Key takeaways:
- POTS (postural orthostatic tachycardia syndrome) is a problem with how the autonomic nervous system regulates heart rate, causing it to increase suddenly when standing.
- Genetic variants can make you more susceptible to the condition, and understanding your genes can help you target the right therapy.
- Genes linked to POTS include HLA genes, SLC6A2 (norepinephrine transporter), GNB3, and NOS3 (nitric oxide synthase).
- Epigenetic modifications, especially those affecting the norepinephrine transporter, are also associated with POTS.
Postural Orthostatic Tachycardia Syndrome (POTS): Background Science, Dysautonomia
POTS (postural orthostatic tachycardia syndrome) is a problem with how your autonomic nervous system regulates heart rate when you change position, such as going from lying down to standing up. It is classified as a type of dysautonomia, or a dysfunction of the autonomic nervous system.
The change in heart rate and the autonomic function gives rise to several symptoms, including fatigue, brain fog, and shaking, in addition to lightheadedness when standing up.
Doctors define POTS as:
- A heart rate increase of 30 BPM within the first 10 minutes of standing for adults (40 BPM for children and teens)
- Or an increase in heart rate to over 120 BPM within the first 10 minutes of standing[ref]
These requirements define POTS as long as the person doesn’t have orthostatic hypotension, a condition where blood pressure initially drops when standing. The Dysautonomia International website explains that POTS impacts 3 million people in the US and more around the world.
Symptoms of Postural Orthostatic Tachycardia Syndrome:
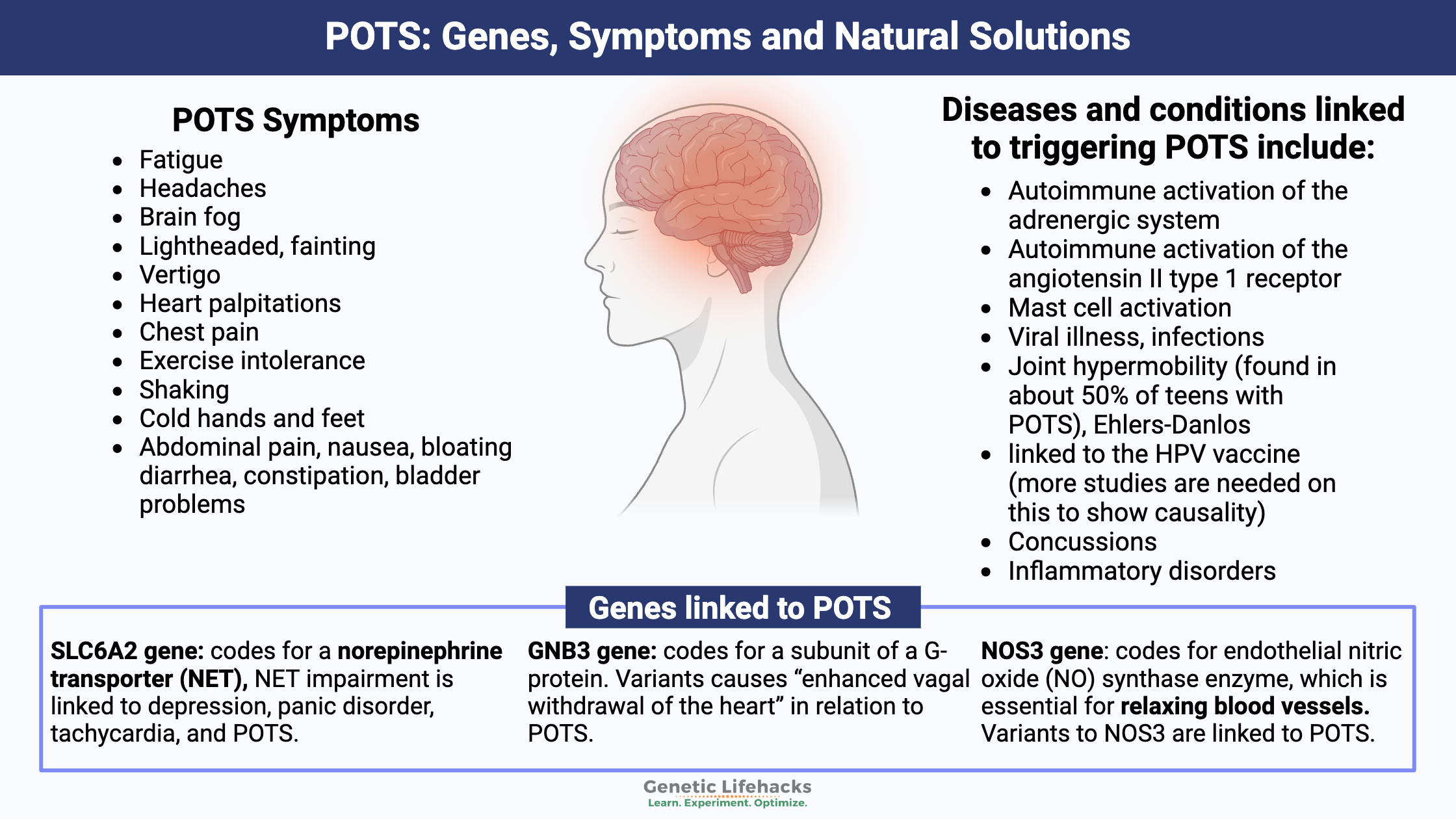
POTS symptoms defined in research studies include[ref][ref][ref][ref][ref][ref]:
- fatigue
- headaches, brain fog, impaired attention
- lightheaded, fainting, vertigo
- heart palpitations, chest pain
- exercise intolerance
- shaking, cold hands and feet
- abdominal pain, nausea, bloating, nerve pain
- diarrhea, constipation, bladder problems
POTS symptoms are often made worse by heat stress.
What happens to blood pressure when standing up?
When someone who does not have POTS stands up, the body goes through a series of regulatory changes that alter heart rate. Because blood pressure and heart rate regulation are automatic, we aren’t even aware that they are taking place.
- Upon standing, gravity causes blood to go from the chest to the lower abdomen and legs. Within the first 30 seconds of standing, there is a fluid shift between the blood vessels and the space in between the cells.
- This change in blood volume causes receptors in the heart to be activated and alters the heart’s stroke volume.
- All of this causes the heart rate to increase just a little bit normally. It also alters blood pressure slightly (decreased systolic BP, increased diastolic BP).[ref]
Thus, when standing up, a slight increase in heart rate and BP is normal.
What happens when a person with POTS stands up?
There are a couple of different scenarios of what is happening in people with POTS (more details below). But generally, POTS can be caused by low blood volume or by the blood vessels in the legs not constricting enough.[ref]
- Reduced blood volume:
If the person with POTS is hypovolemic, with reduced blood volume, they usually have an elevated heart rate even when at rest. This heart rate elevation worsens upon sitting or standing, and the heart rate increases substantially. - Not enough vasoconstriction:
For someone with reduced vasoconstriction in the legs, the heart rate increases dramatically to maintain normal blood pressure. This increase could be worsened in high-heat conditions, causing more blood to flow to the skin.[ref]
Theories on the underlying causes of POTS:
Research points to several theories on what causes POTS, and it is likely that the individual causes of the symptoms can be different for different people.
| Mechanism | Description |
|---|---|
| Hypovolemia | Low blood volume leads to higher baseline/resting HR and even greater HR increase upon standing |
| Reduced Vasoconstriction | Blood vessels don’t constrict enough, leading to blood pooling and compensatory HR increase |
| RAAS Dysfunction | ↑ Angiotensin II, poor sodium retention, altered blood volume |
| Vasoconstriction Failure | Insufficient SNS-induced vasoconstriction in lower limbs; blood pools in legs |
| Hyperadrenergic State | Excess norepinephrine released or cleared too slowly, resulting in tachycardia, anxiety, etc. |
| NET Dysfunction | SLC6A2 gene/NET malfunction: reduced norepinephrine clearance |
Let’s look at a couple of the different systems that regulate heart rate and blood pressure, and then we will dive into some of the known causes of dysfunction in these systems.
The autonomic nervous system and blood regulation:
POTS is usually classified as a type of dysautonomia. Essentially, dysautonomia means a dysfunction of the autonomic nervous system. It is a catch-all term, with several chronic conditions falling under the umbrella of dysautonomia.[ref]
The autonomic nervous system controls involuntary functions in the body, including heart rate, blood pressure, and the motility of the digestive tract.
Blood pressure is tightly regulated through the renin-angiotensin-aldosterone system (called RAAS). The RAAS system balances out the volume of blood by regulating sodium and water levels. In the kidneys, this system can either increase sodium reabsorption or water reabsorption to alter blood volume in the body. (More water kept in the bloodstream = more blood volume.)[ref]
Angiotensin system and POTS:
For some people, the renin-angiotensin-aldosterone system is involved in POTS. When RAAS dysfunction is involved in causing POTS, there can be an increase in plasma angiotensin II. This increase causes an imbalance in blood volume due to the kidneys not retaining enough sodium. Additionally, some researchers have found that there is inadequate ACE2 activity.[ref][ref]
Vasoconstriction and POTS:
Vasoconstriction is the tightening (constriction) of blood vessels, which increases blood pressure. This is controlled by the sympathetic nervous system.
Some people with POTS have issues with the sympathetic nervous system not working correctly in the feet and legs. When standing up, it can result in insufficient vasoconstriction (e.g., blood vessels are too relaxed). As a result, blood pools in the legs and feet, as well as the abdominal cavity. The lack of vasoconstriction and the pooling of blood then kick the heart into high gear, pumping hard to make up for the lack of blood flow.[ref]
Sympathetic nervous system, NET, medications, and POTS: Hyperadrenergic
One branch of the autonomic nervous system is the sympathetic nervous system (fight-or-flight response).
The activation of the sympathetic nervous system releases norepinephrine. Norepinephrine (aka noradrenaline) functions as a neurotransmitter and a stress hormone in the body.
Adrenergic system and POTS:
A subset of people with POTS have what is known as the ‘hyperadrenergic’ form. When these people stand, their bodies release an excess of norepinephrine. It can cause heart palpitations, tremors, rapid heartbeat, feeling anxious, and increased blood pressure. Some patients with hyperadrenergic POTS also get headaches upon standing.[ref][ref]
Alternatively, instead of releasing excess norepinephrine, the body may not clear out the normal norepinephrine quickly enough. Or both could occur together.
Norepinephrine transporter, autonomic nervous system:
Alterations in the norepinephrine transporter (NET) can cause reduced clearance of norepinephrine, leaving the sympathetic nervous system in a state of excessive activation. Norepinephrine is the primary signaling neurotransmitter in the autonomic nervous system, where it controls heart rate. When norepinephrine is released into the synapse between neurons, the amount of norepinephrine available there is controlled by the norepinephrine transporter, NET.[ref]
In POTS, research points to an excess of norepinephrine signaling, and this could be due to decreased NET activity, essentially allowing too much norepinephrine to remain in the synapse.
Both rare mutations and more common gene variants that code for the norepinephrine transporter (SLC6A2 gene) are linked to POTS. Additionally, common medications can be NET inhibitors, such as some tricyclic antidepressants and certain ADHD medications.[ref]
Medications that impact NET may be helpful in POTS. For example, atomoxetine (Strattera) is an ADHD medication that targets the norepinephrine transporter. In a clinical trial, it has been shown to significantly increase the standing heart rate in POTS patients.[ref] Note that this may not be a good option for everyone. An animal study showed that atomoxetine increased histamine levels (in the brain).[ref]
Epigenetic changes and POTS:
Research points towards epigenetic modification of the SLC6A2 gene as a cause of POTS. Epigenetic changes are ways that genes can be turned off (or on) for transcription. In this case, the researchers think that epigenetic markers decrease the availability of the norepinephrine transporter in POTS.[ref]
Autoimmune involvement in POTS:
Research shows that for some people, POTS can be due to an autoimmune attack on either the adrenergic system or the renin-angiotensin system.[ref][ref] Both systems are important in heart rate and blood flow.
For example, a small study in 2018 found that most patients with POTS in their study had angiotensin II type 1 receptor antibodies (IgG) as well as adrenergic activation, showing an autoimmune activation of that receptor. Interestingly, losartan, a commonly used hypertension medication that acts on the angiotensin II receptor, reduced the receptor activity down to the same levels as the control.[ref]
Another recent study showed that patients with POTS were likely to have autoimmune activity towards the adrenergic receptors (α1 receptor, β2 receptor, cholinergic, and opioid receptor-like 1).[ref] The α1 and β2 adrenergic receptors are activated by epinephrine and norepinephrine, tying back to the norepinephrine disruption of the autonomic nervous system.[ref]
However, not everyone with POTS has autoimmune activity that interacts with the angiotensin receptors or the adrenergic receptors.
So far, we have autonomic dysfunction causing vasoconstriction or alterations to the angiotensin system or adrenergic receptors. But what initiates these changes?
Triggers of POTS:
Researchers have identified several different triggering events that can initiate POTS. Diseases and conditions linked to triggering POTS include:
| Trigger/Condition | Notes |
|---|---|
| Autoimmune activation (adrenergic/RAAS)[ref][ref] | Example: angiotensin II type 1 receptor antibodies[ref] |
| Mast cell activation[ref] | MCAS often overlaps with POTS |
| Viral illness (COVID, West Nile, etc.)[ref][ref] | COVID/long COVID common trigger |
| Vaccines (HPV, mRNA COVID)[ref] | Possible molecular mimicry response; research not consistent |
| Concussions, surgery, neck injury, bed rest[ref][ref] | Often reported as triggers, inflammation involved |
| Inflammatory disorders[ref] | Possible contributing role |
Let’s dig into the research on a few of these triggers:
Can vaccines trigger POTS?
One trigger for POTS for some individuals seems to be certain vaccines given to teens or adults. For example, there are dozens of case reports of POTS shortly following the HPV vaccine. Additionally, several studies followed some women after the HPV shot, finding an increase in dysautonomia and POTS.[ref][ref][ref][ref][ref]
Why would an HPV vaccine cause POTS in a minority of people? Research points to an autoimmune response (adrenergic receptor antibodies) due to molecular mimicry with specific HPV peptides.[ref] There are a lot of unanswered questions on the links between the HPV vaccine and POTS. Epidemiological studies often don’t find a statistical, population-wide link between the introduction of the HPV vaccine and the number of POTS diagnoses.[ref][ref] Additionally, some researchers point to media coverage as a cause for any post-vaccination symptom spikes.[ref]
Does long Covid cause POTS?
Interestingly, one of the mechanisms that causes POTS is a disturbance in the renin-angiotensin system, possibly due to inadequate ACE2 activity, which is one of the receptors used by the SARS-CoV-2 virus in causing Covid.[ref][ref][ref] POTS is a common autonomic disorder following Covid and in long Covid.[ref][ref]
Some researchers point to SARS-CoV-2 invading the central nervous system in POTS, while others point to the changes in endothelial function. Yet other research shows that neuroinflammation may be involved in POTS after a Covid infection.[ref][ref] Multiple case studies also report a new onset of POTS following the Covid mRNA vaccine.[ref][ref][ref]
Related article: Long Covid: Genetics, Multiple Causes, and Possible Solutions
Cervical spine injury in POTS:
Injury to the neck or cervical spine can lead to autonomic dysfunction and POTS. This is an uncommon cause, but multiple case studies show the connection.[ref][ref]
Overlapping conditions: POTS + hEDS + MCAS + ME/CFS
POTS is a syndrome – meaning a collection of symptoms – rather than a specific disease. Thus, multiple diseases or chronic conditions can cause POTS symptoms. While POTS can be a stand-alone diagnosis, there are several conditions that have a high frequency of overlap with POTS.
| Condition | Description | Overlap with POTS |
|---|---|---|
| hEDS | Hypermobility Ehlers-Danlos Syndrome; joint hypermobility | ~50% of teens with POTS[ref][ref] |
| ME/CFS | Chronic fatigue, PEM, light sensitivity, etc | Subset have POTS HR response[ref] |
| MCAS | Mast cell overactivity and histamine release; promotes wide range of symptoms | High overlap—up to 66% in some studies.[ref] |
Is POTS Genetic or Inherited?
The big question with POTS, especially in long Covid or other post-viral illnesses, is why some people get it and others do not.
Research shows that certain genetic variants increase the susceptibility (or risk) to POTS. There isn’t a single genetic mutation that causes POTS. Rather, POTS is a syndrome that can have its genetic roots in various genes. Understanding where your genetic susceptibility lies may help you find your best treatment options.
| Gene | Function/Pathway | Key Variant(s) | Effect on POTS Risk/Symptoms | Data Availability in Consumer Tests |
|---|---|---|---|---|
| HLA-DQB1*0609 | Immune system/self-recognition | *0609 | 8x increased risk | Not available |
| SLC6A2 | Norepinephrine transporter | rs5569 | Increased susceptibility | Available |
| GNB3 | Signal transduction, blood pressure | C825T | Alters heart rate response | Available |
| NOS3 | Nitric oxide synthesis, vasodilation | rs2070744, others | Some protective, some increase risk | Some available |
Genetic mutations linked to POTS:
For many people, an autoimmune condition (adrenergic system, angiotensin II receptor) triggers POTS, and genetic variants can increase the risk of certain autoimmune conditions. The HLA genes code for an important part of our adaptive immune system. They help the body understand what is foreign (bacteria, viruses) and needs to be attacked. They also help the body understand which tissue is ‘self’ and should be left alone by the immune system. One POTS risk factor is an HLA variant that is not included in 23andMe or AncestryDNA data. A study in 2019 identified the HLA-DQB1*0609 serotype as increasing the risk of POTS by over 8-fold.[ref]
Other genetic variants increase susceptibility to POTS in different ways. The genes that have been identified so far by researchers include nitric oxide genes, norepinephrine (noradrenaline) transporters, and the beta2-adrenergic receptor – all of which impact blood volume regulation. These are all included in the Genotype Report section below.
Sympathetic nervous system:
The SLC6A2 gene codes for a norepinephrine transporter (NET), which removes norepinephrine from the junction between sympathetic nerves. Norepinephrine transporter impairment is linked to depression, panic disorder, tachycardia, and POTS.[ref] Rare mutations in the SLC6A2 gene have been strongly linked to POTS, and a more common variant increases susceptibility a little bit.
The GNB3 gene codes for part of a G-protein that is involved in signal transduction. It can impact a lot of systems in the body, including metabolism and blood pressure. Specifically, a common variant in GNB2 has been shown to interact with the vagus nerve in regulating blood pressure in pots. This variant causes “enhanced vagal withdrawal of the heart” in relation to POTS.[ref]
Vasoconstriction of blood vessels:
The NOS3 gene codes for endothelial nitric oxide (NO) synthase enzyme, which is essential for relaxing blood vessels. Nitric oxide production in the lining of the blood vessels helps to regulate blood flow. A common genetic variant in the NOS3 gene is protective against POTS. Other NOS3 variants, not included in 23andMe or AncestryDNA data, are also linked to POTS.
Epigenetics and POTS:
Epigenetics refers to alterations in how the genetic code is turned on or off for translation. In a nutshell, the nucleus of every cell contains the complete genome, but only certain genes remain essential for that cell to function. Epigenetic markers control how often a gene is translated into its protein.
Studies showed that people with POTS were more likely to have epigenetic modifications that reduced the function of the norepinephrine transporter (SLC6A2 gene).[ref][ref] This goes hand-in-hand with the research showing that people with genetic variants that reduce the function of the SLC6A2 gene are also more susceptible to POTS.
Genotype Report: POTS
Research shows that genetic variants can increase – or decrease – the likelihood of developing POTS. These genes do not cause POTS by themselves. Instead, the variants increase susceptibility, which, when combined with environmental triggers, may lead to the onset of the syndrome. Understanding your susceptibility factors may help you to understand which treatments are more likely to work for you.
Access this content:
An active subscription is required to access this content.
Lifehacks for POTS: Diet, supplements, apps, and treatments
Decreasing POTS symptoms:
Research studies show that the following are recommended for POTS, but please talk with your doctor before making any changes – including lifestyle changes.
- Increase your fluid intake:
Hypovolemia, or lower levels of blood volume, may be helped, in part, by increasing the amount of water – or electrolyte sports drink – that you consume.[ref] - Increased sodium intake:
Similar to increasing fluid intake, if your sodium intake is low or your electrolyte balance is off, it can affect blood volume.[ref] Often, people with POTS are encouraged to drink sports drinks such as Gatorade. - Compression socks:
If your blood pools in your lower extremities, compression socks or compression leggings may help. - Thermal comfort wearables:
A study involving POTS patients found that more than half of them had temperature intolerance that was improved significantly with thermal comfort wear.[ref]
Mast cell activation and POTS:
Check out the full article on mast cell activation syndrome, along with the lifehacks section there. Reducing mast cell activation through supplements, such as luteolin or quercetin, may help reduce symptoms.
Specific interventions with genetic connections:
These genetic connections are theoretical, but they may give you a starting point for talking with your doctor or doing more research.
Vagal nerve stimulation:
A clinical trial showed that transcutaneous vagus nerve stimulation significantly reduced POTS symptoms compared to a sham (placebo) treatment.[ref] The GNB3 T allele is associated with an increased risk of POTS and links to vagal nerve stimulation in the heart.
Beta-blockers:
Talk with your doctor about whether beta-blockers (β2 adrenergic receptor antagonists) are an option for you. Clinical trials show that they are helpful for some people with POTS.[ref] The ADRB2 gene codes for the beta2-adrenergic receptor and the SLC6A2 gene encodes the norepinephrine transporter. Both may be relevant for beta-blockers.[ref]
Increase NOS3:
Reduced nitric oxide activity is associated with POTS for some people.[ref] BH4 (tetrahydrobiopterin) is an essential cofactor for nitric oxide production. Your body naturally produces BH4, and under normal conditions, it gets recycled and reused in the cell. But when oxidative stress is high (excess reactive oxygen species), it can be used up more quickly.[ref] Studies show that Vitamin C increases nitric oxide through increasing BH4 bioavailability.[ref]
Curcumin for NOS3?
Similarly, curcumin supplementation has been shown to increase NO by reducing oxidative stress.[ref] A clinical trial of 2,000 mg/day of curcumin had beneficial effects on increasing nitric oxide in relation to endothelial function tests for cardiovascular disease.[ref]
Diet and Supplements for POTS Syndrome:
The recommendations and studies seem to vary a lot on specific dietary interventions. For example, some caffeine constricts the blood vessels and can be helpful in certain situations, but caffeine worsens POTS for others.
Overall, a healthy diet with fresh vegetables and fruits, fish, and/or grass-fed meat should help meet your needs for vitamins.
Low iron or low vitamin D levels are also linked to an increased risk for POTS in teens.[ref][ref] It is easy to check both your iron and vitamin D levels with a quick blood test. You can order tests online (e.g., UltaLab tests) or through your doctor.
Caution with exercise:
People with POTS are often ‘deconditioned’ due to fatigue and not being able to exercise. Researchers recommend mild to moderate exercise for short periods several times a week to reverse the deconditioning.[ref] Talk with your doctor to figure out a plan that will work best for you.
POTS and Heart Rate Apps:
Several different heart rate apps are available for both Android and iPhone. Some apps, such as Cardiogram, integrate with Apple Watch to track heart rate. The Instant Heart Rate app for the iPhone is easy to use and has good ratings.
Vagus Nerve Stimulation:
A 2023 randomized clinical trial found that ‘transcutaneous vagus nerve stimulation’ via a device attached to the ear was effective in treating POTS symptoms. [ref] In 2025, a clinical trial in long covid patients with POTS confirmed these results and found that vagus nerve stimulation was an effective treatment for POTS and that the results persisted for a year following treatment. [ref]
Talk with your doctor about:
An angiotensin II receptor blocker, Losartan, may also be something to talk with your doctor about. Several studies show that it could be effective for some individuals with POTS if they have autoimmune activation of the angiotensin II receptor.[ref][ref][ref]
Natural Supplements for POTS:
Get enough B12:
One study noted that teens with POTS were about three times more likely to have low vitamin B12 levels.[ref] Good food sources of B12 include liver, fish, meat, poultry, eggs, and dairy products. If you are planning to supplement with methylB12, be sure to check your COMT gene first. People with slow COMT may want to choose a different type of B12.
Thiamine for POTS:
Access this content:
An active subscription is required to access this content.
Frequently asked questions
Is POTS genetic?
POTS appears to have both genetic and environmental components, with several genes involved in immune function, autonomic regulation, and connective tissue linked to increased risk.
Which genes are associated with POTS?
Research has implicated HLA genes, the norepinephrine transporter gene SLC6A2, GNB3, NOS3, and genes related to connective tissue and mast cell activation in people with POTS.
Can Covid-19 or other infections trigger POTS?
Viral infections, including Covid-19, are commonly reported triggers for new-onset POTS, likely through effects on the immune system, autonomic nerves, and blood vessel regulation.
Are vaccines linked to POTS?
There are case reports of POTS following vaccination, but these events are rare, and large studies have not shown a clear overall increase in risk compared to background rates.
What conditions commonly overlap with POTS?
POTS often overlaps with hypermobile Ehlers–Danlos syndrome, mast cell activation, autoimmune thyroid diseases, and gastrointestinal dysmotility or irritable bowel-type symptoms.
How is POTS treated or managed?
Management usually combines non-pharmaceutical strategies such as increased fluids and salt, compression garments, and exercise, along with medications tailored to the patient’s POTS subtype.
Related Articles and Topics:
Histamine Intolerance: Genetic Report, Supplements, and Real Solutions
HLA-B27: Genetic Variant That Increases Susceptibility to Autoimmune Diseases
References:
Anjum, Ibrar, et al. “Postural Orthostatic Tachycardia Syndrome and Its Unusual Presenting Complaints in Women: A Literature Minireview.” Cureus, vol. 10, no. 4, p. e2435. PubMed Central, https://doi.org/10.7759/cureus.2435. Accessed 12 Apr. 2022.
Bayles, Richard, et al. “Epigenetic Modification of the Norepinephrine Transporter Gene in Postural Tachycardia Syndrome.” Arteriosclerosis, Thrombosis, and Vascular Biology, vol. 32, no. 8, Aug. 2012, pp. 1910–16. PubMed, https://doi.org/10.1161/ATVBAHA.111.244343.
Blitshteyn, Svetlana, et al. “Autonomic Dysfunction and HPV Immunization: An Overview.” Immunologic Research, vol. 66, no. 6, Dec. 2018, pp. 744–54. PubMed, https://doi.org/10.1007/s12026-018-9036-1.
Bonamichi-Santos, Rafael, et al. “Association of Postural Tachycardia Syndrome and Ehlers-Danlos Syndrome with Mast Cell Activation Disorders.” Immunology and Allergy Clinics of North America, vol. 38, no. 3, Aug. 2018, pp. 497–504. PubMed, https://doi.org/10.1016/j.iac.2018.04.004.
Boris, Jeffrey R., and Thomas Bernadzikowski. “Demographics of a Large Paediatric Postural Orthostatic Tachycardia Syndrome Program.” Cardiology in the Young, vol. 28, no. 5, May 2018, pp. 668–74. PubMed, https://doi.org/10.1017/S1047951117002888.
Brinth, Louise, et al. “Suspected Side Effects to the Quadrivalent Human Papilloma Vaccine.” Danish Medical Journal, vol. 62, no. 4, Apr. 2015, p. A5064.
Brinth, Louise S., et al. “Orthostatic Intolerance and Postural Tachycardia Syndrome as Suspected Adverse Effects of Vaccination against Human Papilloma Virus.” Vaccine, vol. 33, no. 22, May 2015, pp. 2602–05. PubMed, https://doi.org/10.1016/j.vaccine.2015.03.098.
Bryarly, Meredith, et al. “Postural Orthostatic Tachycardia Syndrome: JACC Focus Seminar.” Journal of the American College of Cardiology, vol. 73, no. 10, Mar. 2019, pp. 1207–28. ScienceDirect, https://doi.org/10.1016/j.jacc.2018.11.059.
Chandler, Rebecca E., et al. “Current Safety Concerns with Human Papillomavirus Vaccine: A Cluster Analysis of Reports in VigiBase®.” Drug Safety, vol. 40, no. 1, Jan. 2017, pp. 81–90. PubMed, https://doi.org/10.1007/s40264-016-0456-3.
Dysautonomia International: Dysautonomia Awareness, Dysautonomia Advocacy, Dysautonomia Advancement. http://www.dysautonomiainternational.org/. Accessed 12 Apr. 2022.
Dysautonomia International: Postural Orthostatic Tachycardia Syndrome. http://www.dysautonomiainternational.org/page.php?ID=30. Accessed 12 Apr. 2022.
Fedorowski, Artur, et al. “Antiadrenergic Autoimmunity in Postural Tachycardia Syndrome.” Europace: European Pacing, Arrhythmias, and Cardiac Electrophysiology: Journal of the Working Groups on Cardiac Pacing, Arrhythmias, and Cardiac Cellular Electrophysiology of the European Society of Cardiology, vol. 19, no. 7, July 2017, pp. 1211–19. PubMed, https://doi.org/10.1093/europace/euw154.
Fountain, John H., and Sarah L. Lappin. “Physiology, Renin Angiotensin System.” StatPearls, StatPearls Publishing, 2022. PubMed, http://www.ncbi.nlm.nih.gov/books/NBK470410/.
Garland, Emily M., et al. “Endothelial NO Synthase Polymorphisms and Postural Tachycardia Syndrome.” Hypertension (Dallas, Tex.: 1979), vol. 46, no. 5, Nov. 2005, pp. 1103–10. PubMed, https://doi.org/10.1161/01.HYP.0000185462.08685.da.
Green, Elizabeth A., et al. “Effects of Norepinephrine Reuptake Inhibition on Postural Tachycardia Syndrome.” Journal of the American Heart Association, vol. 2, no. 5, Sept. 2013, p. e000395. DOI.org (Crossref), https://doi.org/10.1161/JAHA.113.000395.
Ikeda, Shu-Ichi, et al. “Suspected Adverse Effects after Human Papillomavirus Vaccination: A Temporal Relationship.” Immunologic Research, vol. 66, no. 6, Dec. 2018, pp. 723–25. PubMed, https://doi.org/10.1007/s12026-018-9063-y.
Kanjwal, Khalil, et al. “Clinical Presentation and Management of Patients with Hyperadrenergic Postural Orthostatic Tachycardia Syndrome. A Single Center Experience.” Cardiology Journal, vol. 18, no. 5, 2011, pp. 527–31. PubMed, https://doi.org/10.5603/cj.2011.0008.
Khan, Abdul Waheed, et al. “NET Silencing by Let-7i in Postural Tachycardia Syndrome.” JCI Insight, vol. 2, no. 6, Mar. 2017, p. e90183. PubMed, https://doi.org/10.1172/jci.insight.90183.
Li, Hongliang, et al. “Autoimmune Basis for Postural Tachycardia Syndrome.” Journal of the American Heart Association, vol. 3, no. 1, Feb. 2014, p. e000755. PubMed, https://doi.org/10.1161/JAHA.113.000755.
Liu, Yonghong, et al. “Postural Orthostatic Tachycardia Syndrome Presenting With Recurrent Syncope After Cervical Spinal Cord Injury.” Clinical Case Reports, vol. 14, no. 3, Mar. 2026, p. e72181. PubMed, https://doi.org/10.1002/ccr3.72181.
Marques, F. Z., et al. “A Polymorphism in the Norepinephrine Transporter Gene Is Associated with Affective and Cardiovascular Disease through a MicroRNA Mechanism.” Molecular Psychiatry, vol. 22, no. 1, Jan. 2017, pp. 134–41. PubMed, https://doi.org/10.1038/mp.2016.40.
Miglis, Mitchell G., et al. “Use of a Thermal Comfort Wearable Improves Temperature Intolerance in Patients with Postural Tachycardia Syndrome.” Autonomic Neuroscience: Basic & Clinical, vol. 264, Feb. 2026, p. 103398. PubMed, https://doi.org/10.1016/j.autneu.2026.103398.
Miranda, Nicole A., et al. “Activity and Exercise Intolerance After Concussion: Identification and Management of Postural Orthostatic Tachycardia Syndrome.” Journal of Neurologic Physical Therapy, vol. 42, no. 3, July 2018, pp. 163–71. PubMed Central, https://doi.org/10.1097/NPT.0000000000000231.
Müller, Katharina, et al. “Impaired Attention and Cognitive Deficits Associated with Pain and Autonomic Symptoms in Hypermobile Ehlers-Danlos Syndrome: A Pilot Study.” Clinical Autonomic Research: Official Journal of the Clinical Autonomic Research Society, Mar. 2026. PubMed, https://doi.org/10.1007/s10286-026-01191-7.
Mustafa, Hossam I., et al. “Abnormalities of Angiotensin Regulation in Postural Tachycardia Syndrome.” Heart Rhythm, vol. 8, no. 3, Mar. 2011, pp. 422–28. PubMed, https://doi.org/10.1016/j.hrthm.2010.11.009.
Nakao, Ryota, et al. “GNB3 C825T Polymorphism Is Associated with Postural Tachycardia Syndrome in Children.” Pediatrics International: Official Journal of the Japan Pediatric Society, vol. 54, no. 6, Dec. 2012, pp. 829–37. PubMed, https://doi.org/10.1111/j.1442-200X.2012.03707.x.
Raj, Satish R. “The Postural Tachycardia Syndrome (POTS): Pathophysiology, Diagnosis & Management.” Indian Pacing and Electrophysiology Journal, vol. 6, no. 2, Apr. 2006, pp. 84–99. PubMed Central, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1501099/.
Segal, Yahel, and Yehuda Shoenfeld. “Vaccine-Induced Autoimmunity: The Role of Molecular Mimicry and Immune Crossreaction.” Cellular and Molecular Immunology, vol. 15, no. 6, June 2018, pp. 586–94. PubMed Central, https://doi.org/10.1038/cmi.2017.151.
Shibao, Cyndya, et al. “Hyperadrenergic Postural Tachycardia Syndrome in Mast Cell Activation Disorders.” Hypertension (Dallas, Tex.: 1979), vol. 45, no. 3, Mar. 2005, pp. 385–90. PubMed, https://doi.org/10.1161/01.HYP.0000158259.68614.40.
Skufca, J., et al. “Incidence Rates of Guillain Barré (GBS), Chronic Fatigue/Systemic Exertion Intolerance Disease (CFS/SEID) and Postural Orthostatic Tachycardia Syndrome (POTS) Prior to Introduction of Human Papilloma Virus (HPV) Vaccination among Adolescent Girls in Finland, 2002–2012.” Papillomavirus Research, vol. 3, Mar. 2017, pp. 91–96. PubMed Central, https://doi.org/10.1016/j.pvr.2017.03.001.
Stewart, Julian M., et al. “Defects in Cutaneous Angiotensin-Converting Enzyme 2 and Angiotensin-(1-7) Production in Postural Tachycardia Syndrome.” Hypertension, vol. 53, no. 5, May 2009, pp. 767–74. ahajournals.org (Atypon), https://doi.org/10.1161/HYPERTENSIONAHA.108.127357.
Thomsen, Reimar Wernich, et al. “Hospital Records of Pain, Fatigue, or Circulatory Symptoms in Girls Exposed to Human Papillomavirus Vaccination: Cohort, Self-Controlled Case Series, and Population Time Trend Studies.” American Journal of Epidemiology, vol. 189, no. 4, Apr. 2020, pp. 277–85. PubMed Central, https://doi.org/10.1093/aje/kwz284.
Vojdani, Aristo, and Datis Kharrazian. “Potential Antigenic Cross-Reactivity between SARS-CoV-2 and Human Tissue with a Possible Link to an Increase in Autoimmune Diseases.” Clinical Immunology (Orlando, Fla.), vol. 217, Aug. 2020, p. 108480. PubMed Central, https://doi.org/10.1016/j.clim.2020.108480.
Yadav, Aditi, et al. “Postural Orthostatic Tachycardia Syndrome in Spinal Cord Injury.” Cureus, vol. 15, no. 6, p. e41124. PubMed Central, https://doi.org/10.7759/cureus.41124. Accessed 7 Mar. 2026.
Yu, Xichun, et al. “Angiotensin II Type 1 Receptor Autoantibodies in Postural Tachycardia Syndrome.” Journal of the American Heart Association, vol. 7, no. 8, Apr. 2018, p. e008351. PubMed, https://doi.org/10.1161/JAHA.117.008351.
Zhang, Qingyou, et al. “Clinical Features of Hyperadrenergic Postural Tachycardia Syndrome in Children: Hyperadrenergic POTS.” Pediatrics International, vol. 56, no. 6, Dec. 2014, pp. 813–16. DOI.org (Crossref), https://doi.org/10.1111/ped.12392.