Key takeaways:
~ Long Covid is the persistence of symptoms after having had and recovered from COVID-19.
~ Fatigue, brain fog, heart problems, autonomic nervous system, and breathing issues are the most common symptoms, but other systemic issues can also occur.
~ It is a heterogeneous condition with individuals having different groups of symptoms.
~ Genetic research shows which genes are involved in susceptibility.
~ Research on long Covid points towards multiple pathways being involved, which means that the solutions may be different for individuals.
Long Covid: Long-term effects from SARS-CoV-2 and/or the spike protein
Long Covid, also known as post-acute sequelae of COVID (PASC), refers to the range of health problems that can develop after a Covid infection. Symptoms vary a lot and can affect multiple systems in the body for months or years after Covid.
Common symptoms include fatigue, brain fog, breathing problems, and heart-related issues.
Let’s start with some background on how viruses can persist in the body, and then go into the research on the body systems involved. We’ll also look at how genetic variants increase susceptibility (and what that means), as well as the clinical trials on possible solutions for long Covid.
How does Covid work? Viral replication and receptors
Viruses are non-living strands of RNA or DNA that can only replicate inside a host cell. Essentially, they have to be taken into a cell, and then the host cell replicates the viral genetic material. The viral genes are then translated into their proteins and packaged up by the host cell. The replicated virus then gets released (usually) into the host so that it can infect other cells. Rinse and repeat.
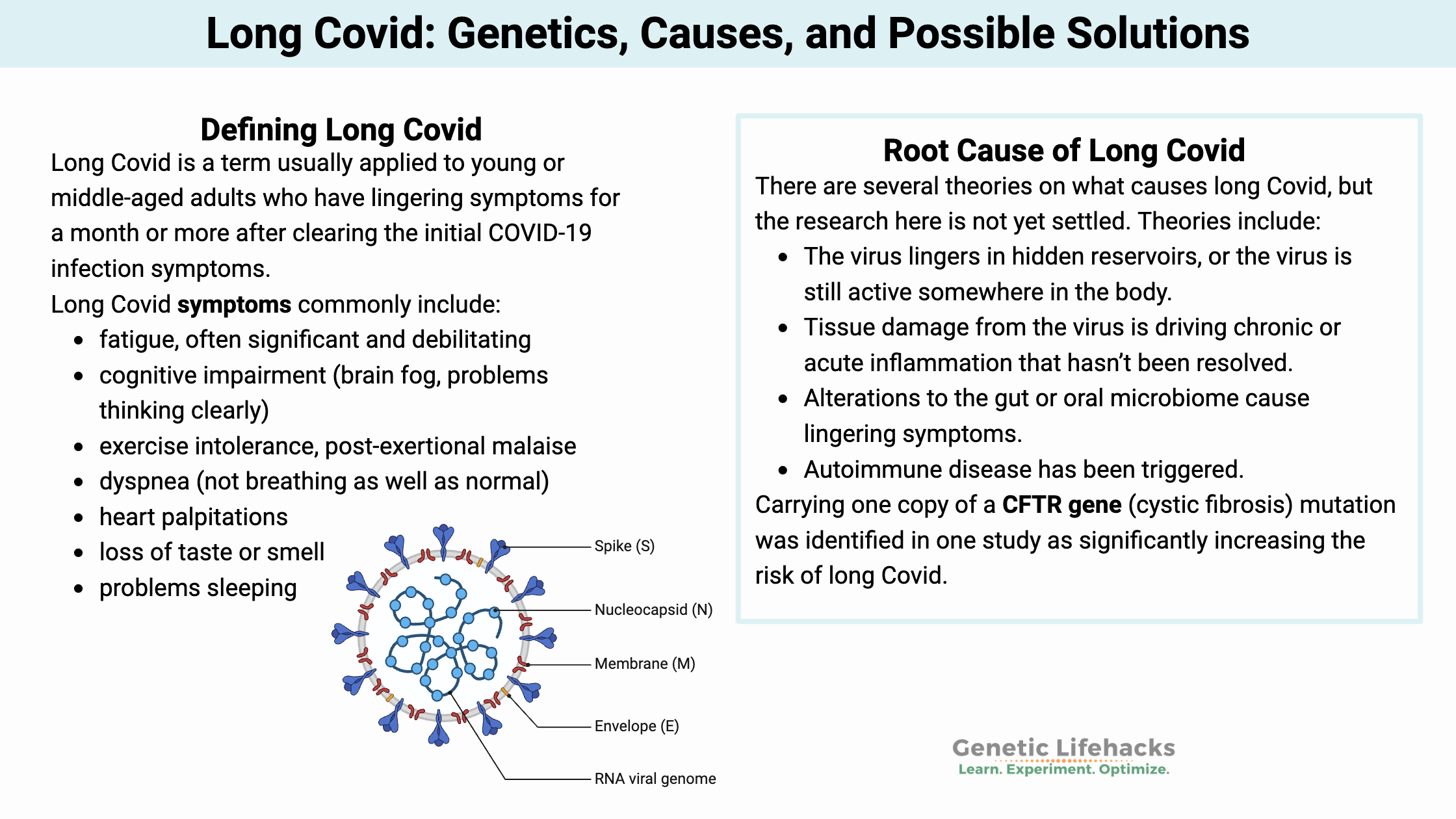
SARS-CoV-2 is a member of the coronavirus family. It is an airborne enveloped, single-stranded RNA virus. Other coronaviruses cause the common cold in people, or respiratory diseases, such as kennel cough, in animals. The outside layer of the virus contains spike proteins that bind to a couple of receptors located on the membranes of host cells. [ref]
Receptors that the spike protein can bind to include:
- ACE2 receptor: Found on the membranes of cells in the lungs, stomach, nose, brain, prostate, testis, placenta, kidney, intestines, and liver.[ref][ref][ref]
- CD147 receptor: platelets express the CD147 receptor, allowing the virus to enter platelets through this receptor.[ref][ref]
- NRP1 receptor: neuropilin-1, which is a receptor highly expressed in the brain. [ref][ref][ref]
Related article: Neuropilins
As the SARS-CoV-2 virus enters the host cell, it releases its viral RNA into the cell. The host cell then replicates and transcribes the viral RNA into the viral proteins. The viral proteins are taken into the host cell’s endoplasmic reticulum (ER) membrane, where the virus is assembled. It is then translocated to the cell surface to be released via exocytosis to infect other cells.[ref]
For many people, the immune system kicks in and gets rid of the virus. However, for people with long Covid, there is either viral or spike protein persistence – or – there are long-lasting cellular or immune system changes.
Let’s take a quick look at how the immune response works to understand what could be going on here.
Immune response to viruses: Innate and Adaptive
For any virus or bacterial infection, there’s a two-phase response: innate (immediate, non-specific) and adaptive (specific antibodies).
Innate Immune Response:
The innate immune system mounts an immediate response to any foreign pathogens, such as viruses, that is not specific to which virus has invaded. Interferons are produced as one of the first responders in fighting a virus.
For the SARS-CoV-2 virus, the host’s cells recognize patterns associated with viral particles (pattern recognition receptors). This recognition is a general response to any virus with particles that fit a recognized pattern.
In SARS-CoV-2 infections, parts of the RNA genome are recognized by Toll-like receptors (TLRs) 3, 7, and 8, as well as MDA5 and RIG-I. Activation of these immune system receptors triggers an antiviral response, leading to the induction of a couple of types of interferon as well as proinflammatory cytokines.
When this initial innate immune response is enough to control the infection, it results in the SARS-CoV-2 virus causing mild, cold-like symptoms. Research points to an appropriately strong innate immune response being key to not having severe Covid symptoms. This innate response can be dysregulated in the elderly, leading to susceptibility to viral infections. Genetics also plays a role in whether the innate immune response will kick the virus to the curb. People with rare mutations in specific toll-like receptors or interferon-related genes end up with severe Covid-19, even if young and healthy.[ref]
Related article: TLR7 common variants and rare mutations
Of note here – if parts of the virus persist, then the innate immune response may remain active.
Adaptive Immune Response:
The adaptive immune response takes a bit longer (days), but it is specific to the virus. This additional time is important for the body to produce specific antibodies to bind to the virus and make it non-infective. IgM antibodies are produced in large amounts for a few weeks, and then IgG antibodies can last for a while after an infection to prevent reinfection.
There is a second type of adaptive immune response called cell-mediated immunity. This type of immunity refers to T cells, which are a type of white blood cell that present antigens on their surface. Several different subtypes of T cells are involved in the immune response to SARS-CoV-2, including CD4+ and CD8+ T cells.
In SARS-CoV-2 infections, neutralizing antibodies are produced that bind to the S1 unit of the spike protein and block it from entering cells. In looking at the immune response of people with either asymptomatic, mild, or severe Covid, the researchers found that they all had a similar antibody response. Asymptomatic people, in this particular study, had a better natural killer cell response.[ref]
Research studies on long Covid:
First, is it all in your head?
Ask anyone with long Covid, and they have likely had doctors, friends, or relatives who have scoffed at their symptoms. Psychosomatic, psychogenic, or functional neurological disorder — are all terms that have been applied to indicate that long Covid is just a psychological condition that is all in the patient’s head.[ref] This echoes a lot of research articles about ME/CFS from the 80s and 90s, which inferred that the fatigue was psychosomatic and the patients should be prescribed antidepressants.[ref][ref][ref]
Here are just a few examples in print:
- A Wall Street Journal article from March 2021 implied that long Covid could be all in the patients’ heads.
- A Spectator article in 2022 echoes the sentiment, asking “Is long Covid all in the mind?” and referencing studies on the overlap with anxiety.
- A peer-reviewed study highlights this thinking: “…we should not dismiss their complaints as being all in their head. This may not be true and, even if correct, is not helpful.”[ref]
The answer is “NO”. The “it’s all in your head” idea has now been thoroughly debunked with a multitude of studies showing significant, physiological, and cellular changes occurring in both long Covid and ME/CFS.[ref][ref]
Defining Long Covid: Symptoms for more than 3 months
Long Covid is a term usually applied to children or adults who have lingering symptoms for a month or more after clearing the initial Covid infection symptoms.[ref][ref][ref] Older people who had severe COVID-19 and ended up in the ICU often have long-term decreased functional capacity due to cell damage. For example, lung problems persist for many months in Covid patients after an ICU stay. This is sometimes also lumped into the long Covid terminology. [ref]
Long Covid symptoms commonly include:
| Symptom Category | Specific Symptoms | Frequency | Possible Mechanism |
|---|---|---|---|
| Fatigue | Debilitating exhaustion, post-exertional malaise, problems sleeping | Very Common | Mitochondrial dysfunction, reduced ATP |
| Cognitive | Brain fog, memory problems, headache, concentration issues | Very Common | Neuroinflammation, reduced brain metabolism |
| Respiratory | Shortness of breath, exercise intolerance | Common | Lung tissue damage, complement activation |
| Cardiovascular | Heart palpitations, tachycardia, POTS | Common | Heart tissue damage, autonomic dysfunction |
Post-infectious symptoms aren’t new or unexpected:
It’s important to understand that the long-term effects of viral infections aren’t new, but they are specific to the virus. For example, hearing loss is a complication of the mumps, and poliovirus can cause long-term disability. Lyme disease causes chronic symptoms in some people. Herpes simplex 1 increases neurodegenerative disorders later in life, and post-viral ME/CFS is linked to EBV. [ref][ref][ref][ref][ref]
Root cause(s) of long Covid:
With the wide range of symptoms, different causes could be impacting individuals independently.
There are several theories on what is going on with long Covid, and the research here is not yet settled. Theories include:[ref]
| Mechanism | Key Finding | Prevalence | Clinical Significance |
|---|---|---|---|
| Mitochondrial dysfunction | Reduced ATP in muscle, immune cells | High in PEM patients | Explains exercise intolerance |
| EBV reactivation | 67% vs 10% in controls | High | Secondary immune activation |
| Viral persistence | SARS-CoV-2 RNA >100 days | Variable | Ongoing immune activation |
| Complement activation | Alternative/classical pathways | Common | Systemic inflammation |
| Persistent Immune activation | T cells, B Cells | Unknown | Immune exhaustion, inflammatory cytokines |
| Gut microbiome | Changed composition | Common | Altered metabolites, immune activation |
Let’s dig into some of the research findings:
Mitochondrial dysfunction in immune cells:
Researchers looked at gene expression in multiple cell types in people with long Covid, acute Covid, and no known Covid. The results showed very significant differences in pathways involved in mitochondrial dysfunction, but only in T cells, B cells, and natural killer cells. Importantly, these immune system cells were not infected with SARS-CoV-2, nor did they have higher-than-normal viral RNA. The dysregulated mitochondria-related genes included both mtDNA and nuclear DNA, with upregulated genes causing an increase in ATP production. This suggests chronic immune activation. The authors theorize that either stem cells are infected or there is persistent viral antigen production that is activating the immune system.[ref]
Muscle and mitochondrial abnormalities in post-exertional malaise (PEM):
A January 2024 study explains a lot about what is occurring at a cellular level in people who have exercise intolerance or post-exertional malaise (PEM) after Covid. (Note that post-exertional malaise also includes mental exertion.)
The study found significant physiological changes in muscle tissue in long Covid patients with PEM. First, they found that exercise or muscle exertion causes amyloid-containing deposits to form in the muscles. Additionally, there were systemic metabolic disturbances in the way the skeletal muscle cells worked. The long Covid patients had lower oxidative phosphorylation capacity, meaning their mitochondria weren’t producing enough ATP for cellular energy. The long Covid patients’ cells had a normal number of mitochondria, but they weren’t functioning like they should. In addition, there was evidence of severe muscle tissue damage with a blunted T-cell response.[ref – open access, worth reading in full]
Also important is what the study didn’t find. The researchers didn’t find that the muscles had increased breakdown products or higher cortisol levels. They also didn’t find activation of damaged protein systems (DAMP). While there was evidence of SARS-CoV-2 nucleocapsid protein in the muscle, the levels did not differ from the healthy controls.
Complement system activation:
The complement system is part of the innate immune response. It can be activated by pathogens or it can respond to damaged cells.[ref]
A recent proteomics study of 268 long Covid patient samples found that there was ongoing activation of the alternative and classical complement pathways. There were also increased antibodies against several herpesviruses.[ref]
Related article: Mannose-binding lectin genes
Lingering spike protein:
Research shows that the spike protein alone (without the virus) causes an immune response, including abnormal clotting.[ref][ref]
- A recent study found that CD16+ monocytes had persistent spike (S1) proteins in patients with PASC (Post-acute sequelae of COVID) long after the infection should have cleared. CD16+ monocytes interact with the endothelium and with platelets.[ref]
- Another study found similar results with spike protein fragments found in a long Covid patient’s monocytes 15 months post-infection.[ref]
- The spike protein is also given in vaccines. These studies on PASC/long Covid patients don’t include information on whether the patients were also vaccinated. A recent study published in Cell shows that the spike protein mRNA is present in some lymph nodes eight weeks after vaccination.[ref]
Reactivation of Epstein-Barr Virus:
A study of patients with long Covid showed that 67% had positive Epstein-Barr titers, showing a reactivation of the virus. (Only 10% of the healthy control group had active Epstein-Barr virus titers.) The study concludes: “These findings suggest that many long COVID symptoms may not be a direct result of the SARS-CoV-2 virus but may be the result of COVID-19 inflammation-induced EBV reactivation.”[ref]
Related article: Epstein-Barr Virus: Genetic Risks, Reactivation, and Chronic Illnesses
Another recent study showed that people who had reactivated EBV before getting Covid were twice as likely to develop long Covid. Interestingly, having had cytomegalovirus was protective against the neurocognitive symptoms of long Covid.[ref]
Persistent clotting problems and inflammation:
A study looked at plasma samples from long Covid /PASC patients and found large anomalous deposits (microclots). The clots were resistant to fibrinolysis. Additionally, the researchers noted various inflammatory cytokines that were elevated in long Covid patients, including “α(2)-antiplasmin (α2AP), various fibrinogen chains, as well as Serum Amyloid A (SAA)”.[ref] Serum amyloid A is an acute-phase protein indicating active inflammation.
Post-COVID-19 Tachycardia Syndrome:
Researchers have identified that a subset of patients end up with heart palpitations or tachycardia that persists for 3+ months after Covid. This abnormality can also include POTS (postural tachycardia syndrome) patients. Interestingly, the researchers did not find that Holter ECG monitoring matched with reported symptoms. The researchers think that direct and indirect damage to the heart from the viral infection may lead to post-Covid tachycardia syndrome. Additional causes are theorized to include persistent lung injury, persistent fevers, pain, anxiety, or depression.[ref]
Related article: POTS, genes
Altered microbiome:
Changes to the gut microbiome or oral microbiome due to SARS-CoV-2 could lead to immune dysregulation and chronic inflammation.
- A study of COVID-19 patients looked at changes to the gut microbiome in the participants who still had lingering symptoms at 6 months (mainly fatigue, brain fog, and hair loss). The gut microbiome of long Covid patients showed persistent differences. “Butyrate-producing bacteria, including Bifidobacterium pseudocatenulatum and Faecalibacterium prausnitzii, showed the largest inverse correlations with PACS at 6 months.”[ref]
- A study of the oral microbiome in patients with COVID-19 showed that those who developed long Covid had significant differences in the oral microbiome. The long Covid patients had “significantly higher abundances of microbiota that induced inflammation… The oral microbiome of patients with long COVID was similar to that of patients with chronic fatigue syndrome.”[ref]
Persistent Immune System Changes:
A study of upregulated genes in people with long Covid showed that platelet-related pathways were downregulated, and genes involved in transcription, translation, and the cell cycle were upregulated. The downregulation of platelet-related genes (e.g. platelet factor 4, coagulation factor XIIII) may correlate to thrombocytopenia (low platelet count) seen in COVID-19 patients – and be a source of fatigue. Interestingly, the researchers also identified interferon-related genes as being downregulated.[ref]
Systemic inflammation:
Elevated inflammatory cytokines are found in people with long Covid.
- A PET/CT study of people with long Covid showed that systemic inflammation might be the underlying cause. The imaging study found hypometabolism in the brain, which is typical of whole-body inflammatory changes. It means that regions of the brain are literally not getting enough energy, and the cause is likely inflammation in the body.[ref] What the study doesn’t show is whether the systemic inflammation is due to lack of resolution of inflammation, persistence of viral infection, or sterile inflammation due to cell death from the inflammatory response to the virus.
- TNF-alpha and interferon-gamma: Another preprint describes the differences between people who fully recovered from Covid and people with long Covid. “participants with respiratory PASC had up to 34-fold higher frequencies of IFN-γ- and TNF-α-producing SARS-CoV-2-specific CD4+ and CD8+ T cells in peripheral blood and elevated levels of plasma CRP and IL-6.”[ref]
Spike protein in the brain:
Some researchers theorize that the spike protein causes neurological issues in long Covid. This researcher points to indirect evidence of the spike protein being released and possibly crossing the blood-brain barrier. Animal studies show that the spike protein alone can cross the blood-brain barrier.[ref][ref]
Persistence of virus:
Many viruses can stick around in the body, usually in a latent form, for a long time. For example, the varicella-zoster virus that causes chickenpox lies dormant in the nervous system and can reactivate as shingles in adulthood. The question is whether long Covid symptoms are caused by the SARS-CoV-2 virus still lingering, at low levels, somewhere in the body.
- A study on viral persistence shows that people with long Covid have higher cytokine levels, such as TNF-alpha and IL-6. The researchers thought that this might indicate persistent infection.[ref]
- Lung tissue biopsy showed the persistence of SARS-CoV-2 RNA in the lungs more than 100 days after a mild infection.[ref]
- Intestinal biopsies of asymptomatic individuals also showed SARS-CoV-2 RNA in half the patients at four months post-infection.[ref]
- A trial using sweat samples and dogs that can sniff Covid found that about half the long Covid patients tested (sniffed) positive for viral infection.[ref]
Autoimmune antibodies:
A study of patients with severe COVID-19 found that 34% of them were positive for antinuclear antibodies, another 34% were positive for anti-β2GPI antibodies, and several other antibodies were also determined. Importantly, none of the patients in the study had previously been diagnosed with a rheumatic disease (which would be a source of these specific autoimmune-related antibodies).[ref][ref] The question, though, is how long the autoimmune antibodies last after an illness. It is thought that the low-level autoimmune antibodies will likely fade away fairly quickly after the illness.[ref]
Genetics of long Covid:
A number of genetic studies have looked at why some people get long Covid while others don’t. In general, genes related to innate immune function as well as T cell function have been identified.
Genome-wide association studies (GWAS) compare the genomes of people with a specific disease to the population as a whole in order to see which genes are likely involved. GWAS studies on long Covid have found:
Transcription factor, lung function:
The FOXP4 gene was identified in a huge GWAS study as being likely involved in the lung function problems of long Covid. The variant was tied to a more than doubled risk of long Covid. In people with long Covid, researchers also found higher FOXP4 levels, including in the alveolar cells in the lungs.[ref]
FOXP4 codes for a transcription factor that is found in mucous-secreting cells of the lungs and intestines, as well as in B-cells, natural killer cells, and T reg cells. In the lungs, FOXP4 promotes the transcription of genes needed for airway epithelial cell regeneration. Changes in FOXP4 are also linked to lung fibrosis.[ref]
Sex differences:
An analysis of genetic data from 2400 covid patients showed that there were differences in genetic susceptibility for males vs. females. The results showed that in males with long Covid, genetic differences were seen in CD5 and VPS37C, which are both linked to immune system function. CD5 is interesting in that it is expressed on T cells and regulates immune response. VPS37C has previously been identified as a gene involved in Kawasaki disease in children.[ref]
23andMe GWAS Study:
A genome-wide association study by 23andMe is available as a preprint study. Using information from about 180,000 23andMe users, this GWAS and Mendelian randomization study looked at the genetic variants of people with persistence of symptoms for more than a month after infection. The results of the study found that long Covid is likely related to the HLA region and to blood type.
| Gene | RS ID | Risk Allele | Function | Impact on Long COVID |
|---|---|---|---|---|
| PEDS1 | rs6020298 | G | Plasmalogen synthesis enzyme | 2x increased susceptibility (G/G) |
| NADSYN1 | rs12785878 | T | NAD synthetase for ATP production | 2x increased risk (T/T) |
| IL10 | rs1800896 | C | Anti-inflammatory cytokine | Increased risk (C/C, C/T) |
| FOXP4 | rs2496644 | A | Transcription factor, lung function | Increased risk (A/A, A/C) |
| CLOCK | rs62303689 | A | Circadian rhythm regulation | Affects fatigue susceptibility |
Long Covid reveals rare genetic diseases:
Stressing the body and the immune system may reveal the genetic susceptibility due to rare mutations that are linked to genetic diseases. A study involving whole genome sequencing of 95 long Covid patients found that 57 of the patients had mutations in genes that were classified as pathogenic or linked to causing a genetic disease. The researchers then did a genetic analysis to determine if the pathogenic mutations were likely to play a role in the long Covid symptoms, and many of them did. For example, CFTR mutations that can cause cystic fibrosis were twice as common in the long Covid cohort compared to normal, and POLG mutations related to mitochondrial diseases were also much more prevalent than they should have been.[ref]
Epigenetic changes and gene expression:
New research suggests that SARS-CoV-2 infection causes changes in gene expression in important ways. Infection has been shown to cause changes in the chromatin structure of DNA, which alters which genes are accessible for translation into proteins.
One study found that there is an upregulation of pro-inflammatory cytokine production and cell adhesion proteins. It also found that there were overall alterations in transcription factors.[ref]
More research is needed here to understand whether some of the changes in gene expression are continuing after the active infection and possibly causing long Covid symptoms. Epigenetic changes are seen for up to a year in hematopoietic stem and progenitor cells, which produce immune cells (T cells) and red blood cells. [ref][ref]
Gut microbiome, genetics interaction:
A Mendelian randomization study used known genetic variants in the host (human) genome to understand likely gut microbiome alterations in people with long Covid. The results showed that variations in Parasutterella, Oscillospira, and Eisenbergiella levels in the gut microbiome (due to host genetic variant) could be contributing causally to long Covid susceptibility.[ref] Parasutterella is particularly interesting. Not only is it associated with the mucosal inflammatory response, but it is also linked to tryptophan and kynurenine metabolism.[ref][ref]
Genotype report: Long Covid
Access this content:
An active subscription is required to access this content.
Lifehacks: Possible solutions for long Covid
First, I’ll present information on natural supplements that may work for specific genetic variants, and then a roundup of the natural supplements with clinical studies for long Covid.
Genetic interactions to target with long Covid supplements:
The question for many people with long Covid when looking at supplements or treatment options is: Where do I start? Below are research studies on natural supplements for long Covid, along with possible genetic connections. I want to be very clear that for many of these supplements, there are no direct studies on them for long Covid.
Access this content:
An active subscription is required to access this content.
Related Articles and Topics:
References:
“Axcella Commences Trial of AXA1125 for Long Covid-19 Treatment.” Clinical Trials Arena, 27 Oct. 2021, https://www.clinicaltrialsarena.com/news/axcella-trial-long-covid-treatment/.
Barrett, Tessa J., et al. “Platelets Contribute to Disease Severity in COVID‐19.” Journal of Thrombosis and Haemostasis, vol. 19, no. 12, Dec. 2021, pp. 3139–53. PubMed Central, https://doi.org/10.1111/jth.15534.
Carsetti, Rita, et al. “Different Innate and Adaptive Immune Responses to SARS-CoV-2 Infection of Asymptomatic, Mild, and Severe Cases.” Frontiers in Immunology, vol. 11, Dec. 2020, p. 610300. PubMed Central, https://doi.org/10.3389/fimmu.2020.610300.
Ceulemans, Laurens J., et al. “Persistence of SARS-CoV-2 RNA in Lung Tissue after Mild COVID-19.” The Lancet. Respiratory Medicine, vol. 9, no. 8, Aug. 2021, pp. e78–79. PubMed Central, https://doi.org/10.1016/S2213-2600(21)00240-X.
Devine, Jeremy. “Opinion | The Dubious Origins of Long Covid.” Wall Street Journal, 22 Mar. 2021. www.wsj.com, https://www.wsj.com/articles/the-dubious-origins-of-long-covid-11616452583.
Eskandari-Nasab, Ebrahim, et al. “Meta-Analysis of Risk Association Between Interleukin-17A and F Gene Polymorphisms and Inflammatory Diseases.” Journal of Interferon & Cytokine Research: The Official Journal of the International Society for Interferon and Cytokine Research, vol. 37, no. 4, Apr. 2017, pp. 165–74. PubMed, https://doi.org/10.1089/jir.2016.0088.
Feng, Bo, et al. “Association of Tumor Necrosis Factor α -308G/A and Interleukin-6 -174G/C Gene Polymorphism with Pneumonia-Induced Sepsis.” Journal of Critical Care, vol. 30, no. 5, Oct. 2015, pp. 920–23. PubMed, https://doi.org/10.1016/j.jcrc.2015.04.123.
Glynne, Paul, et al. “Long COVID Following Mild SARS-CoV-2 Infection: Characteristic T Cell Alterations and Response to Antihistamines.” Journal of Investigative Medicine, vol. 70, no. 1, Jan. 2022, pp. 61–67. jim.bmj.com, https://doi.org/10.1136/jim-2021-002051.
Gold, Jeffrey E., et al. “Investigation of Long COVID Prevalence and Its Relationship to Epstein-Barr Virus Reactivation.” Pathogens, vol. 10, no. 6, June 2021, p. 763. PubMed Central, https://doi.org/10.3390/pathogens10060763.
Grandjean, Dominique, et al. Screening for SARS-CoV-2 Persistence in Long COVID Patients Using Sniffer Dogs and Scents from Axillary Sweats Samples. medRxiv, 12 Jan. 2022, p. 2022.01.11.21268036. medRxiv, https://www.medrxiv.org/content/10.1101/2022.01.11.21268036v1.
Hoffer, Edward P. “Long COVID: Does It Exist? What Is It? We Can We Do For Sufferers?” The American Journal of Medicine, vol. 134, no. 11, Nov. 2021, pp. 1310–11. PubMed Central, https://doi.org/10.1016/j.amjmed.2021.05.023.
Lebeau, Grégorie, et al. “Deciphering SARS-CoV-2 Virologic and Immunologic Features.” International Journal of Molecular Sciences, vol. 21, no. 16, Jan. 2020, p. 5932. www.mdpi.com, https://doi.org/10.3390/ijms21165932.
Littlefield, Katherine M., et al. SARS-CoV-2-Specific T Cells Associate with Reduced Lung Function and Inflammation in Pulmonary Post-Acute Sequalae of SARS-CoV-2. bioRxiv, 15 Feb. 2022, p. 2022.02.14.480317. bioRxiv, https://www.biorxiv.org/content/10.1101/2022.02.14.480317v1.
Liu, Qin, et al. “Gut Microbiota Dynamics in a Prospective Cohort of Patients with Post-Acute COVID-19 Syndrome.” Gut, Jan. 2022, p. gutjnl-2021-325989. PubMed Central, https://doi.org/10.1136/gutjnl-2021-325989.
“Long Covid-19: Drug Trial Results to Watch in 2022.” Clinical Trials Arena, 25 Jan. 2022, https://www.clinicaltrialsarena.com/analysis/long-covid-19-drug-trial-results-to-watch-in-2022/.
MacIntyre, Elaina A., et al. “GSTP1 and TNF Gene Variants and Associations between Air Pollution and Incident Childhood Asthma: The Traffic, Asthma and Genetics (TAG) Study.” Environmental Health Perspectives, vol. 122, no. 4, Apr. 2014, pp. 418–24. PubMed, https://doi.org/10.1289/ehp.1307459.
Majumder, Poulami, et al. “Association of Tumor Necrosis Factor-α (TNF-α) Gene Promoter Polymorphisms with Aggressive and Chronic Periodontitis in the Eastern Indian Population.” Bioscience Reports, vol. 38, no. 4, Aug. 2018, p. BSR20171212. PubMed, https://doi.org/10.1042/BSR20171212.
Patterson, Bruce, et al. Targeting the Monocytic-Endothelial-Platelet Axis with Maraviroc and Pravastatin as a Therapeutic Option to Treat Long COVID/ Post-Acute Sequelae of COVID (PASC). 2 Mar. 2022. Research Square, https://www.researchsquare.com/article/rs-1344323/v1.
Peluso, Michael J., et al. Markers of Immune Activation and Inflammation in Individuals with Post-Acute Sequelae of SARS-CoV-2 Infection. medRxiv, 11 July 2021, p. 2021.07.09.21260287. medRxiv, https://www.medrxiv.org/content/10.1101/2021.07.09.21260287v1.
Pretorius, Etheresia, et al. “Persistent Clotting Protein Pathology in Long COVID/Post-Acute Sequelae of COVID-19 (PASC) Is Accompanied by Increased Levels of Antiplasmin.” Cardiovascular Diabetology, vol. 20, Aug. 2021, p. 172. PubMed Central, https://doi.org/10.1186/s12933-021-01359-7.
Ramakrishnan, Rakhee K., et al. “Unraveling the Mystery Surrounding Post-Acute Sequelae of COVID-19.” Frontiers in Immunology, vol. 12, June 2021, p. 686029. PubMed Central, https://doi.org/10.3389/fimmu.2021.686029.
Ricci, Daniela, et al. “Innate Immune Response to SARS-CoV-2 Infection: From Cells to Soluble Mediators.” International Journal of Molecular Sciences, vol. 22, no. 13, June 2021, p. 7017. PubMed Central, https://doi.org/10.3390/ijms22137017.
Röltgen, Katharina, et al. “Immune Imprinting, Breadth of Variant Recognition, and Germinal Center Response in Human SARS-CoV-2 Infection and Vaccination.” Cell, vol. 0, no. 0, Jan. 2022. www.cell.com, https://doi.org/10.1016/j.cell.2022.01.018.
Rowe, Regina K., and Emma L. Mohr. “Special Issue ‘Pediatric Viral Infection Long-Term Consequences.’” Viruses, vol. 14, no. 2, Feb. 2022, p. 343. www.mdpi.com, https://doi.org/10.3390/v14020343.
Ryan, Feargal J., et al. “Long-Term Perturbation of the Peripheral Immune System Months after SARS-CoV-2 Infection.” BMC Medicine, vol. 20, Jan. 2022, p. 26. PubMed Central, https://doi.org/10.1186/s12916-021-02228-6.
Ryu, Jae Kyu, et al. SARS-CoV-2 Spike Protein Induces Abnormal Inflammatory Blood Clots Neutralized by Fibrin Immunotherapy. bioRxiv, 13 Oct. 2021, p. 2021.10.12.464152. bioRxiv, https://www.biorxiv.org/content/10.1101/2021.10.12.464152v1.
Saheb Sharif-Askari, Narjes, et al. “Enhanced Expression of Autoantigens During SARS-CoV-2 Viral Infection.” Frontiers in Immunology, vol. 12, June 2021, p. 686462. PubMed Central, https://doi.org/10.3389/fimmu.2021.686462.
Sapkota, Hem Raj, and Arvind Nune. “Long COVID from Rheumatology Perspective — a Narrative Review.” Clinical Rheumatology, vol. 41, no. 2, 2022, pp. 337–48. PubMed Central, https://doi.org/10.1007/s10067-021-06001-1.
Sollini, Martina, et al. “Long COVID Hallmarks on [18F]FDG-PET/CT: A Case-Control Study.” European Journal of Nuclear Medicine and Molecular Imaging, vol. 48, no. 10, 2021, pp. 3187–97. PubMed Central, https://doi.org/10.1007/s00259-021-05294-3.
Ståhlberg, Marcus, et al. “Post-COVID-19 Tachycardia Syndrome: A Distinct Phenotype of Post-Acute COVID-19 Syndrome.” The American Journal of Medicine, vol. 134, no. 12, Dec. 2021, pp. 1451–56. PubMed Central, https://doi.org/10.1016/j.amjmed.2021.07.004.
Stappers, M. H. T., et al. “Polymorphisms in Cytokine Genes IL6, TNF, IL10, IL17A and IFNG Influence Susceptibility to Complicated Skin and Skin Structure Infections.” European Journal of Clinical Microbiology & Infectious Diseases: Official Publication of the European Society of Clinical Microbiology, vol. 33, no. 12, Dec. 2014, pp. 2267–74. PubMed, https://doi.org/10.1007/s10096-014-2201-0.
Theoharides, Theoharis C. “Could SARS-CoV-2 Spike Protein Be Responsible for Long-COVID Syndrome?” Molecular Neurobiology, Jan. 2022, pp. 1–12. PubMed Central, https://doi.org/10.1007/s12035-021-02696-0.
Tirelli, U., et al. “Fatigue in Post-Acute Sequelae of SARS-CoV2 (PASC) Treated with Oxygen-Ozone Autohemotherapy – Preliminary Results on 100 Patients.” European Review for Medical and Pharmacological Sciences, vol. 25, no. 18, Sept. 2021, pp. 5871–75. PubMed, https://doi.org/10.26355/eurrev_202109_26809.
Vlachoyiannopoulos, Panayiotis G., et al. “Autoantibodies Related to Systemic Autoimmune Rheumatic Diseases in Severely Ill Patients with COVID-19.” Annals of the Rheumatic Diseases, vol. 79, no. 12, Dec. 2020, pp. 1661–63. ard.bmj.com, https://doi.org/10.1136/annrheumdis-2020-218009.
Walitt, Brian, and Elizabeth Bartrum. “A Clinical Primer for the Expected and Potential Post-COVID-19 Syndromes.” Pain Reports, vol. 6, no. 1, Feb. 2021, p. e887. PubMed Central, https://doi.org/10.1097/PR9.0000000000000887.
Wang, Ke, et al. “CD147-Spike Protein Is a Novel Route for SARS-CoV-2 Infection to Host Cells.” Signal Transduction and Targeted Therapy, vol. 5, no. 1, Dec. 2020, pp. 1–10. www.nature.com, https://doi.org/10.1038/s41392-020-00426-x.
Wu, Jun-Cang, et al. “Gene Polymorphisms and Circulating Levels of the TNF-Alpha Are Associated with Ischemic Stroke: A Meta-Analysis Based on 19,873 Individuals.” International Immunopharmacology, vol. 75, Oct. 2019, p. 105827. PubMed, https://doi.org/10.1016/j.intimp.2019.105827.
Yong, Shin Jie. “Long COVID or Post-COVID-19 Syndrome: Putative Pathophysiology, Risk Factors, and Treatments.” Infectious Diseases (London, England), pp. 1–18. PubMed Central, https://doi.org/10.1080/23744235.2021.1924397. Accessed 2 Mar. 2022.
Yucesoy, Berran, et al. “Genetic Variants in TNFα, TGFB1, PTGS1 and PTGS2 Genes Are Associated with Diisocyanate-Induced Asthma.” Journal of Immunotoxicology, vol. 13, no. 1, 2016, pp. 119–26. PubMed, https://doi.org/10.3109/1547691X.2015.1017061.