| Maintain healthy oral hygiene | P. gingivalis & oral microbiome linked to AD risk |
| Reduce saturated fat intake (APOE E4) | Some evidence this may help in E4 carriers reduce heart disease risk |
| Weigh hormone therapy risk | In APOE E4 women, HRT increases risk markers |
| Use of luteolin, DHA/EPA (fish oil) | Potential neuroprotective effects, variable by genotype |
| Minimize alcohol (APOE E4 carriers) | Alcohol increases the risk of cognitive decline in E4 |
Circadian Rhythm and Melatonin:
Number one on my list for preventing Alzheimer’s is blocking blue light at night to boost melatonin and help with circadian rhythm. There is a ton of research showing a strong connection between circadian rhythm disruption, melatonin production, insulin regulation, and healthy brain aging.[ref][ref][ref][ref]
Our natural circadian rhythm causes melatonin to rise in the evening and stay elevated until morning. Light in the shorter, blue wavelengths signals through receptors in our eyes to turn off melatonin production in the morning.
However, our modern environment with electric lights at night, especially TVs and phones, disrupts our natural circadian rhythm.
You can either block blue light with 100% blue-light-blocking glasses or simply avoid electronics and bright overhead light for a couple of hours before sleep. Blue-blocking glasses, worn in the evening for several hours before bed, increase natural melatonin production by about 50% in just two weeks.[ref]
Read more about Light at Night and Alzheimer’s Risk. (in-depth on all the research on circadian rhythm)
What about supplemental melatonin?
Clinical trials are also evaluating the use of melatonin supplements for Alzheimer’s.[ref] Additionally, melatonin has been shown to positively reduce any increase in cardiovascular disease risk associated with the APOE E4 allele.[ref]
Read more about Supplemental Melatonin research studies and dosage details.
Brain energy and neurodegenerative diseases:
Some researchers have dubbed Alzheimer’s ‘type 3 diabetes’ due to changes in how glucose is metabolized in the brain, but it isn’t a true form of diabetes.
A study in mice investigated the changes in metabolism in brain cells linked to Alzheimer’s and cognitive decline in aging. In this study, the researchers showed that energy production is reduced in microglia and macrophages in response to increased prostaglandin E2, an inflammatory signal. Specifically, prostaglandin E2 caused glucose to be stored as glycogen rather than being used for cellular energy production. Most importantly, the study showed that inhibiting the EP2 receptor for prostaglandin E2 in myeloid cells was sufficient to increase cellular energetics. Blocking that prostaglandin EP2 receptor reversed cognitive aging in mice.[ref]
Creatine: A 2025 pilot study found that 20g/day of supplemental creatine was well tolerated by patients with Alzheimer’s, and the researchers hypothesized that this may increase cognitive performance in Alzheimer’s. [ref]
Related article: Creatine and cognitive performance
Coconut or MCT oil: Medium-chain triglycerides can be used by the brain more easily for energy. A small placebo-controlled clinical trial looked at using coconut oil, a medium-chain triglyceride, in elderly adults diagnosed with mild or moderate dementia. The coconut oil arm of the study took about 1.8 tablespoons per day, and for the second half of the study, the crossover placebo arm received olive oil. The results showed that 80% of the participants had improved or stable cognitive scores regardless of APOE4 status.[ref]
Red meat consumption and APOE E4:
A 2026 study showed that processed meat consumption, such as sausages and deli meats, increases the risk of Alzheimer’s (a little bit) for all APOE types. However, the same study showed that high unprocessed red meat consumption decreased Alzheimer’s risk significantly in APOE E4 allele carriers (APOE E3/E4 or E4/E4). There was no benefit from red meat for APOE3 or E2 genotypes. The researchers theorized that the APOE E4 genotype is ancestral to a time before agriculture. The E3 allele is thought to be an adaptation that was beneficial when agriculture and farming became more common.[ref]
Hormone replacement therapy in menopause:
There are conflicting epidemiological studies on whether hormone replacement therapy is beneficial or detrimental for dementia, and part of the discrepancy may be APOE types. A recent (2025) study involving women in the ongoing, long-term Wisconsin Registry for Alzheimer’s Prevention project looked at the effect of hormone replacement therapy in women with different APOE types. The results showed that women with APOE E4 had increased markers for Alzheimer’s, including increased amyloid beta and phosphorylated tau when using HRT. Women without the APOE E4 allele did not have negative effects (regarding Alzheimer’s markers) from HRT.[ref]
Low-dose (microdose) Lithium:
Research shows that the mineral lithium may help prevent Alzheimer’s. Lithium is naturally found in food sources; most people consume up to 3 mg/day from food.
Lithium Orotate can be purchased as a mineral supplement in 5 mg doses. (Note, this is a much, much lower dose than is used in the lithium carbonate prescription medication for bipolar disorder.)
Epidemiological studies show that higher lithium levels in drinking water may help protect against dementia. A clinical trial showed that low lithium doses helped AD patients have no decrease in cognitive impairment over 15 months.[ref]
Cautions: There are interactions between lithium and several prescription drugs, so always check with your doctor or pharmacist before starting any supplement.
Full article on lithium with references and details: Lithium Orotate: Mood, Alzheimer’s, and Aging
Resveratrol for neuroinflammation
For mild to moderate Alzheimer’s disease, a small, year-long study involving <1g/day of resveratrol showed that it attenuated declines in mini-mental status examination (MMSE) scores. Neuroinflammation markers improved significantly.[ref]
Related article: Resveratrol Studies, Genetic Interactions, and Bioavailability
Lemon balm (Melissa officinalis) extract with rosmarinic acid:
A small placebo-controlled clinical trial showed that Melissa officinalis extract actually improved cognitive function in people with mild to moderate Alzheimer’s. The study was done in Iran and involved 42 Alzheimer’s patients.[ref]
Lemon balm contains rosmarinic acid, which other studies have shown may affect memory and cognition. It also contains low amounts of harmine, which could help to prevent glutamate toxicity.[ref][ref]
Lifestyle changes for Alzheimer’s prevention:
Lifestyle plays a significant role in Alzheimer’s risk and timing of onset. Modifying lifestyle and diet has been shown to prevent or delay Alzheimer’s disease. [ref]
Bredesen Protocol:
Dr. Dale Bredesen has developed a lifestyle and diet protocol for preventing cognitive decline in Alzheimer’s patients. A 2024 study showed that the protocol, individualized for the patient, was effective for most in sustaining cognitive improvements.[ref] His book is called The End of Alzheimer’s. It has good reviews, but there are also published critiques if you want a more nuanced picture of the protocol.
Access this content:
An active subscription is required to access this content.
Conclusion:
Understanding your APOE type is a starting point for knowing what to prioritize for brain health. For some, this information can be alarming at first, but for most, it simply confirms what family history already suggests. Either way, research shows that APOE is only part of the picture for Alzheimer’s risk. Lifestyle choices made years or even decades before symptoms appear can definitely influence your brain health in aging.
Whether you carry zero copies of the E4 allele or two, the prevention strategies in this article are simple but powerful tools for protecting your brain. Finally, knowing your risk is only valuable if you use that knowledge. Make a plan and implement it, based on the current research, but revisit your plan as the research evolves over time.
Related Articles and Topics:
Serotonin 2A receptor: Psychedelic response and Alzheimer’s disease
Alzheimer’s and Light at Night: Taking action to prevent this disease
References: